This article provides an overview of the technical considerations of endoscopy of the posterolateral skull base and cerebellopontine angle (CPA). Specific areas of focus are on the instrumentation requirements for neuroendoscopy of the CPA; the learning curve associated with this technique; and a complete description of the surgical techniques necessary to perform the procedure, along with outcomes and results. The article provides a general overview of the endoscopic approach to the CPA. For a variety of pathologies, the emphasis is on performing this technique for acoustic tumors and hearing preservation. Insights as to how the author’s practice evolved in its use of neuroendoscopic procedures are provided.
| BAER | Brainstem auditory evoked responses |
| CPA | Cerebellopontine angle |
| CSF | Cerebrospinal fluid |
| HFS | Hemifacial spasm |
| IAC | Internal auditory canal |
| MVD | Microvascular decompressions |
| REZ | Root entry zone |
| TS | Transverse/sigmoid |
The necessity of improved visualization during surgery with regard to clarity, magnification, and illumination has remained paramount in all aspects of surgery, but it is one of the most essential elements in skull base surgery. The idea of surgical endoscopy is not new. Max Nitze is credited with the first design of a surgical endoscope in 1879. This initial invention, using a series of multiple lenses illuminated by a light at the tip, was developed to overcome the same obstacles surgeons still encounter: magnification and improved visualization in the depths of a surgical field. Doyen, in 1917, further developed this idea with the introduction of a crude endoscope used to visualize the cerebellopontine angle (CPA). However, the primitive technology of lenses and light source at the time significantly limited this technology and it was quickly abandoned. With the microscope’s superior illumination and magnification the idea of the endoscope was quietly dismissed.
Because of this fundamental tenet, the introduction of the operating microscope was an essential advancement in neurosurgery in the latter part of the twentieth century. Surgical techniques and instruments were developed to build on the strengths and minimize the limitations of the operating microscope. Advancements in the field of microsurgery allowed the surgeon to compensate for the microscope’s deficiencies by creating instruments and techniques that create linear corridors of visualization. These advancements, which provided accessibility to the posterolateral skull base, specifically the CPA, required either extensive removal of the temporal bone or mobilization and retraction of the cerebellum to provide a channel for the optics and the light source to adequately illuminate the surgical field ( Fig. 1 ). Although the operating microscope provides significant improvement in visualization compared with surgical loupes and headlight, there are significant limitations to the microscope because of the location of the light source and optics outside of the surgical incision. Because the light source is located a distance from the surgical incision, a significant portion of the light is disbursed, and therefore unavailable for illumination of the deep recess of the CPA ( Fig. 2 A). Additionally, because light and optics travel in a straight line, illumination and visualization around corners is virtually impossible. As such, additional brain retraction or removal of bone is necessary to broaden the field of view, especially for surgery within the CPA.
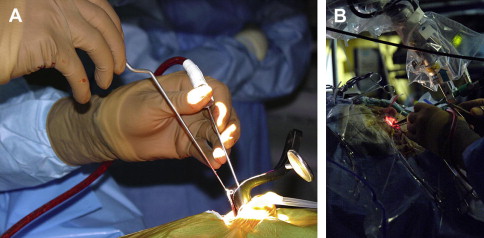
It was not until the 1970s that the surgical endoscope would once again emerge and gain wider acceptance in the field of surgery. The introduction of the endoscope to neurosurgery has been at a much slower pace compared with in general, otolaryngology, and orthopedic surgery. The major criticism of these early endoscopes was their bulkiness and poor resolution compared with the superiority of the operating microscope. As camera technology improved, the endoscope was introduced to neurosurgery for use in the ventricular system. The intraventricular procedures required that the endoscope be maintained in a relatively fixed position over the course of the procedure and therefore could be safely used by the addition of a rigid endoscope holder or a surgical assistant. The early results showing less tissue damage and enhanced visualization with faster patient recovery times fueled the interest of endoscopy within neurosurgery. However, it was not until the collaborative efforts between endoscopically trained otolaryngologists and neurosurgeons while accessing the pituitary fossa that the endoscope evolved into the world of skull base surgery.
In the 1990s the endoscope was reintroduced to the CPA, almost 80 years since Doyen had described his experience during a trigeminal neurectomy. Since then a small but ever growing number of surgeons have reported their experiences using the endoscope as an adjunct to the surgical microscope and microsurgical techniques. Shahinian and colleagues in 2001 were the first to describe a fully endoscopic approach to the CPA.
