Chapter 13 Ultrasound of the Thyroid and Parathyroid Glands
![]() Please go to expertconsult.com to view related video Ultrasound of the Thyroid and Parathyroid Glands: Equipment and Techniques.
Please go to expertconsult.com to view related video Ultrasound of the Thyroid and Parathyroid Glands: Equipment and Techniques.
Introduction
![]() Please see the Expert Consult website for a brief discussion of the limitations of ultrasound.
Please see the Expert Consult website for a brief discussion of the limitations of ultrasound.
Physics and Principles of Ultrasound
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Sound waves travel relatively slowly in comparison to light and require some form of support medium. When traveling through air, the relative lack of density of the transferring molecules prevents rapid transport and permits attenuation. In contrast, sound waves in fluid travel over long distances with much less degradation depending on their frequency. The process of sono localization in many sea creatures such as dolphins and whales serves both to communicate with others of the same species and to receive feedback information from emitted sounds to locate food. Dolphins and other odontocetes such as toothed whales emit single high-frequency clicks for echolocalization.2 Many other animals, including bats, owls, and elephants, emit and receive sound waves for localization and communication.
Many of the applied concepts have been extracted and simplified from the most commonly referenced text by Kremkau3 titled “Diagnostic Ultrasound, Principles and Instruments.” Sound is transmitted as linked sine waves with peak and trough elements whose height represents amplitude or loudness. A single full cycle is measured from peak to peak or trough to trough, and the number of these cycles per 1-second unit of time represents the frequency. The human ear can recognize sounds as low as 20 Hz and as high as 20,000 Hz or 20 kilohertz (20 kHz). Ultrasound is so named because its frequency emission is well above these frequency levels, in the range of millions of cycles per second or megahertz (MHz). In fact, ultrasound systems used in medicine vary from 3 to 17 MHz with advantages and disadvantages for selected emissions in low, intermediate, or high ranges. Just as a fluid wave along the surface of a body of water is generated from the physical deformation produced by a dropped pebble, an ultrasound wave is transmitted to human tissues through the transducer by physical deformation. One of the key elements of the transducer is the piezoelectric crystal, which elongates and shortens in response to applied alternating electrical current. The actual physical change in these crystals imparts vibrational contact with the skin and deeper tissues to produce a transmitting wave. Special backing materials surrounding the transducer, except for the surface of the footprint that touches the skin, prevent random scatter of the wave energy. This design produces directional transmission. In a linear array transducer where the crystals are oriented such that emitted waves are perpendicular to its footprint, commonly a composite of 128 individual crystals in parallel array will generate the signal. Each crystal vibrates in synchrony with adjacent crystals such that multiple parallel sound waves of equal frequency enter the tissue.
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
The image can be further refined by manipulation after the echoes are returned to the signal processor. Brightness can be adjusted by a turn of the gain control knob. As previously described, ultrasound waves attenuate at greater depth from surface entry. The attenuation is especially problematic when higher frequencies are employed as in thyroid and parathyroid imaging. For example, the deeper aspects of a large goiter (with anterior-posterior dimension of 6 to 7cm or more) will be difficult to see, as these areas are affected by attenuation. These deeper attenuated waves can be selectively recovered with time-gain compensation by increasing the gain of these waves while leaving unaltered the more superficial waves. In this way the overall image has a more even distribution of illumination. Other methodologies such as “sono CT”4 change the way sound waves are delivered from the transducer. Rather than simple linear delivery of sound packets from the center of the transducer, some ultrasound units and transducer combinations have a selected option that sends sound waves in parallel from the edges of the long axis of the transducer. The waves converge at intervals somewhat similar to CT scan configurations to improve image resolution. Electronic noise is an undesirable but unavoidable element in amplification systems. Bandpass filtering can reduce the noise, which eliminates the frequencies above and below the ideal selected frequency. Harmonic imaging is a refinement that observes both the fundamental and second harmonic frequency echo reception. The fundamental frequency is filtered while permitting passage of the second harmonic, a postprocessing method that improves image quality. These are but a few examples of methods that modern ultrasound units have employed to refine image quality to a remarkable degree without any risk to the patient. In fact, even the small, basic portable machines now produce images that were simply unimaginable less than a decade ago.
