Treatment of Thyroid Neoplasms
Irene Zhang
Samantha DeMauro-Jablonski
Robert L. Ferris
It is estimated that 45,000 new cases of thyroid cancer were diagnosed and 1,690 of them will die of this disease in 2010 (1,2). The lifetime risk of developing thyroid cancer is 0.9%, and the annual incidence has increased from 3.6 per 100,000 in 1973 to 10.2 per 100,000 in 2007 (1). This trend may be due to increasing use of neck ultrasound as well as other imaging modalities during routine physical exam. CT, MRI, or PET scans performed for other medical reasons can subsequently result in the detection of incidental benign and malignant thyroid nodules. Thyroid nodules are a common presentation of thyroid cancer. Although the prevalence of thyroid nodules is around 50% at autopsy, and up to 67% detected by ultrasonography (US), only 5% to 10% of these are malignant (3,4). The challenge exists to identify the 5% to 10% of patients with malignancy of clinical significance. Recent advances in molecular markers for thyroid cancer have improved the ability to detect subsets of cancer with aggressive behavior and to assist in the setting of indeterminate fine-needle aspiration (FNA). The diagnosis and treatment of thyroid cancer have significantly evolved over last decade, emphasizing the need for a thorough understanding of the underlying molecular pathways and risk factors, as well as the surgical anatomy, diagnostic, and treatment algorithms necessary to provide excellent care for patients with thyroid neoplasm.
SURGICAL ANATOMY AND EMBRYOLOGY
The thyroid gland originates from both primitive pharyngeal and the neural crest cells. It is the first endocrine gland to develop in the human body. The medial portion of the gland derives from the endodermal diverticulum of the first and second pharyngeal pouches at the foramen cecum. It then descends to its pretracheal position along the midline neck during 4 to 7 weeks of gestation, and the proximal portion degenerates into a fibrous stalk. If any of these portions persists, a thyroglossal duct cyst (TGDC) may result. The distal portion gives rise to the pyramidal lobe. The lateral portion of the gland derives from the fourth and fifth pharyngeal pouches, which descend to fuse with the medial portion of the gland.
Parafollicular C cells originate from the ectoderm neural crest cells as the ultimobranchial body, which secretes calcitonin. In mammals, the ultimobranchial body and medial portion of the fourth pharyngeal fuse into the lateral lobes of the thyroid. Because of this, the majority of C cells are located deep within the upper one-third of the lateral lobe, the most common location for medullary thyroid carcinoma (MTC).
Ectopic thyroid tissue can be found anywhere along the course of its developmental descent in the midline anterior neck. Lingual thyroid can be found at its origin at the foremen cecum due to failure to descend. Seventy percent of the patients with lingual thyroids have no thyroid tissue in the neck. Thyroid function should be evaluated before the surgical removal of the lingual thyroid glands. A TGDC is the most common congenital midline cervical anomaly. It may develop at any location along the thyroglossal duct tract, most commonly at the midline upper cervical location close to the hyoid bone. Rarely, this entity can occur in a lateral position. The hyoid bone divides this tract into upper and lower segments. Thus, surgical management of the TGDC (Sistrunk procedure) should remove the middle segment of hyoid bone to prevent recurrence. Preoperative imaging will aid in delineating the nature of a midline mass as well as identifying functional thyroid tissue prior to complete TGDC excision. High-resolution ultrasound, CT, and MRI are commonly used in detecting TGDC. Calcification seen in a CT scanning raises suspicion of papillary carcinoma arising from TGDC.
RISK FACTORS OF THYROID CANCER
Risk factors for thyroid cancer have been studied extensively. They include age of the patient, gender, history of previous radiation, family history, environmental exposures, and molecular genetic factors. It has been widely accepted that papillary thyroid carcinoma (PTC) is twice as common in women as in men (5). Young patients have a better prognosis than older patients. The clinical presentation differs significantly in terms of the size, extent of the cancer, the presence and the number of lymph node metastases, and response to the treatment (6). Indeed, AJCC has incorporated age as part of the staging criteria for differentiated thyroid cancer (DTC).
