Fig. 7.1
Neutrophil on blood smear, Wright’s Giemsa stain. Original magnification 100 × oil immersion. Note the characteristic multi-segmented nucleus and mostly lilac-staining specific (secondary) granules and a few azurophilic primary granules. Surrounding cells are mature erythrocytes (Credit: Ruth F. Padmore MD, FRCPC, PhD, Ottawa, Canada)
Neutrophils have a diameter of 12–15 μm. In circulating blood, neutrophils are in a quiescent, or resting state, and have a vaguely circular form. However, when activated, the shape changes, with the cell becoming more amoeba-like, with pseudopods extending in search of antigens.
The granules present in neutrophils contain a number of substances with either bactericidal or proteolytic actions. Primary (azurophilic) granules typically contain the bactericidal enzymes defensins and cationic proteins. They also contain proteolytic enzymes, cathepsin G, lysozyme, and myeloperoxidase. The specific (secondary) granules contain lysozyme, lactoferrin, as well as compounds involved in the formation of toxic oxygen species (Hager et al. 2010).
7.2 Hematologic Progenitors
As granulocytes, neutrophils share their origins with basophils and eosinophils. These are derived from progenitor cells in the bone marrow and then differentiate through myelocytes into promyelocytes and then into the final differentiated cell.
Neutrophils have a relatively short half-life, up to 6 days. This is believed both to prevent the spread of pathogens that may parasitize within neutrophils to facilitate survival and dispersion and also to limit local tissue damage caused by the intense antibacterial activity of the neutrophil in a tissue.
7.3 Physiology and Function
Neutrophils are principally responsible for bacterial killing, which they enact through a variety of mechanisms. Prior to serving this role, neutrophils are primarily present in the circulation in an inactivated form. They then migrate into target tissues to exert their effects following activation by a variety of proinflammatory signals.
The transit from bloodstream to tissue begins with a process called diapedesis. This leukocyte extravasation process occurs when the activated neutrophil approaches the periphery of the blood vessel and then becomes attached to and migrates through the blood vessel wall in a process called diapedesis. This involves an interaction with receptors called intracellular adhesion molecules (ICAMs) and various selectins and integrins.
Arrived at the site of infection, the neutrophil exerts its antibacterial action through the following mechanisms:
1.
Phagocytosis and killing of pathogens
2.
Neutrophil extracellular trap (NET)
3.
Protease digestion through the release of granule contents
The activated neutrophil can phagocytose bacteria by engulfing them and killing them within the cell through the generation of toxic substances. This process called the “oxidative burst” creates a high concentration of reactive oxygen species (ROS) through a process involving NADPH oxidase activation and the creation of superoxide dismutase (SOD). Through several steps, this leads to the production of hypochlorous acid (HClO), which may be bactericidal in itself and/or lead to the activation of the necessary proteases (van den Berg and Kuijpers 2011).
More recently, a role for extracellular trapping and killing of bacteria by neutrophils has been described by the formation of “extracellular NETs.” In this mechanism, neutrophils secrete a mesh composed of DNA and various proteins outside the neutrophil, which serves to trap and destroy bacteria (Fig. 7.2). This may also limit the propagation of infection as well. NETs which form intravascularly are responsible for many of the clinical manifestations of sepsis.
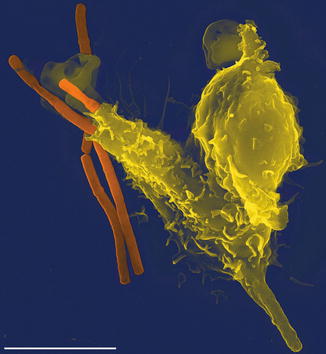

Fig. 7.2
Neutrophil extravasation trap (NET). In this scanning electron microscope image, an Anthrax bacteria (orange) is being engulfed by a single neutrophil (yellow), by generation of a NET (By Volker Brinkmann [CC-BY-2.5 (http://creativecommons.org/licenses/by/2.5)], via Wikimedia Commons)
The neutrophil also plays a role in limiting potentially negative effects of inflammation through the secretion of various serine proteases. The most well known of these is alpha-1-antitrypsin, which serves to limit the extent of damage caused by neutrophil elastase released from granules in tissue. Individuals with low levels of alpha-1-antitrypsin may have inordinate responses to trauma. In smokers, this may lead to the development of emphysema.
7.4 Implication in Disease
Low levels of circulating neutrophils, or neutropenia, may occur from genetic causes or from infectious or toxic causes, one of which the most familiar is chemotherapy. Individuals with neutropenia are particularly susceptible to infection by bacterial pathogens.
The neutrophil may also be involved primarily in inflammatory diseases. Familial Mediterranean fever is a relapsing disorder where individuals present with bouts of acute inflammation characterized by hyperthermia, arthralgia, and peritonitis. Due to a dysfunction in the MEFV gene, individuals have a reduced amount or structural malformations of a protein called pyrin which makes up part of the cytoskeleton of leukocytes. Pyrin abnormalities lead to defective inflammation regulation and, subsequently, to inappropriate or prolonged inflammatory activity (Savic et al. 2012). It is a disorder not directly linked to neutrophil level, but rather to function, as described for alpha-1-antitrypsin deficiency above.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