Artifacts
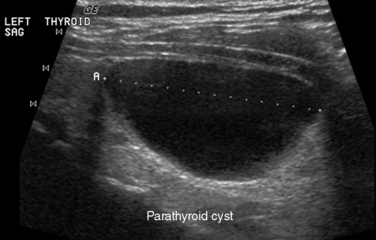
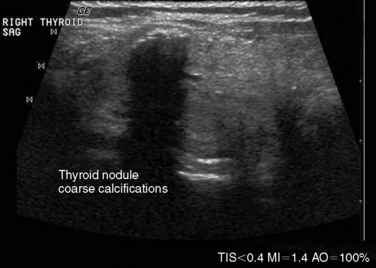
In ultrasonography, artifacts are images that appear on the display and do not represent real anatomic structures. Rather, they are shadows or enhanced representation of tissue elements that are consistent and tell a story. Pure cysts have a thin discrete capsule and are entirely fluid-filled without significant solid components. Sound enters from a superficial direction and as yet unattenuated waves easily penetrate the anterior capsule. Because the interior of the cyst is fluid without elements that reflect sound, the parallel sound waves strike the posterior capsule, which through the acoustical mismatch acts as a reflector. A large proportion of these waves penetrate this capsule and return as bright, high amplitude signals from just beyond. This produces a relatively broad reflected area that is hyperechoic to adjacent tissues and the cyst itself. This artifact is designated “posterior enhancement” and is usually diagnostic of a cyst (Figure 13-1). In contrast, coarse calcifications block transmission of the sound waves to deeper tissue planes, producing a dark rectangular void deep to the densely hyperechoic structure. Known as posterior shadowing (Figure 13-2), this particular artifact represents coarse calcifications often seen in portions of a multinodular goiter and to a lesser degree in some cancers. Microcalcifications (Figure 13-3) generally seen in papillary carcinoma of the thyroid gland, unless closely aggregated do not produce posterior shadowing artifact as a result of their small size. These microcalcifications are small points of hyperechoic signal and represent psammoma bodies defined histologically in papillary carcinoma of either the primary thyroid or metastatic adenopathy. When planning fine-needle aspiration cytology, these areas are good sampling targets under ultrasound guidance. Other artifacts may be confused with microcalcifications. One such confusion can occur with “comet tail” artifact (Figure 13-4). These are hyperechoic points with a tapering core of hyperlucency extending from and deep to the circular dot. When examined more closely, the “tail” portion is actually a form of reverberation artifact. One accepted explanation is that areas of colloid crystallize and serve both as finite obstructions to transmission and deeper reverberation of the ultrasound waves. Ahuja has studied comet tail artifacts in a large number of thyroid conditions, and invariably he has found that the underlying processes are benign.5 Examples of structures that commonly demonstrate reverberation (Figure 13-5, A-C) are the anterior wall of the trachea, anterior wall of the carotid artery, comet tails from small colloid crystals, and biopsy needles in their long axis.
Doppler
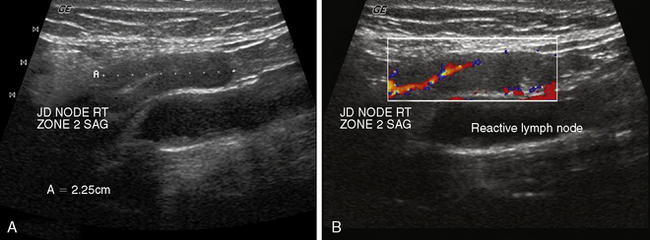
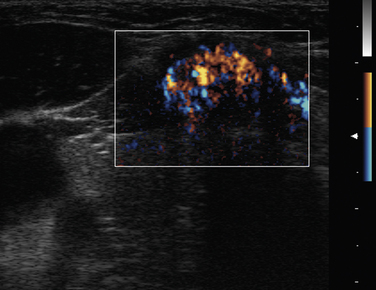
Doppler is a relevant and technically different process than grayscale ultrasound in the assessment of vascularity of anatomic and pathologic elements.6,7 In simplistic terms, the Doppler shift of sound waves occurs when waves imparted at an angle to a blood vessel strike directionally moving red blood cells and are reflected. If the waves are reflected toward the transducer, the velocity of this reflection is augmented. If the sound waves are reflected away from the transducer signifying blood flow away from the transducer, the velocity is reduced. This velocity of red cell movement can be calculated and directional flow given a color designation—that is, flow toward the transducer is red and away from it blue by convention. These Doppler images are superimposed over the corresponding B mode display in a rapidly alternating fashion such that the eye sees a moving color video rendition of this activity. This color Doppler imaging and flow interpolation is highly relevant to the study of carotid and peripheral vascular anatomy and restriction of flow. Power Doppler is a separate technique that ignores these calculations and directional relationships. Power Doppler is more sensitive to and has better resolution for small vessels with low flow such as those found within a lymph node or parathyroid adenoma (Figure 13-6, A and B). In fact, power Doppler can often be used as a differentiating tool between these two structures in the clinical setting. Power Doppler will still reveal large vessels in the head and neck but cannot render actual flow values.