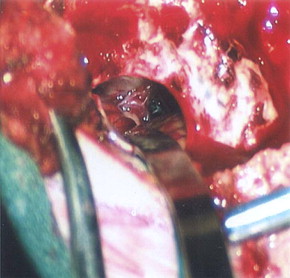
In 2005, our skull base team further developed this technique to access and treat several pathologies within the CPA extending through the incisura and foramen magnum superiorly and inferiorly, and lesions extending anteriorly into Meckel’s cave, the internal auditory canal (IAC), and jugular foramen; posteriorly by the foramen of Luschka; and medially to access lesions involving the contralateral CPA. The major benefit of accessing the CPA through a fully endoscopic approach is the decreased tissue manipulation and retraction while simultaneously widening the field of view and obtaining a true 360-degree visualization of the structures of the CPA. Because the endoscope is a mere 4-mm diameter shaft, the approach around the cerebellar hemisphere can be obtained without the need for retractors, thereby obviating the need to sacrifice venous structures that would otherwise be at increased risk during cerebellar retraction ( Fig. 3 ). The enhanced illumination, allowed by a focal, intradurally placed light source, and an unobstructed wide field of view from the endoscope allows unimpeded visualization from cranial nerve (CN) IV at the level of the incisura to the cervicomedullary junction through a cranial defect as small as 14 mm. However, as with the development of any new technique, significant obstacles were initially encountered. These primarily included stabilization of the endoscope, heat transmission from the light source, lack of endoscope-specific surgical instruments, and the ever-present learning curve.
Technical obstacles in endoscopy
Stabilization in Endoscopy
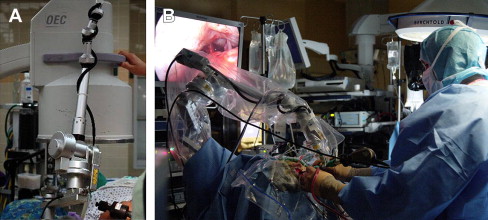
One of the earliest and most difficult technical obstacles was stabilization of the endoscope. In the early reports of endoscope-assisted approaches to the CPA, many authors describe manipulating the endoscope in one hand and working with the other hand. This significantly limits use of the endoscope. To fully use the endoscope in the approach, bimanual surgical technique is essential. Therefore, an effective strategy to stabilize the endoscope while using bimanual microsurgical technique had to be developed. Unlike the intraventricular approaches, visualization of the CPA requires constant changes to the field of view. Therefore, a rigid endoscope holder used in intraventricular procedures was not practical for procedures within the CPA. Additionally, because of the confined space during approaches to the CPA, the ability to use an assistant to stabilize the endoscope, as is typically done with transsphenoidal approaches, is not feasible. We were able to overcome this situation by using a pneumatically controlled multijointed, polyaxial arm (Mitaka Kohki, Tokyo, Japan). This arm works similarly to the operating microscope using a single button to release all of the joints, allowing for one-handed control of the endoscope and immediate and secure stabilization of the endoscope after the button is released, allowing the surgeon to carry out the procedure bimanually ( Fig. 4 ).
Heat Transmission in Endoscopy
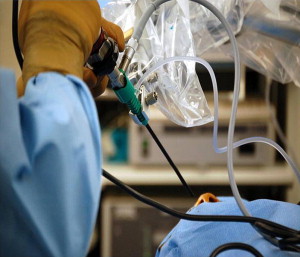
One of the major advantages of the endoscope is the enhanced illumination of the surgical field because the light is not disbursed as with the surgical microscope, but remains focused within the area of interest. However, this advantage is also a major disadvantage because the proximity of the xenon light to the structures within the CPA can become overheated. To overcome this problem we introduced a water-cooled sleeve to the outside of the endoscope (Medtronic, Xomed, Jacksonville, FL, USA) ( Fig. 5 ). A foot pedal is depressed periodically, which bathes the endoscope dissipating the heat generated from the light source allowing the procedure to continue unimpeded without removing the endoscope from the surgical field.