Although there is no clear association between dietary iodine supplementation and thyroid cancer, the pattern of thyroid cancer in iodine-sufficient areas differs significantly from iodine-deficient areas, with the shift from high proportion of follicular carcinoma to PTC (6,7).
It is well established that exposure to ionizing radiation to the head and neck region during childhood increases the risk of thyroid cancer. Radiation therapy (RT) was once a therapy for tonsil and adenoid hyperplasia, thymic enlargement, acne, hemangioma, and Hodgkin disease (mantle irradiation) to the head and neck. The risk of thyroid cancer often RT exposure is dose dependent and is greatest for children less than 15 years old. The 25-year follow-up after the Chernobyl accident demonstrated that children and adolescents exposed to radioiodine from the Chernobyl fallout have a substantial dose-related increase in thyroid cancer, with the risk greatest in those less than 18 years old at exposure (8). Fortunately, the disease-specific mortality rate in this patient population is quite low, at 1% or less (9). The increased incidence of thyroid cancer in those greater than 18 years old during the Chernobyl fallout is less definitive.
A patient with a family history of thyroid cancer has an increased risk to develop thyroid cancer, and 5% to 10% of all thyroid carcinoma cases are hereditary (10). Gardner syndrome and Peutz-Jeghers syndrome are both associated with PTC. Cowden disease is associated with follicular thyroid carcinoma (FTC). MEN IIA and MEN IIB are associated with family history of MTC (11).
MOLECULAR BASIS OF THYROID NEOPLASM
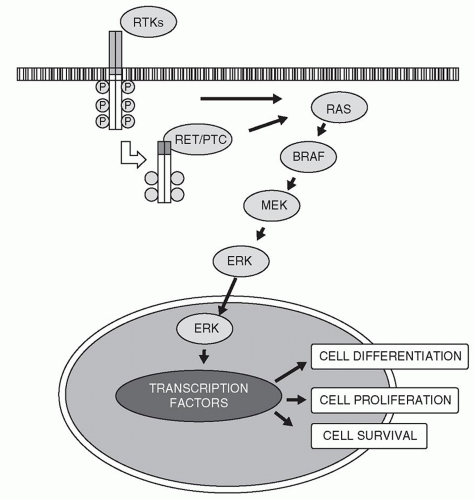
The knowledge of molecular genetics and signaling pathways in thyroid cancer has expanded in recent years. Numerous molecular alterations in thyroid cancer have been identified, and are actively studied in clinical trials as therapeutic targets for recurrent disease. Among them, four major mutations involved in development of differentiated thyroid cancer (DTC), BRAF and RAS point mutation, RET (REarranged during Transfection)/PTC, and PAX8/peroxisome proliferator-activated receptor γ (PPARγ) rearrangements, seem to have most relevant clinical implications (12). Like many other malignancies, genetic alterations in the mitogen-activated protein kinase (MAPK) signaling pathway are involved in development of PTC (Fig. 133.1) (13).
PTC often carries mutually exclusive BRAF and RAS point mutations and RET/PTC rearrangements, which are activated in MAPK signaling pathway. Mutation in one of these genes presents in more than 70% of PTC (14,15,16). BRAF point mutations have been reported in 40% to 45% of PTC. BRAF V600E mutation has been found in 70% to 80% of tall-cell variant of PTC and 60% of classical PTC and strongly correlated with aggressive features of this disease including extrathyroidal extension (ECE), lymph node, or distant metastases (14,17,18). BRAF V600E is not found in FTC or other benign thyroid nodules, which makes it a specific tumor marker of PTC. This association has also been reported in papillary microcarcinomas, which might have some clinical implications in the management of these tumors (19). RET/PTC rearrangements are found in 20% of adult papillary carcinomas, most commonly in patients with history of radiation (20,21). It is common in patients with history of radiation and young adults with PTC. However, it has been found in other benign thyroid lesions or hyalinizing trabecular tumors (21,22). In MTC, RET is activated by point mutation rather than chromosomal rearrangement, as found in PTC. Point mutations of the RAS genes (HRAS, KRAS, and NRAS) are found in FTC, PTC, and follicular adenoma. In thyroid carcinoma, KRAS and NRAS are the most commonly altered. RAS mutations are found in 45% of follicular carcinoma and 10% papillary carcinoma (13). In the latter, PTC with RAS mutation is almost always of the follicular variant (12). These are also found in other follicular adenomas/hyperplasia or goiters. PAX8/PPARγ rearrangement is present in 35% of classic follicular carcinomas. RAS mutation and PAX8/PPARγ rearrangement are rarely overlapping. Mutations in the RAS genes or PAX8/PPARg rearrangement occur in approximately 70% of FTC (23).