Thyroid Ultrasound
Ultrasound (US) is the first-line recommended imaging modality for thyroid nodules.8,9 Its use in thyroid disorders is widely acknowledged, and its benefits and indications continue to expand. Surgeons and endocrinologists in the office-based setting have adopted thyroid US for the evaluation and management of patients with thyroid, parathyroid, and many other head and neck disorders. Its versatility, convenience, safety profile, ability to offer dynamic real-time images, and low cost compared to other radiologic modalities, combined with the outstanding quality of the images produced by high frequency, high-resolution ultrasound, have all contributed to its popularity. Table 13-1 lists the principal goals of and indications for thyroid ultrasonography.
Table 13-1 Thyroid Ultrasonography: Principal Goals and Indications
| Assess palpable thyroid nodules and enlargement Assess nonpalpable thyroid nodularity and disease Identify characteristics associated with malignancy Assess the thyroid and extrathyroidal neck in patients with thyroid cancer prior to treatment Monitor treated thyroid cancer patients for early evidence of recurrence Monitor nodules, goiters, or lymph nodes in patients undergoing treatment or observation of thyroid disease Screen high-risk patients (with familial forms of thyroid cancer, a history of radiation exposure, FDG avidity on PET, etc.) Screen for thyroid lesions in patients with other diseases in the neck, such as hyperparathyroidism, who are undergoing treatment planning Guide fine-needle aspiration biopsy and other interventions |
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
In past decades, palpable thyroid nodules were surgically excised to establish a pathologic diagnosis. Advances in two techniques, namely US and fine-needle aspiration cytology, and ultimately the combination of these techniques served to revolutionize the treatment of thyroid nodules and to reduce the incidence of unnecessary surgery for nodules that are predictably benign. Continued improvements in ultrasound technology have further improved the ability to identify and distinguish malignant features of thyroid lesions.10–13 These same improvements have led to an enhanced ability to localize pathologic parathyroid glands, as will be discussed later in this chapter.
Role of Ultrasound in the Initial Evaluation of the Thyroid Nodule
Thyroid US is recommended for all patients with suspected thyroid nodules,8,9 including patients with palpable abnormalities, nodular goiter, and thyroid lesions incidentally found by other imaging studies.
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Routine screening thyroid US is not recommended for the general population because of the high incidence of thyroid nodules that are not clinically significant. Palpable thyroid nodules occur in approximately 7% of the general adult population, but the incidence of nonpalpable thyroid nodules visible by ultrasound is up 70%.14–16
US can help confirm the presence of a thyroid nodule; objectively characterize the nodule’s size, location, and appearance; evaluate for benign or suspicious features; and evaluate for the presence of other thyroid nodules or cervical lymphadenopathy.1 Although certain ultrasound characteristics of thyroid nodules are associated with malignancy, fine-needle aspiration (FNA) cytology remains the gold standard for diagnosis. FNA has until recently been recommended for cytologic evaluation of all thyroid nodules greater than 1 cm in diameter or nodules under 1 cm that exhibit suspicious features.8,9 The 2009 Revised ATA Guidelines for Management of Thyroid Nodules and Differentiated Thyroid Cancer include more selective recommendations for FNA of certain thyroid lesions based on ultrasound criteria. These include FNA for mixed solid and cystic nodules 1.5 to 2 cm or greater in diameter with any suspicious ultrasound characteristics and limiting FNA of predominantly spongiform nodules to 2 cm or greater in diameter.8 Despite its utility, the diagnostic role of FNA is limited by an overall 3% false-negative rate and a 10% nondiagnostic rate.17
The use of ultrasound guidance improves the overall sensitivity, specificity, and accuracy of fine-needle aspiration (FNA) compared to palpation-guided FNA.18–20 Ultrasound-guided FNA appears to be most valuable in patients with nonpalpable nodules, small palpable nodules, posterior nodules, multiple nodules, partially cystic nodules, or concomitant glandular disease. It is also beneficial for sampling specific areas of a nodule, such as the solid part of a mixed solid-cystic nodule. Cesur et al. found the rates of inadequate FNA samples to be significantly improved in palpable nodules 1 to 1.5 cm using ultrasound- versus palpation-guided FNA (37.6% versus 24.4%, p = 0.009), but not for palpable nodules 1.6 cm or larger.18 Ultrasound-guided FNA is also specifically recommended when repeating FNA for a nodule with an initial nondiagnostic cytology result.8
Ultrasonography Technique and Measurements
![]() Please see the Expert Consult website for discussion of this topic, including Figures 13-7 and 13-8.