Surgical Instruments for Endoscopy
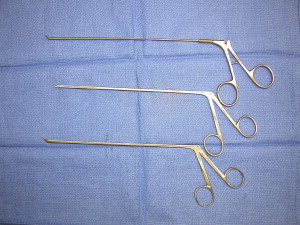
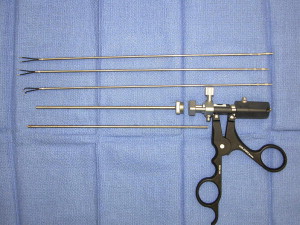
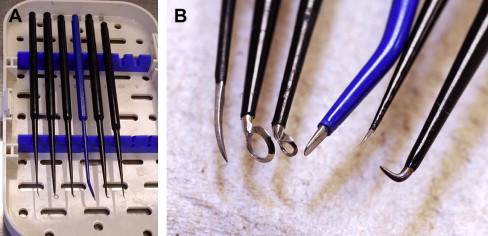
Instruments specifically designed for microsurgery are, in many cases, counterproductive for use in endoscopic procedures. This is not unique to neurosurgery but is well established and a necessary growing pain experienced by clinicians in other surgical specialties. The bayoneted and opposable grip instruments, specifically forceps, scissors, and bipolars, in most cases cannot be transferred from the microscope to the endoscopic procedure. To overcome this we surveyed instruments from the otolaryngology and sinus surgery sets to obtain perspective in designing straight-shafted pistol-grip forceps and scissors for use in these CPA endoscopic procedures ( Fig. 6 ). Because of the limited corridor to the CPA bounded by neural tissue posteriorly and petrous bone anteriorly, the trajectory for instruments is somewhat limited; therefore, each of these instruments has rotational capabilities to allow maneuverability in a 360-degree plane. The major instrument limitation encountered was the lack of a straight-shafted, rotational pistol-grip bipolar. Our initial attempts to use the bayoneted opposable bipolars from the microsurgical sets was unsuccessful because the tips kept scissoring and therefore could not be used reliably. Since our initial experience in 2005, such a bipolar is now developed that comes with three interchangeable tips, angled and straight (Karl Storz, Tuttlingen, Germany) ( Fig. 7 ). We also use a set of straight-shafted monitored microdissecting instruments ( Fig. 8 ). These instruments allow us to continually monitor cranial nerve activity while actively dissecting and resecting neoplasms within the CPA without changing instruments.
Learning Curve in Endoscopy
Most neurosurgeons have little if any formal training with endoscopy. As such, before beginning a neuroendoscopy practice, time must be spent familiarizing oneself with the technique, the instruments, and manipulating the endoscope during surgery. Currently, most cameras provide a high-definition picture in two-dimensions. Therefore, the surgeon must learn to accommodate the loss of depth perception. This is easily overcome with practice, but is necessary to become proficient with endoscopy. Recent advances in camera technology may overcome this disadvantage in the near future, but until then this fundamental limitation is a major step in the learning curve.
Another significant learning obstacle is also one of the true advantages of the endoscope. The ability to use angled endoscopes provides the surgeon with a way to effectively look around corners without additional retraction or further drilling. I have found that the 30-degree endoscope is adequate for visualizing the IAC or Meckel’s cave ( Fig. 9 ). A significant learning curve exists for these angled endoscopes, and the greater the angle the greater chance for disorientation.
Patient Selection for Endoscopic Surgery
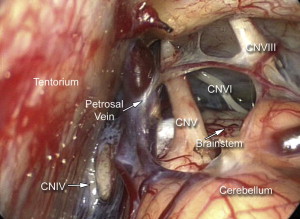
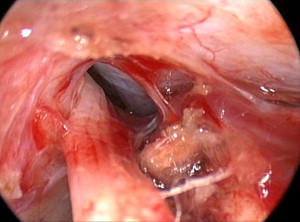
To create an adequate working corridor, especially to resect acoustic tumors, accessing the basilar cisterns is mandatory. This is never done blindly. The endoscope is directed along the superior-ventral aspect of the cerebellum until the arachnoid of the basilar cistern is visualized. The petrosal veins should be identified and preserved if possible ( Fig. 10 ). After the arachnoid has been opened sharply, the egress of the cerebrospinal fluid (CSF) allows further relaxation of the cerebellar hemisphere and a larger working corridor. Therefore, a contraindication of this approach is a tumor that has filled the prepontine cistern or involves significant mass effect elevating the posterior fossa pressure. In these cases a microscope-assisted approach is more prudent allowing the surgeon early access to the cisterna magna and ultimately providing a decrease in the intracranial pressure. We also limit the endoscopic approach to those tumors and pathologies where hearing is present and preservation is planned. In cases where hearing preservation is not a consideration we advocate a translabyrinthine approach, which in our experience offers the lowest risk to the facial nerve.
Technique for endoscopic skull base surgery
The following describes the technique for endoscopic skull base surgery.
- •
Anesthesia is typically positioned at the patient’s feet to allow adequate room for placement of the monitors and sterile tables around the patent’s head.
- •
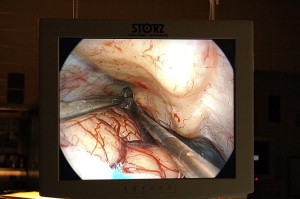
The endoscope monitor should be positioned so that it is in a direct line of sight for the surgeon as he or she is working ( Fig. 11 ). Placing the monitor out of the surgeon’s line of sight can be extremely disorienting.