Compelling data indicate that molecular testing of thyroid FNA specimens may significantly improve the accuracy of preoperative cytologic diagnosis of thyroid nodules, especially in the subgroups including follicular lesion of indeterminate significance (FLUS), follicular neoplasm, and suspicious for malignancy. Current ATA guidelines recommend that the molecular test results may be considered for indeterminate FNA cytology to help guide the management of those patients. The feasibility and diagnostic utility of molecular testing in FNA of thyroid nodules has been reported in a recent large prospective study including 470 FNA samples. Molecular testing demonstrated 100% accuracy for malignancy in all mutation-positive FLUS cases (24). BRAF was the most common mutation and had 100% positive predictive value (PPV) for PTC, whereas RAS mutation confers 87.5% probability of malignancy (24). In light of the finding in this study, the patients with
mutation-positive and cytologic indeterminate nodules are considered for total thyroidectomy, especially with BRAF mutations, to reduce the need for intraoperative pathologic consultation and to avoid a second surgery. In addition to the diagnostic value of molecular testing, the aggressive behavior of BRAF-positive PTCs provides prognostic information to refine patients’ management, including consideration of central neck dissection. Although still early in clinical application, the expanding knowledge of thyroid tumor biology has started translating into clinical practice, and molecular testing in thyroid FNA will be a valuable tool for diagnosis and better prognostication of thyroid cancer.
mutation-positive and cytologic indeterminate nodules are considered for total thyroidectomy, especially with BRAF mutations, to reduce the need for intraoperative pathologic consultation and to avoid a second surgery. In addition to the diagnostic value of molecular testing, the aggressive behavior of BRAF-positive PTCs provides prognostic information to refine patients’ management, including consideration of central neck dissection. Although still early in clinical application, the expanding knowledge of thyroid tumor biology has started translating into clinical practice, and molecular testing in thyroid FNA will be a valuable tool for diagnosis and better prognostication of thyroid cancer.
EVALUATION OF THYROID NODULES
Most commonly thyroid neoplasms present as either palpable thyroid nodules or nonpalpable nodules incidentally found by imaging studies, so-called incidentalomas. These “incidentalomas” carry the same risk of malignancy as palpable nodules with the same size. Generally, any nodule greater than 1 cm warrants evaluation. Occasionally, subcentimeter nodules with worrisome ultrasound features, previous history of head and neck irradiation, family history of thyroid cancer, or associated neck lymphadenopathy require further evaluation. Recent studies demonstrated 27% to 42% risk of malignancy in focal or unilateral uptake in 18FDG-PET scan (25,26,27). The term “PAINs” (PET-Associated Incidental Neoplasm) was introduced, and the prompt evaluation and treatment of these patients are warranted (28).
Clinical Assessment
For patients presenting with a thyroid nodule, a thorough history and physical examination should be performed. Pertinent elements increasing the possibility of malignancy in the history are previous history of irradiation to the neck, history of thyroid cancer in first-degree relatives, rapid growth, dysphasia, hoarseness, male gender, and presentation at extreme age (less than 20 or older than 70) (29).
The worrisome findings on physical examination which raise concern for malignancy are size of the nodules greater than 4 cm, lateral cervical lymphadenopathy, vocal cord immobility, and fixation of the nodule to surround structures. The accuracy of physical examination may be limited by the patient’s body habitus, and further imaging may be of value for the patients with large BMI or posterior located nodules.
Laboratory Studies
Most patients with thyroid nodules are euthyroid. A serum thyroid stimulating hormone (TSH) level should be obtained as part of the evaluation. Subsequent T3, T4, and
a radionuclide thyroid scan should be obtained for those with a low TSH to further evaluate the thyroid function. Thyroglobulin (Tg) level is not routinely measured as initial evaluation of thyroid nodule due to its lack of sensitivity and specificity as a screening tool. Serum calcitonin level and urine metanephrine should be obtained in patients with family history of medullary thyroid cancer or MEN IIA or IIB.
a radionuclide thyroid scan should be obtained for those with a low TSH to further evaluate the thyroid function. Thyroglobulin (Tg) level is not routinely measured as initial evaluation of thyroid nodule due to its lack of sensitivity and specificity as a screening tool. Serum calcitonin level and urine metanephrine should be obtained in patients with family history of medullary thyroid cancer or MEN IIA or IIB.
Imaging Modalities
Ultrasound
High-resolution thyroid ultrasound should be performed in all patients with thyroid nodule(s) to assess the thyroid bed, central compartment, as well as lateral neck, according to the 2009 ATA guideline. US helps to confirm the presence of a nodule corresponding to the physical examination, size of the nodule(s), presence of other nodules in the thyroid bed and/or lymphadenopathy in lateral necks, components of the nodule(s) (cystic vs. solid), and posteriorly located nodules. It also plays an important role for monitoring the interval changes of preexisting nodule(s) and posttreatment surveillance for thyroid cancer. In addition, it allows identification of certain suspicious ultrasound features suggesting malignancy. Although no single sonographic finding is pathognomonic for thyroid malignancy, certain ultrasound features of thyroid nodule are associated with higher probability of malignancy (30,31). These include hypoechogenicity, increased intranodular vascularity, irregular or infiltrated margins, presence of microcalcifications, absence of halo (for papillary carcinoma), and length greater than the width in transverse dimension. The sonographic features highly predictive of benign nodules are pure cystic, coarse calcifications, and regular margins. These features also help to select appropriate nodule(s) and/or site within the nodule (cystic vs. solid) to aspirate for fine needle aspiration biopsy (FNAB).
Radionuclide Thyroid Scan
The role of radionuclide scanning in the initial workup of a thyroid nodule is limited. It is indicated in a nodule(s) with low TSH to determine the functional status of the nodule, since the rate of malignancy in hyperfunctioning thyroid nodule is less than 1%. A hyperfunctioning nodule demonstrated on a radionuclide scan obviates the need for FNAB.
CT, MRI, and PET/CT
Unlike high-resolution, US, CT, or MRI imaging are insensitive for intrathyroid nodule(s), and routine use is rarely indicated in initial workup for thyroid nodule(s). However, both CT and MRI are excellent to evaluate the mediastinal extension or to assess, trachea, esophageal, and/or extrathyroid invasion. CT with contrast as a preoperative evaluation will delay potential postoperative radioactive iodine (RAI) therapy by roughly 3 months. Although PET/CT has been used in initial staging of oncocytic (Hurthle cell) carcinoma or poorly differentiated thyroid carcinomas, it is not indicated in initial workup for a thyroid nodule. On the other hand, PAINs deserves prompt evaluation due to its substantial risk of malignancy. Although, some suggest that PET/CT might play a role to reduce the need of diagnostic lobectomy for nodules with inconclusive cytologic results due to its high negative predictive values, further study is needed before this expensive test becomes universally accepted.
Fine-Needle Aspiration and Molecular Markers
FNA is the procedure of choice to evaluate the thyroid nodule and may be performed via palpation or via US guidance. According to 2009 ATA guideline, FNA is indicated in following situations:
Any nodule with high-risk history if it is greater than 5 mm, with microcalcification, or any abnormal cervical lymph nodes
Greater than 1-cm solid nodule
Greater than 1.5-cm mixed nodule with any suspicious ultrasound features
Greater than 2.0-cm spongiform nodules or mixed nodule without any suspicious ultrasound features
FNA is not indicated for pure cystic nodule. In case of multinodular gland, preference should be given to those hypo- or isofunctioning nodules with suspicious ultrasound features. The diagnostic accuracy is improved with US-guided FNA because of a decreased number of inadequate samples or false negative results (32). According to 2009 ATA guidelines, US-guided FNA is recommended for nonpalpable nodules, predominately cystic nodules, and posterior nodules. The cytologic results from FNA should be reported under six categories to reflect the risk of malignancy according to 2007 Bethesda system for reporting thyroid cytopathology (Table 133.1) (33). PTC can be reliably diagnosed from FNA due to its characteristic nuclear features. Benign or malignant follicular neoplasm cannot
be distinguished on cytology alone, because capsular or angiolymphatic invasion is required to make the diagnosis, which cannot be assessed from cytopathology.
be distinguished on cytology alone, because capsular or angiolymphatic invasion is required to make the diagnosis, which cannot be assessed from cytopathology.
TABLE 133.1 THE BETHESDA SYSTEM FOR REPORTING THYROID CYTOPATHOLOGY | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
As discussed before, recent advances in molecular testing in thyroid FNA specimens have enhanced the ability to distinguish malignant from benign nodules. The most commonly studied molecular markers in thyroid neoplasm are BRAF and RAS point mutations, as well as RET/PTC and PAX8/PPARγ rearrangements. Although the exact indications of molecular testing need to be defined, it seems that most clinical utility lies in assisting the decision making between thyroid lobectomy and total thyroidectomy when the cytology is under the category of “follicular neoplasm” or “follicular lesion of undetermined significance (FLUS).” Also, BRAF V600E-mutated cancers may be considered for central neck dissection, particularly if greater than 1 cm, to reduce the need for reoperation.
Workup
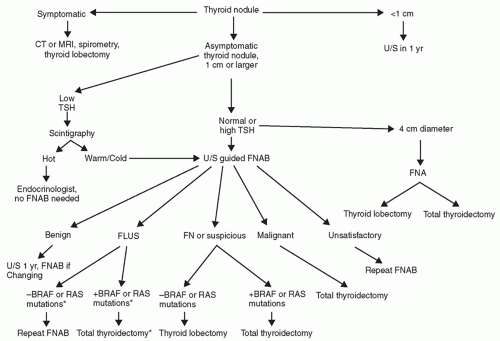
The authors have proposed the algorithm in Figure 133.2 for workup of a thyroid nodule. It is based on the 2009 American Thyroid Association guideline, 2009 Bethesda system for reporting thyroid cytopathology, and the most recent advances in thyroid FNA molecular basis of thyroid cancer.
Any symptomatic thyroid nodule should be evaluated with TSH level and diagnostic ultrasound. Thyroid uptake scan should be ordered in those with low TSH levels. The patients with hyperfunctioning nodule can be worked up by an endocrinologist, and no FNA is recommended. FNA is indicated in the patients with normal or high TSH.
A repeat FNA should be performed if FNA is reported as “unsatisfactory.” Patients who have a benign FNA report can be followed. There is a 3% false negative rate. Annual US is used to monitor the nodule for growth or
development of suspicious sonographic features for malignancy, at which time a repeat FNA is warranted. Total thyroidectomy is recommended when the FNA report indicates malignancy. The FNA has a sensitivity of 97% to 99% (9). Recent prospective data from University of Pittsburgh Medical Center demonstrated 100% PPV and 100% specificity for BRAF-positive “follicular neoplasm” or “suspicious for malignancy” FNA (15). Another study showed 100% PPV in mutation-positive “FLUS” group, while the probability of malignancy for mutation-negative FLUS group is only 7.6% (34). The asterisk part in Figure 133.2 reflects our clinical practice based on above findings. Currently, it has not yet been fully validated or universally accepted. We believe the treatment algorithms will change in the near future as more data accumulate and molecular testing becomes more widely available.
development of suspicious sonographic features for malignancy, at which time a repeat FNA is warranted. Total thyroidectomy is recommended when the FNA report indicates malignancy. The FNA has a sensitivity of 97% to 99% (9). Recent prospective data from University of Pittsburgh Medical Center demonstrated 100% PPV and 100% specificity for BRAF-positive “follicular neoplasm” or “suspicious for malignancy” FNA (15). Another study showed 100% PPV in mutation-positive “FLUS” group, while the probability of malignancy for mutation-negative FLUS group is only 7.6% (34). The asterisk part in Figure 133.2 reflects our clinical practice based on above findings. Currently, it has not yet been fully validated or universally accepted. We believe the treatment algorithms will change in the near future as more data accumulate and molecular testing becomes more widely available.