Please see the Expert Consult website for discussion of this topic, including Figures 13-7 and 13-8.
Ultrasonography Technique and Measurements
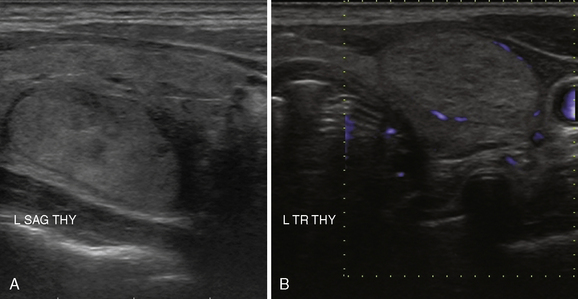
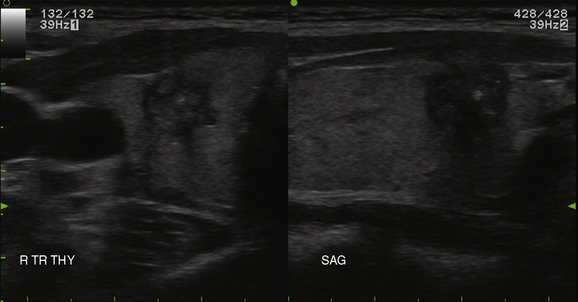
A normal thyroid lobe measures approximately 1.5 to 2 cm in the transverse dimension, 4 to 5 cm in the sagittal plane, and 2 cm in the anterior-posterior dimension. If the sagittal length is greater than the width of the ultrasound transducer and its borders extend beyond the image, the length can be approximated by using the dual-screen option when available and “splicing” together the superior and inferior portions of the lobe (Figure 13-7). The thyroid isthmus has an average length of 1.2 to 1.5 cm in the sagittal plane, and the pyramidal lobe is seldom visible. Normal thyroid parenchyma echoes are fine, uniform, and slightly hyperechoic when compared to the surrounding muscles because of the gland’s iodine content.5,21
Methods exist for estimating thyroid volume by applying a mathematical formula to measurements obtained from US.22,23 However, prospective studies have shown high interobserver variability in estimating thyroid nodule volume24 and poor correlation between predicted and actual thyroid volumes.25 Nevertheless, some use thyroid volume to facilitate 131I dosimetry in patients with thyrotoxicosis.26
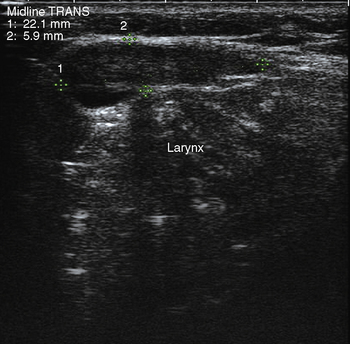
A complete US examination of the thyroid gland should also include examination of the regional lymphatics, noting any abnormally enlarged or atypical lymph nodes. Lymphatic metastases of thyroid carcinoma most often occur in the pretracheal and paratracheal lymph nodes or along the lateral neck jugulodigastric chain. Absence of a cervical thyroid gland in an untreated patient or presence of a thyroglossal duct cyst (Figure 13-8) should also prompt ultrasound inspection of the midline neck from the base of the tongue to the sternum, to assess for possible lingual or undescended thyroid. Laryngeal function can often be evaluated in the process.
Ultrasound Characteristics of Thyroid Nodules
Many distinctive ultrasound characteristics of malignant thyroid nodules have been identified (Table 13-2). Although these ultrasound characteristics offer high sensitivity, no single criterion offers sufficient specificity to differentiate benign from malignant lesions.27 When characteristics are combined, however, specificity improves. One large prospective, observational study comparing US and FNA results with surgical pathology found that performing FNA on nodules with one of three ultrasound criteria—microcalcifications, blurred margins, or hypoechoic pattern—missed only 2% of cancers.28 Kim et al. prospectively analyzed 155 incidentally discovered, nonpalpable, solid thyroid nodules and found a mean number of 2.6 suspicious ultrasound characteristics per malignant nodule and an overall sensitivity and specificity of 94% and 66%, respectively.29
Table 13-2 Ultrasound Features Associated with Malignancy
| Margins Halo/rim Shape Echo structure Echogenicity Calcifications Vascular pattern Elastography Lymph nodes | Blurred, ill-defined Absent, avascular Irregular, spherical, tall Solid Hypoechoic (especially markedly so) Microcalcifications, internal Intranodular, hypervascular Decreased elasticity Abnormal lymphadenopathy |
Size
Nodule size does not predict malignancy. The risk of malignancy for palpable thyroid nodules is approximately 10%, and a similar incidence of malignancy is found in nodules smaller than 1 cm.30–32 Thyroid cancers less than 1 cm in size behave clinically similar to larger cancers, and therefore suspicious subcentimeter lesions that are not treated should be followed with periodic ultrasound surveillance with the option for further evaluation by FNA if growth is observed.21 The ATA recommends FNA biopsy of subcentimeter nodules if there is high risk of malignancy (family history of thyroid cancer, history of external beam or ionizing radiation, history of thyroid cancer, or PET positive nodules) or if there is suspicious concomitant lymphadenopathy, in which case FNA of the lymph node should be performed.8
Margins and Halo/Rim
Benign lesions are often associated with a hypoechoic circumferential vascular “halo” (Figure 13-9, A and B) thought to represent compressed vascularity within thyroid parenchyma adjacent to a nodule.33 Neoplasms may display a partial or absent halo, and its presence or absence has been found to be suggestive but not diagnostic.34,35 For malignant lesions, the halo is typically avascular and represents a true capsule that may by invaded by neoplastic cells. Blurred or ill-defined margins have been associated with an increased risk of malignancy.28,34,35 The mobility of the nodule with respect to surrounding structures can be assessed by dynamic palpation during US, and fixation suggests malignant invasion of the surrounding tissue.
Shape
Nodule shape has been implicated as a prognostic factor. Nodules that are more tall than wide on transverse view (i.e., greater anteroposterior diameter than transverse diameter; Figure 13-10) are more likely to harbor cancer.29 This characteristic has been found to predict malignancy in breast US as well.36 However, one retrospective analysis also found that thyroid nodules with a more spherical shape had a higher incidence of malignancy.33 Irregular shape and microlobulated borders (Figure 13-11) have also been implicated in malignancy.29,35
Echo Structure
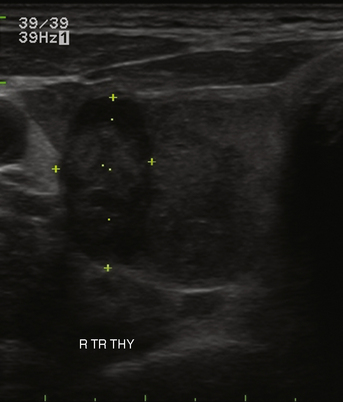
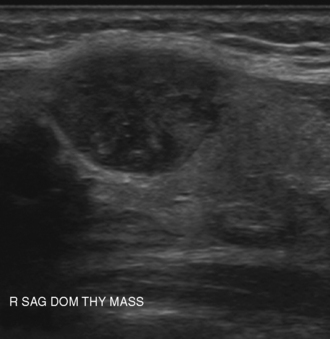
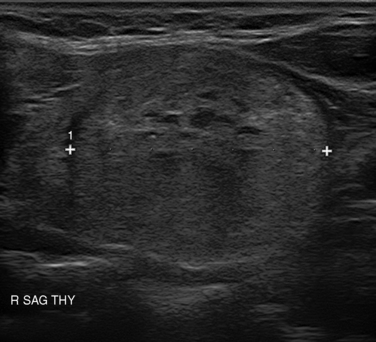
Many thyroid nodules are cystic or have cystic components, such as cystic degeneration of a follicular adenoma (Figure 13-12) or in the setting of multinodular goiter. Malignancy has been more closely associated with solid nodules than with cystic or mixed nodules.28,35 Purely cystic nodules are unlikely to be malignant,37 and those with a spongiform appearance (Figure 13-13), defined as an aggregation of multiple microcystic components in more than 50% of the nodal volume, are more than 99% specific for benign histology.38,39
Echogenicity
The echogenicity of a thyroid nodule should be compared with that of surrounding thyroid tissue. Most benign adenomas or adenomatous nodules are slightly hyperechoic to slightly hypoechoic when compared to normal thyroid tissue (Figure 13-14), whereas malignant nodules are frequently markedly hypoechoic (Figure 13-15), or more hypoechoic than adjacent muscle.29,35
Calcifications
The presence of calcifications has variable significance. Peripheral calcification, also referred to as “eggshell calcification,” has traditionally been considered a benign feature representing previous hemorrhage and degenerative change, although debate exists about its correlation with benignity, especially when the calcification is disrupted. Coarse calcifications, however, are more worrisome for potential malignancy when seen in the center of a solid hypoechoic nodule (see Figure 13-2). Microcalcifications are even more strongly associated with an increased risk of malignancy.28 Forty-five percent to 60% of malignant nodules demonstrate microcalcifications, as opposed to 7% to 14% of benign nodules.29,40 Approximately 60% of patients with microcalcifications have malignant disease.41 Microcalcifications in malignant nodules are often attributed to psammoma bodies in papillary thyroid carcinoma (Figure 13-16) but are also frequently seen in medullary thyroid carcinoma. Although suggestive of malignancy, the overall specificity of microcalcifications for thyroid carcinoma has been reported to range from 71% to 94% with a sensitivity of 35% to 72%,28,42,43 and therefore should not be solely relied on to differentiate benign from malignant lesions.
Vascular Pattern
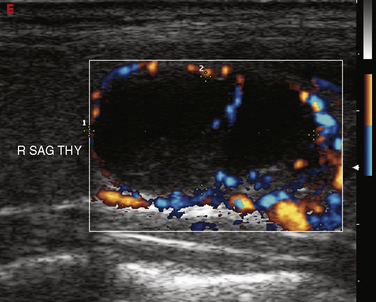
The vascular pattern around or within a nodule may correlate with the probability of malignancy. Chammas et al. classified thyroid nodules according to the pattern of vascularity seen with power Doppler into five types: absent blood flow, perinodular flow only, perinodular flow as great or greater than central blood flow, mainly central nodular flow, and central flow only.44 Nodules with exclusively central blood flow or central blood flow greater than perinodular flow had a higher incidence of malignancy (Figure 13-17). Follicular carcinomas also tend to show a moderate increase in central vascularity by power Doppler compared to follicular adenomas that favor peripheral flow (Figure 13-18).45 In general, increased vascularity in the interior of a thyroid nodule suggests malignancy but should not be considered a pathognomonic feature.
Capsular Contact
The presence of contact between a nodule and the adjacent thyroid capsule is defined when there is no intervening thyroid tissue between the thyroid nodule and the capsule. Capsular contact of greater than 25% by US has been found to be a useful marker for predicting extrathyroidal extension of cancer46 (Figure 13-19).
Ultrasound Characteristics of Benign Thyroid Nodules
![]() Please see the Expert Consult website for discussion of this topic, including Figures 13-20 and 13-21.
Please see the Expert Consult website for discussion of this topic, including Figures 13-20 and 13-21.
Ultrasound Characteristics of Benign Thyroid Nodules
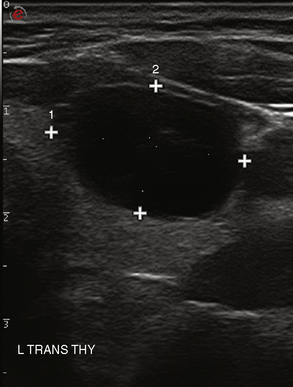
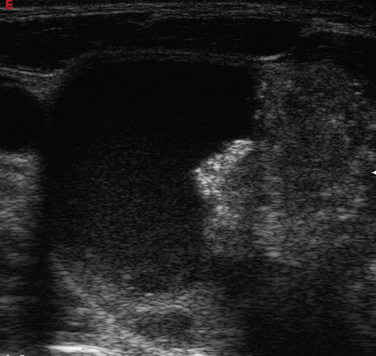
Overall, the most common types of nodules are benign hyperplastic (colloid) nodules and benign follicular adenomas (Figure 13-20). Colloid nodules consist of colloid and benign follicular cells and are associated with small, hyperechoic, internal lucencies on US, known as the “comet tail” sign (see Figure 13-4).47 Follicular adenomas are the most common type of thyroid neoplasm and are, with few exceptions, not considered a forerunner of carcinoma.48 They are typically round, well-encapsulated lesions with a clear margin or “halo” distinguishing them from the surrounding normal thyroid tissue. Spontaneous or traumatic hemorrhage may occur into the nodule (Figure 13-21).
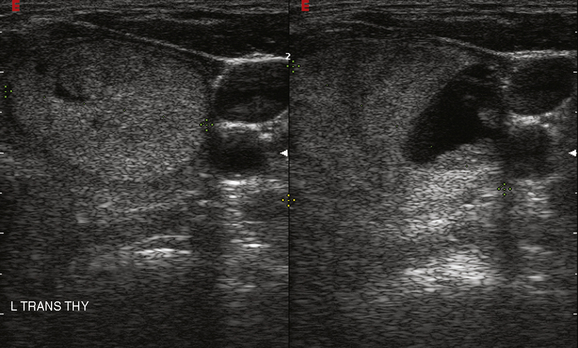
Figure 13-21 Hemorrhage (right panel) into a follicular adenoma (left transverse view). Note halo surrounding the nodule.
Thyroid Cysts
Cystic nodules represent approximately 20% of all thyroid nodules.27 Purely cystic lesions are nearly uniformly benign; however, these comprise only 2% of all cystic lesions.37 Approximately 15% of cystic nodules represent necrotic papillary cancers and 30% represent hemorrhagic adenomas.15 Rates of nondiagnostic FNA are high with cystic lesions, and therefore ultrasound-guided FNA is recommended to ensure sampling of the solid component.8
Ultrasound Characteristics of Malignant Lesions
![]() Please see the Expert Consult website for discussion of this topic.
Please see the Expert Consult website for discussion of this topic.
Ultrasound Characteristics of Malignant Lesions
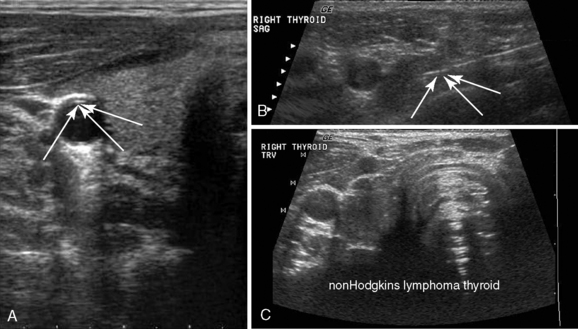
Although the majority of thyroid nodules are benign, a top priority in the management of thyroid nodules is to identify the approximately 10% of nodules that harbor malignancy.49 Most thyroid cancers are of follicular cell origin, including papillary, follicular, and Hurthle cell carcinoma, collectively known as well-differentiated thyroid cancers. Other malignancies including medullary thyroid carcinoma, anaplastic carcinoma, lymphoma, and metastatic disease are less common.
Although ultrasound characteristics of thyroid nodules do not offer sufficient specificity to diagnose malignancy, US represents an invaluable tool for identifying thyroid lesions and determining which lesions should undergo further evaluation through biopsy or other investigations. Clinical features of the various thyroid malignancies are discussed elsewhere throughout this textbook (see Chapters 18, Papillary Thyroid Cancer, 19, Papillary Thyroid Microcarcinoma, 20, Follicular Thyroid Cancer, 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer, 22, Hurthle Cell Tumors of the Thyroid, 23, Sporadic Medullary Thyroid Carcinoma, 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, 25, Sporadic Medullary Thyroid Microcarcinoma, and 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma). Certain ultrasound characteristics are associated with certain thyroid malignancies, as described in this chapter.
Papillary Carcinoma
Ultrasound features typical of PTC include a solid, hypoechoic lesion with microcalcifications. Cystic components may be present within a solid lesion (Figure 13-22), and although an incomplete halo may be seen, ill-defined margins are more common. Doppler examination may also reveal disorganized hypervascularity. Of these features, microcalcifications may be the most specific for PTC because psammoma bodies are a histopathologic feature considered pathognomonic for PTC; they are composed of tiny laminated, spherical collections of calcium that reflect sound waves and appear as tiny bright foci.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree