17
Otitis Media
Nathan Sautter and Keiko Hirose
Treatment of children and adults with otitis media (OM) is a substantial component of the practice of otolaryngology—head and neck surgery. OM is second only to viral upper respiratory tract infection as the most common reason for visits to pediatricians. In a prospective study of children in the greater Boston area, 93% of children had one or more episode of acute otitis media (AOM) and 74% had at least three episodes of AOM during the first 7 years of life.1 Epidemiologic studies at the University of Pittsburgh revealed a 90% incidence of OM in urban children within the first 2 years of life.2 The vast majority of OM occur in children, and although the greater part of these cases resolves by adolescence, the disease may also occur in adults. Tympanostomy with tube placement is the primary surgical treatment for OM and remains the most common surgical procedure performed under general anesthesia in the United States.
In spite of substantial research, controversy exists regarding optimal therapy and treatment guidelines, which vary widely across the United States and in other nations. The large annual health care expenditure in the United States for treatment of OM, estimated at more than $5 billion, has caused health care providers and payers to adopt several tactics aimed at reducing costs.
The first attempt at development of an evidence-based and cost-effective clinical treatment guideline for OM was introduced by the Agency for Health Care Policy and Research (AHCPR)3 in 1994. These recommendations were updated in 2004 by the Subcommittee on Otitis Media with Effusion, composed of experts selected by the American Academy of Otolaryngology—Head and Neck Surgery in conjunction with the American Academy of Pediatrics and the American Academy of Family Physicians.4 These recommendations apply to children aged 2 months to 12 years without developmental disabilities or an underlying medical condition causing predisposition to OM. The evidence-based guidelines proposed by the Subcommittee are intended to maximize efficiency of health care expenditures while maintaining the highest standard of care. They are intended as a guide, not a substitute, for clinical judgment.
 Terminology
Terminology
OM is an infectious, inflammatory condition of the middle ear associated with an effusion behind an intact tympanic membrane. It also may be associated with several inciting factors, most commonly upper respiratory tract infection and eustachian tube dysfunction. OM may be classified according to the composition of the effusion: serous (SOM), mucoid (MOM), and purulent (POM). Each classification is not in itself a distinct entity; that is, a specific etiology may not be inferred by the composition of the effusion. Rather, OM is a dynamic process encompassing a somewhat broad spectrum of disease. For example, SOM may progress to MOM, and POM typically progresses to SOM during the course of resolution.
Acute otitis media usually is characterized by rapid onset of otalgia and erythema of the tympanic membrane in the presence of middle ear effusion. Otalgia and fever are more evident in younger children and may be absent in older children. Erythema of the tympanic membrane without middle ear effusion is called acute myringitis and may be mistaken for AOM. Erythema of the tympanic membrane also is seen in a small proportion of chronic middle ear effusions. Recurrent acute otitis media (RAOM) is a term commonly assigned to patients with multiple self-limited episodes of AOM punctuated by periods of time in which the patient is symptom-free, either with or without a persistent middle ear effusion. Current practice guidelines define RAOM as three or more episodes of AOM within 6 months, or four or more episodes within a year.
Chronic otitis media with effusion (COME) refers to an accumulation of inflammatory liquid in the middle ear cleft without other signs of inflammation (e.g., fever, otalgia). Alternative names assigned to COME in the literature include glue ear, chronic secretory otitis media, serous otitis media, persistent otitis media, and silent otitis media. Effusions resulting from barotrauma, skull fracture, or leakage of cerebrospinal fluid are not included in this category. Many, if not most, chronic effusions result from resolving AOM. It is appropriate to think of AOM and COME as two points along a continuum of the same disease process. An antecedent history of AOM, however, is not necessary for diagnosis of COME. Chronic effusions of unknown duration as well as all cases of OM persisting more than 30 days are classified as COME. Chronic otorrhea through a perforated tympanic membrane is termed chronic suppurative otitis media and may be associated with cholesteatoma.