OVERVIEW OF THYROID NEOPLASM
Thyroid Adenoma
Thyroid adenomas are true benign thyroid neoplasms, distinctly separate from surrounding thyroid tissue. They are often encapsulated and arise from normal thyroid follicular cells, in multinodular goiter or in thyroiditis. Clinically, patients usually are asymptomatic, and the nodule is often found in routine physical examination. Spontaneous bleeding into the adenoma may cause sudden increase in size and pain. Follicular adenomas are the most common. Other types include oncocytic (Hurthle cell) adenoma, hyalinizing trabecular adenoma, and nodular adenomatous goiter. Thyroid adenomas with benign FNA results warrant follow-up due to the false negative rate of 5% (35). Nodule growth during the follow-up warrants repeat FNA. The 2009 ATA guidelines recommend serial US every 6 to 18 months following initial FNA. The follow-up interval can extend up to 3 to 5 years if the nodule is stable over time. Routine TSH suppression is not recommended for benign thyroid nodules. If surgery is indicated, the minimal surgical intervention should be thyroid lobectomy.
Papillary Carcinoma
PTC is the most common type of malignant thyroid neoplasm, both in adult and children. It is twice as common in women as in men (5). The hallmark for PTC is its papillary architecture, psammoma bodies, and characteristic nuclear features. The distinct nuclear features include nuclear grooves, nuclear pseudo-inclusions, powdery chromatin, and micronucleoli. These features allow a diagnosis of PTC based on cytology of FNA. Several histologic variants of PTC have been identified, including papillary thyroid microcarcinoma, follicular variant, encapsulated variant, diffuse sclerosing variant, and oxyphilic cell variant and two aggressive types—tall cell and columnar cell variant.
Most patients present with a slow growth neck mass. Thirty-five percent PTC were found to have cervical lymph nodes involvement at the presentation. Multicentric involvement and ECE are common. Previous exposure to ionizing radiation, especially during childhood, is wellestablished risk factor for papillary carcinoma. Although most of the cases are sporadic, several syndromes involving familial PTC have been described, which include Cowden syndrome and familial adenomatous polyposis coli.
Follicular Carcinoma
FTC is more aggressive compared with PTC. It is more prevalent in iodine-deficient regions. Although most radiation-induced thyroid cancers are papillary carcinoma, it also plays a role in development of FTC. There are two types of FTC: minimally invasive and widely invasive, each of which carries different prognosis. Diagnosis of FTC is based on capsular and lymphovascular invasion, which makes this entity almost impossible to diagnose based on cytology or frozen section.
The clinical presentation is similar to PTC. The incidence of nodal involvement at presentation is lower than papillary carcinoma, ranging from 15% to 20%. However, the incidence of distant metastasis at presentation is more common than PTC, ranging from 10% to 15% (36,37). The most common sites of metastasis are lung and bone.
Hurthle Cell Carcinoma
Oncoytic (formerly Hurthle cell carcinoma, HCC) is uncommon and currently considered as a variant of follicular carcinoma according to World Health Organization classification. It is twice as common in males as in females. Hurthle cells are large follicular epithelial cells with dense eosinophilic cytoplasm. Similar to follicular carcinoma, diagnosis of HCC is based on its capsular and vascular invasion. The incidence of lymph node involvement is around 6% to 9%. HCC has the highest rate of distant metastases compared to PTC and conventional type FTC. Thirty-four percent of HCC developed distant metastases, and most common sites are lung and bone (36,38).
Anaplastic Carcinoma
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree