Purpose
To examine trends in the treatment of newly diagnosed neovascular age-related macular degeneration (AMD).
Design
Retrospective cohort study.
Methods
Among 284 380 Medicare beneficiaries with a new diagnosis between 2006 and 2008, we used the cumulative incidence function to estimate procedure rates and the mean frequency function to estimate the cumulative mean number of intravitreous injections. We used Cox log-binomial regression to estimate predictors of the use of vascular endothelial growth factor (VEGF) antagonists within 1 year after diagnosis. Discontinuation of anti-VEGF therapy was defined by absence of treatment for 12 months. Discontinuation rates were calculated using the Kaplan-Meier method.
Results
The proportion of patients receiving anti-VEGF therapy increased from 60.3% to 72.7%, photodynamic therapy decreased from 12.8% to 5.3%, and thermal laser treatment decreased from 5.5% to 3.2%. Black patients (hazard ratio, 0.77; 95% confidence interval, 0.75–0.79) and patients of other/unknown race (0.83; 0.81–0.84) were less likely than white patients to receive anti-VEGF therapy. Patients with dementia were less likely to receive anti-VEGF therapy (0.88; 0.88-0.89). Among patients who received anti-VEGF therapy, the mean number of injections within 1 year of the first injection was 4.3 per treated eye. Anti-VEGF therapy was discontinued in 53.6% of eyes within 1 year, and in 61.7% of eyes within 18 months.
Conclusions
Treatment of new neovascular AMD changed significantly between 2006 and 2008, most notably in the increasing use of anti-VEGF therapies. However, few patients treated with anti-VEGF medications received monthly injections, and discontinuation rates were high.
Age-related macular degeneration (AMD) is a disabling condition that affects more than 1.5 million older Americans. Approximately 12% of patients have the exudative form of the condition, in which abnormal growth of blood vessels causes vision loss. Although less prevalent than nonexudative disease, exudative AMD accounts for the most cases of severe vision loss. Before December 2004, thermal laser photocoagulation and photodynamic therapy were the only approved therapies. Thermal laser photocoagulation reduces the likelihood of severe vision loss but may cause scarring of the retina and an immediate reduction in vision. Photodynamic therapy, which uses intravenous verteporfin in conjunction with a diode laser to destroy new blood vessels, halts the progression of vision loss but does not restore vision.
Treatment options for exudative AMD have expanded in recent years. In December 2004, the US Food and Drug Administration (FDA) approved pegaptanib (Macugen; OSI Pharmaceuticals), a selective vascular endothelial growth factor (VEGF) antagonist, for exudative AMD. Pegaptanib is associated with less vision loss than placebo; however, as with intravenous verteporfin, treated patients continue to experience vision loss. Ranibizumab (Lucentis; Genentech), an anti-VEGF agent, was approved for intravitreous use in June 2006. Several studies have shown that monthly injections of ranibizumab yield superior vision outcomes, but the most recent research suggests that ranibizumab administered as needed is equivalent to ranibizumab administered monthly. Additional clinical trials evaluating the safety and efficacy of ranibizumab are under way. Although not approved for use in AMD, intravitreous bevacizumab has been used increasingly by ophthalmologists since the fall of 2005. Derived from the same monoclonal antibody as ranibizumab, bevacizumab is approved for the treatment of some cancers. When used for AMD, bevacizumab is much less expensive than ranibizumab ($42 vs $1593 per injection), but its efficacy is not fully understood. Recent results from the landmark comparative efficacy trial of ranibizumab and bevacizumab showed that comparable treatment regimens of ranibizumab and bevacizumab resulted in equivalent improvements in visual acuity at 1 year. This finding was consistent with research by Fong and associates, which found no difference in 1-year visual acuity outcomes between patients treated with ranibizumab and patients treated with bevacizumab. Comparative efficacy trials of ranibizumab and bevacizumab are under way in the United States, Europe, and South America.
Little is known about how these therapies are used in clinical practice and how practice has changed over time. Using a large Medicare claims database, we examined the use of thermal laser photocoagulation, photodynamic therapy, pegaptanib, ranibizumab, and bevacizumab among patients with newly diagnosed AMD from 2006 through 2008.
Methods
Data Sources
We obtained outpatient and carrier standard analytic files and the corresponding denominator files from the US Centers for Medicare & Medicaid Services for January 1, 2004, through December 31, 2009, for all Medicare beneficiaries having any claim with a diagnosis for neovascular AMD (International Classification of Diseases, Ninth Revision, Clinical Modification [ICD-9-CM] diagnosis code 362.52). This time frame allowed for a 3-year study period with 1 year of history and 1 year of follow-up. The standard analytic files contain encrypted identifiers that allow longitudinal follow-up of beneficiaries. The outpatient files contain claims data for outpatient institutional care covered under Medicare Part B. The carrier files contain claims data for noninstitutional physician services also covered under Medicare Part B. The denominator files include demographic characteristics, information about Medicare eligibility and enrollment, and death date, if applicable. Medicare data are made available for research purposes 12 to 18 months after the claim is filed.
We limited the study population to beneficiaries 65 years or older living in the United States. We restricted the analyses to include beneficiaries only during periods of fee-for-service enrollment, because claims data from Medicare managed care plans were not available. The institutional review board of the Duke University Health System approved this study.
Study Population
We selected all Medicare beneficiaries with a diagnosis of neovascular AMD from 2005 to 2008. For each beneficiary, we identified the earliest outpatient or carrier claim that contained a diagnosis of neovascular AMD. We considered the date of this claim to be the index date. We deemed a beneficiary to be newly diagnosed if we found a subsequent AMD claim in the year following the index date and if no AMD diagnosis was recorded in the year prior to the index date. We required beneficiaries to be enrolled in fee-for-service Medicare throughout the ascertainment period. To coincide with the onset of the use of bevacizumab for neovascular AMD, we limited the sample to patients with an index date between January 1, 2006, and December 31, 2008. We used data from 2009 for follow-up.
To enable comparisons with a recently published analysis in the same database, we assembled the source population of fee-for-service Medicare beneficiaries with at least 1 diagnosis of neovascular AMD on an outpatient or carrier claim.
Diagnostic Procedures, Treatments, and Physician Visits
We searched outpatient and carrier claims in the year after the index date for procedures related to the treatment and management of neovascular AMD. Diagnostic procedures of interest included fluorescein angiography (Current Procedural Terminology [CPT] code 92235) and optical coherence tomography (92135). Therapies of interest included thermal laser photocoagulation (67220, 0017T), photodynamic therapy with intravenous verteporfin (67221, 67225), and intravitreous injection (67028) of pegaptanib, bevacizumab, or ranibizumab. We used 2 methods to identify the medications associated with intravitreous injections ( Table 1 ) . First, we searched for medication-specific Healthcare Common Procedure Coding System (HCPCS) medication reimbursement codes. Second, because medication-specific codes are not available immediately after FDA approval, we identified medications during these periods by using nonspecific reimbursement codes in combination with reimbursement amounts from the Medicare quarterly pricing reports. We defined discontinuation of anti-VEGF therapy as the absence of treatment for 12 months, with the first day of the 12-month period serving as the discontinuation date. For all of the procedures listed above, we used HCPCS modifier codes on each claim to determine which eye (ie, left, right, or both) was involved.
| Medication | Payment Amount for “Unclassified” Codes | HCPCS Reimbursement Code | Date Range for “Unclassified” Codes |
|---|---|---|---|
| Pegaptanib | $750-$1000 | J3490 (unclassified drug) | |
| C9128 (injection, pegaptanib) | |||
| J2503 (injection, pegaptanib) | |||
| Bevacizumab | $1-$100 | J3490/J3590 (unclassified drug/biologic) | July 1, 2006–December 31, 2009 |
| J9035 (injection, bevacizumab) | |||
| Ranibizumab | $1500-$2000 | J3490/J3590 (unclassified drug/biologic) | July 1, 2006–December 31, 2007 |
| C9399 (unclassified drug/biologic) | July 1, 2006–December 31, 2007 | ||
| C9233 (injection, ranibizumab) | |||
| J2778 (injection, ranibizumab) |
We searched carrier claims in the year after the index date for evidence of different types of evaluation and management visits. We defined evaluation and management visits coded with CPT codes 99201 through 99215 (ie, ophthalmologic evaluation and management) or 92002 through 92014 (ie, general evaluation and management) as ophthalmology visits if the physician specialty was ophthalmology.
Other Variables
We obtained demographic characteristics including age, sex, and race from the denominator files. Comorbid conditions were based on the presence of relevant ICD-9-CM diagnosis and procedure codes on any inpatient, outpatient, or carrier claim in the year of the index diagnosis. Specifically, we searched for evidence of atrial fibrillation, cancer, cataract, chronic kidney disease, chronic obstructive pulmonary disorder, dementia, diabetes mellitus, glaucoma, heart failure, ischemic heart disease, myocardial infarction, and stroke or transient ischemic attack using definitions developed by the Chronic Condition Warehouse.
Statistical Analysis
In the source population and in the derived study population, we describe the number of claims and unique patients with a neovascular AMD diagnosis in each year. We describe the baseline characteristics of the study population by presenting frequencies with percentages for categorical variables and medians with interquartile ranges for continuous variables. We tested for differences between annual cohorts using χ 2 tests for categorical variables and Kruskal-Wallis tests for continuous variables. In the source population, we calculated the proportion of beneficiaries who received at least 1 intravitreous injection. In the study population, we estimated the proportions of beneficiaries who underwent diagnostic procedures or were treated for age-related macular degeneration, both overall and with specific procedures, using the cumulative incidence function. Among beneficiaries treated with an anti-VEGF agent, we used the mean frequency function to estimate the cumulative mean number of injections per eye during the year after the initial injection. We used the cumulative incidence function and the mean frequency function to account for the competing risk of mortality and the fact that follow-up differs across individuals. Length of follow-up is not uniform because beneficiaries may die or switch to managed care (at which time fee-for-service claims are no longer filed). In all cases, the data were censored at the time of the event. Because of changes in treatment availability during the study period, we present the results of the analyses stratified by the year of the index diagnosis.
We used a Cox proportional hazards regression model to estimate the relative risks of treatment with intravitreous injection within 1 year after the index date. The model adjusted for age, sex, race, comorbid conditions, and the date of the index diagnosis. We estimated rates of discontinuation of anti-VEGF therapy using the Kaplan-Meier method. To ensure that these estimates were not artificially inflated by right-censoring, we used discontinuation data only through 12 months before the date on which each patient’s data were censored. For example, for a patient with data censored after June 30, 2009, the latest possible discontinuation date was June 30, 2008. Therefore, we only used that patient’s data through June 2008 in the discontinuation estimator. We used a series of Kaplan-Meier estimators to describe the distribution of the number of anti-VEGF injections received within 12 months of the first injection. We used SAS statistical software version 9.2 for all analyses (SAS Institute Inc, Cary, North Carolina, USA).
Results
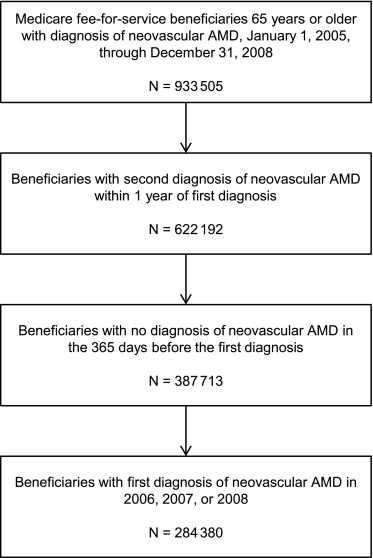
Between January 1, 2005, and December 31, 2008, nearly 1 million Medicare beneficiaries had a diagnosis of AMD ( Figure 1 ) . More than 60% of these beneficiaries had a second diagnosis of AMD within 1 year, and approximately 60% of these had no neovascular AMD diagnosis in the previous year. Therefore, the study population included 284 380 Medicare beneficiaries with a diagnosis of neovascular AMD between January 1, 2006, and December 31, 2008. The source population from which the study population was drawn included more than 400 000 beneficiaries in each calendar year with a diagnosis of neovascular AMD ( Table 2 ) . Approximately one-third of beneficiaries had a single AMD diagnosis during the calendar year, and nearly two-thirds had an AMD diagnosis in the previous year.
| 2006 (n = 434 535) | 2007 (n = 459 312) | 2008 (n = 480 396) | |
|---|---|---|---|
| No. of AMD diagnoses in calendar year, n (%) | |||
| 1 | 169 908 (39%) | 165 035 (36%) | 159 687 (33%) |
| ≥ 2 | 264 627 (61%) | 294 277 (64%) | 320 709 (67%) |
| Any AMD diagnosis in previous calendar year, n (%) | |||
| Yes | 266 182 (61%) | 290 613 (63%) | 323 897 (67%) |
| No | 168 353 (39%) | 168 699 (37%) | 156 499 (33%) |
Distributions of demographic characteristics were consistent throughout the study period ( Table 3 ). Mean age was approximately 81 years, and the cohort was predominantly female and white. Approximately 42% of patients had ischemic heart disease, more than 25% had diabetes mellitus, and approximately 23% had heart failure. The prevalence of atrial fibrillation, cancer, dementia, and diabetes mellitus increased slightly over time, whereas the prevalence of cataracts decreased slightly. The prevalence of chronic kidney disease increased from 9.5% in 2006 to 14.7% in 2008.
| Characteristic | 2006 (n = 95 237) | 2007 (n = 97 515) | 2008 (n = 91 628) | P Value |
|---|---|---|---|---|
| Age, mean (SD), years | 80.9 (6.8) | 81.1 (6.9) | 81.1 (7.0) | <.001 |
| Male, no. (%) | 34 157 (35.9) | 34 963 (35.9) | 33 163 (36.2) | .22 |
| Race, no. (%) | <.001 | |||
| Black | 1236 (1.3) | 1453 (1.5) | 1427 (1.6) | |
| White | 91 397 (96.0) | 93 293 (95.7) | 87 334 (95.3) | |
| Other/unknown | 2604 (2.7) | 2769 (2.8) | 2867 (3.1) | |
| Comorbid conditions, no. (%) | ||||
| Acute myocardial infarction | 967 (1.0) | 1026 (1.1) | 1030 (1.1) | .07 |
| Atrial fibrillation | 11 585 (12.2) | 12 389 (12.7) | 11 656 (12.7) | <.001 |
| Cancer | 7855 (8.2) | 8144 (8.4) | 7875 (8.6) | .02 |
| Cataracts | 31 713 (33.3) | 31 828 (32.6) | 29 096 (31.8) | <.001 |
| Chronic kidney disease | 9005 (9.5) | 13 045 (13.4) | 13 449 (14.7) | <.001 |
| Chronic obstructive pulmonary disease | 13 457 (14.1) | 13 883 (14.2) | 13 037 (14.2) | .76 |
| Dementia | 9455 (9.9) | 11 137 (11.4) | 10 702 (11.7) | <.001 |
| Diabetes mellitus | 24 563 (25.8) | 26 308 (27.0) | 25 641 (28.0) | <.001 |
| Glaucoma | 16 352 (17.2) | 16 925 (17.4) | 16 173 (17.7) | .02 |
| Heart failure | 21 602 (22.7) | 22 323 (22.9) | 20 576 (22.5) | .08 |
| Ischemic heart disease | 39 778 (41.8) | 40 845 (41.9) | 38 269 (41.8) | .83 |
| Stroke or transient ischemic attack | 5029 (5.3) | 5131 (5.3) | 4818 (5.3) | .97 |
Table 4 shows the diagnostic procedures and therapies used within 1 year of the index diagnosis of AMD. The proportion of patients who underwent optical coherence tomography in the year after the index diagnosis increased by 25% between 2006 and 2008, and the mean number of scans increased from 3.8 to 4.7 ( P < .001). In contrast, the proportion of patients who underwent fluorescein angiography decreased slightly, and the mean frequency decreased from 2.4 to 2.1 ( P < .001). The proportion of patients who received any treatment increased from 67.9% in 2006 to 74.6% in 2008 ( P < .001). Use of photodynamic therapy and thermal laser coagulation declined over time, whereas use of anti-VEGF agents rose from 60.3% in the 2006 cohort to 72.7% in the 2008 cohort ( P < .001). Bevacizumab was the most common anti-VEGF agent received by patients with new AMD. Use of bevacizumab increased over time, whereas the use of ranibizumab remained fairly steady and the use of pegaptanib declined dramatically. Among patients initiated on bevacizumab in 2006, 5582 (13.0%) switched to ranibizumab within 1 year. By 2008, 4079 patients (7.9%) newly initiated on bevacizumab switched to ranibizumab. Among patients initiated on ranibizumab in 2006, 3716 (14.6%) switched to bevacizumab within 1 year. In 2008, 3110 (13.6%) patients newly initiated on ranibizumab switched to bevacizumab.
| Procedure | 2006 (n = 95 237) | 2007 (n = 97 515) | 2008 (n = 91 628) |
|---|---|---|---|
| Diagnostic procedures, number of patients (cumulative incidence rate) | |||
| Fluorescein angiography | 76 549 (80.5) | 75 409 (77.4) | 69 463 (75.9) |
| Optical coherence tomography | 62 259 (65.7) | 73 743 (75.9) | 75 614 (82.7) |
| Therapeutic procedures, number of patients (cumulative incidence rate) | |||
| Any treatment (photodynamic therapy, thermal laser, or intravitreous anti-VEGF agent) | 64 485 (67.9) | 70 543 (72.4) | 68 255 (74.6) |
| Photodynamic therapy | 12 150 (12.8) | 6450 (6.7) | 4843 (5.3) |
| Thermal laser | 5261 (5.5) | 3596 (3.7) | 2960 (3.2) |
| Anti-VEGF agent | 57 293 (60.3) | 68 166 (70.0) | 66 588 (72.7) |
| Pegaptanib | 9104 (9.6) | 1125 (1.2) | 548 (0.6) |
| Bevacizumab | 36 561 (38.6) | 46 568 (47.9) | 49 453 (54.0) |
| Ranibizumab | 26 246 (27.8) | 29 238 (30.1) | 23 850 (26.1) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