 Risk Factors
Risk Factors
Known risk factors for OM include recent upper respiratory infection (URI), male gender, a greater number of siblings in the home, attending day care, family history, bottle (rather than breast) feeding, and smoking in the home.5 Familial susceptibility to OM may result from defects in the immune system or in the biologic structure of the middle ear, palate, and eustachian tube. Inherited disorders of mucociliary clearance such as Kartagener’s syndrome or cystic fibrosis may predispose to OM. Socioeconomic factors within a family that may contribute to the development of OM include overcrowding, poor diet, and lack of access to health care. Certain ethnic groups, notably Native Americans, have a high prevalence of OM presumably due to differences in the anatomy of the eustachian tube and skull base.6 Children in day care have a greater incidence of OM than those reared at home because of endemic URI in groups. Similarly, children in early grade school have an increased incidence of OM. Some authors have also suggested that environmental allergy, food allergy, and gastroesophageal reflux disease contribute to the development of OM, although a definite link between any of these conditions and OM has yet to be established.7,8
 Pathophysiology
Pathophysiology
Acute Otitis Media
AOM is an inflammatory disorder induced by microorganisms in the middle ear (Figs. 17–1 through 17–6) characterized by the presence of an effusion (often purulent), and frequently associated with otalgia and fevers, particularly in younger children. The most likely route of bacterial entry into the middle ear is from reflux of infected secretions from the nasopharynx through the eustachian tube, which is known to occur in otitis-prone children. Three factors appear to facilitate bacterial reflux into the middle ear: bacterial colonization of the nasopharynx, incompetence of the protective function of the eustachian tube, and a negative pressure in the middle ear in relation to the nasopharynx.
AOM is principally a sequel of viral URI.9,10 Viral rhinitis breaks down mucosal barriers and mechanisms of mucociliary clearance that prevent bacterial adherence and growth in both the nose and nasopharynx. In addition, swelling of the nasal mucosa alters the aerodynamics of the upper airway.
The bacteriology of AOM has been studied extensively. In a large study at the University of Pittsburgh, the most predominant bacteria cultured from acute middle ear effusions were Streptococcus pneumoniae (35%), Haemophilus influenzae (23%) and Moraxella catarrhalis (14%).11 These same three pathogenic bacteria appear in the nasopharynx following an URI.12,13 Pillsbury et al14 demonstrated higher bacterial colony counts in the adenoids of children with recurrent OM than in those undergoing adenoidectomy for adenoid hypertrophy without OM. Bernstein et al15 have identified the alterations in the microecology of the nasopharynx in children with RAOM. It would appear that loss of the immunologic function of the adenoid and colonization of the nasopharynx with pathogenic bacteria are important factors in the pathophysiology of OM. Normalization of the nasopharyngeal flora in conjunction with a marked decrease in the colonization of pathogenic bacteria has been demonstrated following adenoidectomy in otitis-prone children.13
Eustachian tube dysfunction is generally held to be an underlying cause of OM. However, controversy remains about whether eustachian tube dysfunction causes or is a result of OM. The eustachian tube has three functions: protection of the middle ear, clearance of middle ear secretions, and equalization of pressure between the nose and middle ear. The child’s eustachian tube is short, horizontal, and composed of relatively flaccid cartilage. The protective function of the tube is less effective, and retrograde reflux of nasopharyngeal secretions may occur more readily than in the mature eustachian tube.16
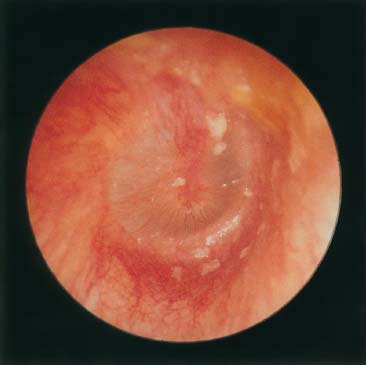
Figure 17–1 Acute otitis media.
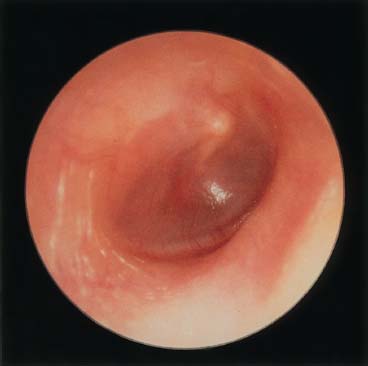
Figure 17–2 Acute otitis media with effusion.
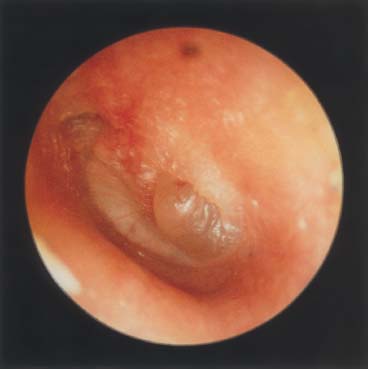
Figure 17–3 Acute otitis media with bulging drum.
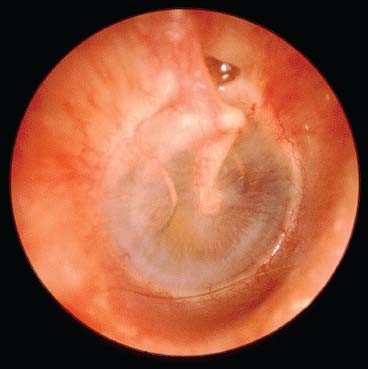
Figure 17–4 Acute otitis media, exudative stage.
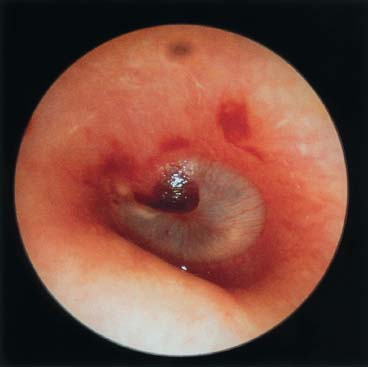
Figure 17–5 Acute otitis media with bleb.
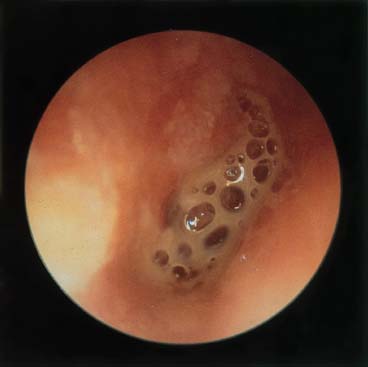
Figure 17–6 Acute otitis media, suppurative stage.
Clearance of secretions results mainly from ciliary action. Viral upper respiratory infection is known to cause transient ciliary dysfunction; it is presumed that ciliary function of the middle ear and eustachian tube mucosa is also impaired during AOM. Fluid accumulates as a consequence of ciliary paralysis, and clearance of fluid follows recovery of ciliary function. However, thick viscous fluid may occlude the tube secondarily because of its rheologic properties. Pressure equalization is normally mediated by tubal opening from contraction of the tensor veli palatini muscle in response to stimuli mediated by the tympanic plexus.17 Normal function of the tensor veli palatini muscle is impaired in patients with submucous or complete cleft palate and is thought to be the main reason for the resultant eustachian tube dysfunction.
The third factor in the genesis of AOM may result from either excessive negative pressure in the middle ear or positive pressure in the nasopharynx. Obstruction of the nose secondary to viral rhinitis may result in the equivalent of the Toynbee maneuver, that is, holding the nostrils closed during swallowing. Because most URIs result in nasal obstruction, swallowing during an URI may increase the nasopharyngeal pressure, which will open the tube and tend to push secretions into the middle ear space. Nose blowing also increases nasopharyngeal pressures. Sniffing is a common symptom of URI and it is known to produce negative middle ear pressure, which may facilitate the reflux process by creating a negative pressure differential that would pull on any material entering the eustachian tube.18
Chronic Otitis Media with Effusion
COME was virtually unknown before antimicrobial therapy began in the 1940s (Figs. 17–7 through 17–10). Whether this is the result of widespread use of antibiotics or better diagnostic methods is unclear. However, perforations of the tympanic membrane are less prevalent after antimicrobial therapy than in the preantibiotic era. Such perforations permitted adequate drainage of the ear and prevented COME.
Early researchers postulated that COME was a primary disorder of multiple causes resulting from the hydrops ex vacuo theory; that is, eustachian tube obstruction resulted in, sequentially, under-aeration of the middle ear, negative middle ear pressure, and fluid transudation.19 When the drop in pressure is acute, these observations adequately explain the development of effusion as in barotrauma.20 However, applying this concept to the pathogenesis of COME does not fully explain more recent observations about the disorder. The Greater Boston Collaborative Otitis Media Study found persistent effusion following AOM for 1 month in 40%, for 2 months in 20%, and for 3 months or longer in 10% of cases.21 More recently, Rosenfeld and Kay22 performed a literature search and meta-analysis of the literature to estimate the natural history of untreated OM. In patients with middle ear effusion following an untreated episode of AOM, 59% resolved within 1 month and 74% resolved within 3 months. Patients with chronic middle ear effusions (observed to be present for longer than 3 months) exhibited 26% resolution within 6 months and 33% resolution by 1 year. Many investigators have demonstrated pathogenic bacteria in the fluid obtained from the middle ears of children with COME.23 The Pittsburgh Otitis Media Research Center study confirmed similar bacteriology in cultured effusions from children with AOM and COME. Children with chronic effusion had a positive culture 70% of the time.2 In one study, reverse-transcriptase polymerase chain reaction (RT-PCR) analysis of culture-negative chronic effusions in children revealed the presence of bacterial messenger RNA (mRNA) (hence, viable and metabolically active bacteria) in a significant proportion of effusions.24 It is possible that, in many chronic effusions, bacteria may be present in an attenuated form, existing as biofilms on the mucosal surface. This was demonstrated by Ehrlich et al25 in a chinchilla model of OM using H. influenzae. Studies in experimental OM26 have demonstrated that eustachian tube obstruction does not result in mucoid effusion in the absence of bacterial infection. Normal middle ear gas is now known to be hypoxic and hypercapneic in relation to inspired air,27 and chronic eustachian tube obstruction per se does not result in severe negative middle ear pressure.28
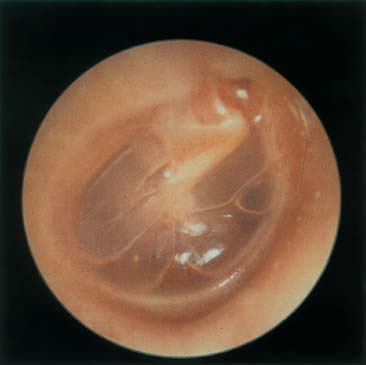
Figure 17–7 Serous otitis media.
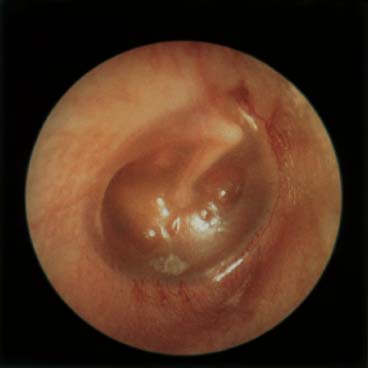
Figure 17–8 Serous otitis media with retracted drum.
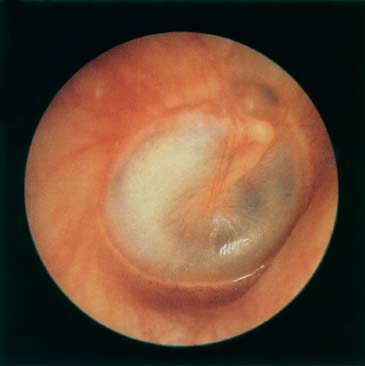
Figure 17–9 Serous otitis media, advanced.
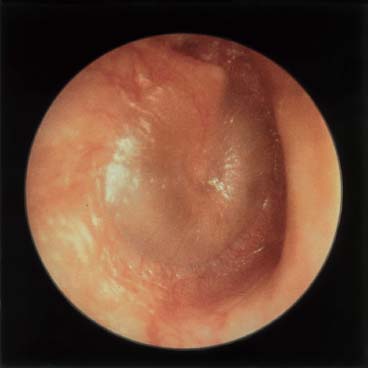
Figure 17–10 Mucoid otitis media.
Thus, the preponderance of available evidence supports the theory that (1) secretory changes in the middle ear in COME are histologic sequelae of chronic infection, rather than a separate pathologic disorder; (2) the majority of cases of COME begin as acute infection of the middle ear; (3) postinflammatory alterations in the middle ear mucosa and eustachian tube (e.g., goblet cell metaplasia and hypersecretion) lead to persistence of effusion; and (4) dysfunction of the eustachian tube is an important part of the process.
 Diagnosis
Diagnosis
The diagnosis of OM is made primarily on a clinical basis. Symptoms such as fever, otalgia, otorrhea, irritability, and decreased hearing should raise suspicion for the presence of OM. In patients with a previous history of OM or with a given history of a preceding URI, the likelihood of middle ear effusion is even greater. COME may be asymptomatic and often is detected incidentally during well-child visits to the pediatrician. Direct visualization of the tympanic membrane with an otoscope is necessary for confirmation of the diagnosis. An erythematous, opaque, and bulging tympanic membrane is consistent with AOM. Clear or amber-colored fluid may be seen with COME; bubbles or an air-fluid level may also be visible behind the tympanic membrane. Pneumatic otoscopy is useful if the diagnosis is in doubt; decreased TM mobility with pneumotoscopy may indicate the presence of fluid in the middle ear. Tympanometry, acoustic reflectometry, and ultrasonography may be useful adjuncts in the diagnosis of OM.29,30 Tympanometry combined with otoscopy has been demonstrated to raise the sensitivity and specificity of the diagnosis of OM to greater than 90%,31 whereas pneumatic otoscopy alone has been shown to have 85% sensitivity and 75% specificity in the diagnosis of OM, although this may vary based on the individual clinician’s training and experience.32 Accordingly, the official recommendation of the Subcommittee on Otitis Media with Effusion is the use of pneumatic otoscopy as the primary diagnostic method for OM with tympanometry reserved as a confirmatory test when the diagnosis is in doubt.4 In the absence of other otologic abnormalities, audiometry may demonstrate a mild or moderate (20–40 dB) conductive hearing loss but is not useful for the initial diagnosis of OM. It is important to document the laterality and duration of the effusion.
The history should also include questions regarding the risk factors for learning and speech disabilities, duration of symptoms, quality of life issues, and the presence of the aforementioned environmental risk factors.
The incidence of OM in adults is much less common than in children. In the absence of an obvious inciting factor (e.g., viral URI, barotrauma) a careful search for the underlying etiology should be undertaken. An adult with unilateral middle ear effusion should undergo evaluation for a nasopharyngeal mass, including nasopharyngoscopy.
 Medical Management
Medical Management
Acute Otitis Media
Clearance of an acute episode of OM usually follows control of the infecting organism through immunologic defense. Most clinicians, however, agree that antimicrobial therapy hastens clearance. The majority of cases are due to aerobic organisms with S. pneumoniae, H. influenzae, and M. catarrhalis—the three most commonly isolated organisms in acute middle ear effusions. Routine culture of the middle ear contents is not done in practice. Tympanocentesis may provide some information regarding the composition and cause of the effusion, but it is not routinely performed and is not considered a therapeutic procedure for any form of OM. It should be reserved for cases in which the clinician suspects a rare or resistant bacterial etiology, or in newborns or immunocompromised patients with OM. In cases of AOM with negative cultures, either the causative agent (virus, anaerobes) is uncertain or the diagnosis is wrong.
Traditional therapy in the United States is amoxicillin 40 mg/kg/day for 7 to 10 days.33 Although shorter courses probably are adequate, the evidence is incomplete. The principal reasons for use of an antimicrobial are to lower the risk of meningitis in babies and shorten the duration of symptoms in a minority of patients. In a meta-analysis of 5400 children with AOM, Rosenfeld et al33 observed clinical resolution within 7 to 14 days in 81% of untreated children and in 94% of children treated with antibiotics. These findings are similar to those in a later meta-analysis.34 Current practice conventions suggest that treatment with another agent, such as amoxicillin clavulanate, is indicated when symptoms fail to resolve after 3 days of treatment. These conventions should be studied in more detail because in some cases the fever is due to a systemic viral infection, and the redness of the tympanic membrane in a crying infant is mistakenly used as evidence of AOM. In such cases, lack of effusion in the middle ear should be considered as clinical evidence against a diagnosis of AOM, and a second antimicrobial agent, therefore, would not be indicated. Further, presence of effusion even up to 3 months after an episode of AOM is not unusual and is not, in itself, an indication for a second or later round of antimicrobial therapy. If the clinician is concerned about lack of response to the agent used, cultures of the middle ear via tympanocentesis is a time-honored method for clarifying the pathogenesis of the signs and symptoms. The role of antimicrobial treatment for AOM is being studied closely, but there is certainly no consensus regarding optimal treatment.
Alternative agents are higher-dose amoxicillin (80–90 mg/kg/day); the second- and third-generation cephalosporins such as cefaclor, cefuroxime, and cefpodoxime; macrolide antibiotics; the combination of sulfisoxazole and erythromycin (Pediazole); or trimethoprim/sulfamethoxazole. Doses are based on body weight.
In an alternative strategy used in many European countries antimicrobial agents are withheld pending spontaneous resolution, which occurs in a majority of cases.22,35 Children are given analgesics and antipyretics; antimicrobials are started if the infection persists after several days or if the tympanic membrane ruptures. Another treatment strategy that is gaining in popularity is “safety net” antibiotic prescriptions.36 Parents of children with AOM are given a prescription for antibiotics and are instructed to fill it only if symptoms persist for 48 hours.
There are no proven preventive measures for AOM other than avoidance of risk factors. A Finnish study of the efficacy of pneumococcal conjugate vaccine (Prevnar) in prevention of AOM in children showed only a modest benefit of 6% reduction in the total number of episodes.37 Although this same vaccine is very effective in preventing invasive pneumococcal infections, its efficacy in prevention of mucosal disease (such as OM) appears to be slight.
Recurrent Acute Otitis Media
The first step in treatment of patients with recurrent or chronic OM should be manipulation of any existing environmental risk factors whenever possible. When this fails, other medical or surgical therapies are considered. Prophylactic medical therapy for RAOM has fallen out of favor. A common practice in the past has been long-term administration of antibiotics for prophylactic purposes. This practice, however, is no longer recommended in the face of rising indices of antibiotic resistance.38 It was estimated in 1995 that approximately 25% of pneumococci were penicillin-resistant, and approximately 25% of H. influenzae isolates and 90% of M. catarrhalis isolates produced ß-lactamase.39 Due to these alarming statistics, some authors have advocated earlier surgical intervention in place of repeated courses of antibiotics as primary treatment of RAOM.40
Chronic Otitis Media with Effusion
Although Mandel et al41
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


