Purpose
To report the visual outcomes and graft survival rates of mushroom keratoplasty for the treatment of postinfectious corneal scars.
Design
Prospective, noncomparative, interventional case series.
Methods
A microkeratome-assisted mushroom-shaped keratoplasty was performed in 31 eyes of 31 patients with a central vascularized full-thickness leukoma, resulting from infectious keratitis of various origin (herpes simplex virus, n = 16; bacteria, n = 10; Acanthamoeba , n = 5), with healthy endothelium. The donor graft consisted of a large anterior stromal lamella (9.0 mm in diameter and ± 250 μm in thickness) and a small posterior button (5 to 6 mm in diameter). Visual acuity, refraction, and endothelial cell density were evaluated before surgery, as well as at 12, 24, and 36 months after surgery, and the postoperative graft survival rate was evaluated.
Results
Three years after surgery, in 26 (83.8%) of 31 patients, best spectacle-corrected visual acuity was 20/40 or better with a refractive astigmatism of 4.5 diopters or less. The endothelial cell count at the last follow-up examination averaged 1584 ± 381 cells/mm 2 , with an average cell loss of 40.7% from the preoperative value. The survival rate at 3 years was 90.3%, improving to 96.7% when excluding nonimmunologic causes for graft failure.
Conclusions
Similarly to penetrating keratoplasty, microkeratome-assisted mushroom keratoplasty restores vision in eyes with postinfectious, full-thickness, central corneal scars. For these vascularized corneas at high risk for immunologic rejection, mushroom keratoplasty combines the visual and refractive advantages of large penetrating keratoplasty grafts with the high survival rate of small penetrating keratoplasty grafts.
Corneal scarring resulting from herpes simplex virus (HSV) or microbial keratitis is one of the main indications for penetrating keratoplasty (PK) in developing countries and is a relatively frequent one also in developed countries. Because deep stromal scars of infectious origin usually affect Descemet membrane and endothelium, to date PK is the standard surgical procedure performed to rehabilitate these patients visually. However, long-term survival of full-thickness standard grafts (7.5 to 8.5 mm in diameter) in vascularized corneas is jeopardized by immunologic rejection as well as possible recurrence in herpetic cases. In the past, small full-thickness grafts (5 to 6 mm in diameter) performed in vascularized corneas have shown higher survival rates than larger grafts (7 to 8 mm in diameter), but were associated with high-degree and irregular postoperative refractive errors, thus making visual rehabilitation practically impossible.
More recently, several deep anterior lamellar keratoplasty (DALK) techniques have been proposed to preserve the healthy endothelium of patients with postinfectious corneal scars. However, reduced visibility makes hand dissection particularly difficult in these eyes, whereas the frequent presence of adhesions between deep stroma and Descemet membrane does not allow easy formation of a big bubble when attempting pneumatic dissection, as in keratoconic patients. In addition, femtosecond lasers cannot obtain smooth surfaces when dissecting through nontransparent corneas, and therefore cannot be used successfully for this purpose. Most of all, even if descemetic dissection is obtained, the Descemet membrane and endothelium underlying the central stromal opacity often also are affected centrally, thus preventing full visual recovery if left in place.
We propose a microkeratome-assisted procedure involving transplantation of a relatively small central area of endothelium and deep stroma in conjunction with a large anterior stromal lamella. The resulting mushroom-shaped 2-piece graft shares the refractive advantages of a large anterior lamellar keratoplasty and that of limited removal of the central recipient endothelium (approximately 25% to 35% of the total), while obtaining a central scar-free optical zone of 5 to 6 mm in diameter. We present herein the 3-year results of a prospective ongoing study evaluating the outcome of mushroom keratoplasty performed in 31 patients with vascularized postinfectious central scars in corneas with otherwise healthy endothelium.
Methods
We recorded the results obtained in all patients undergoing mushroom keratoplasty surgery by the same surgeon (M.B.) at our hospital between January 2004 and December 2006 and followed up prospectively thereafter. The main inclusion criterion was the loss of visual acuity resulting from postinfectious deep corneal scars involving the optical zone in the presence of vascularization and otherwise healthy endothelium; in addition, surgery could not be performed unless a period of 1 year or longer without episodes of reactivation of the underlying disease or inflammation had elapsed. All surgical procedures were performed in a standard fashion as described in detail below.
Before surgery, every patient underwent a complete ophthalmologic evaluation, including slit-lamp examination, both uncorrected and best spectacle-corrected visual acuity, as well as refraction. Corneal vascularization was evaluated by slit-lamp examination and was rated according to extension (number of clock hours involved) and depth (superficial or deep stroma). In addition, at all postoperative examination times (12, 24, and 36 months), corneal topography analysis (EyeSys 2000; EyeSys Technologies, Inc, Houston, Texas, USA) and endothelial cell count (cornea module of HRT-II; Heidelberg Technology, Heidelberg, Germany) were performed. Postoperative endothelial cell density (ECD) was evaluated centrally, that is, in the area of the posterior donor button; the values recorded were compared with those obtained before surgery from the eye bank, thus considering the cell loss as a percentage of the preoperative in vitro value, as described in a previous article from our group.
An analysis of variance was used to determine the significance of changes in ECD values at different postoperative examination times. Analyses were conducted using Stata software version 10.0 (Stata Corp, Inc, College Station, Texas, USA). A P value of less than .05 was considered to be statistically significant.
Surgical Procedure
All but 2 patients who received general anesthesia because they were younger than 14 years were sedated with 3 mL intravenous droperidol immediately before anesthetic injection. Local anesthesia was administered with a peribulbar injection of a mixture of lidocaine hydrochloride 2% and bupivacaine hydrochloride 0.5%. All steps of the surgical technique are shown in the Supplemental Video (Supplemental Material available at AJO.com ).
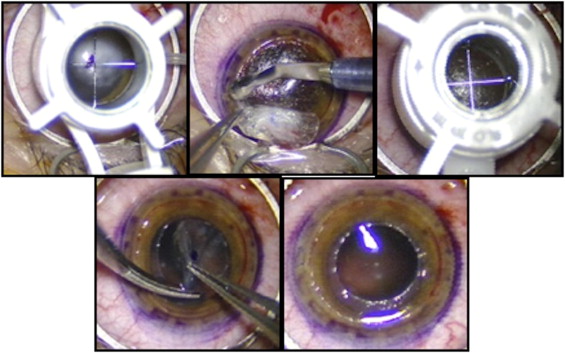
Initially, a Barron suction trephine (Katena Products, Inc, Denville, New Jersey, USA) was used to obtain a circular incision 9.0 mm in diameter and approximately 250 μm in depth in the recipient cornea ( Figure 1 , Top left). Then, a lamellar dissection was carried out by hand from the base of the incision toward the center of the cornea, and the anterior lamella was removed from the recipient cornea ( Figure 1 , Top middle). A trephine 5 to 6 mm in diameter was used to make a full-thickness circular incision in the residual recipient bed, taking particular care to center the incision on the pupil ( Figure 1 , Top right). The central button then was excised with corneal scissors ( Figure 1 , Bottom left and right). At this stage, when necessary, an open-sky extracapsular cataract extraction was performed and an intraocular lens was implanted into the capsular bag.
The donor cornea was mounted on the artificial anterior chamber of the automated lamellar therapeutic keratoplasty system (Moria SA, Antony, France); a 200-μm head was used to split the donor cornea into anterior and posterior lamellae ( Figure 2 , Top left), which then were both punched to proper size ( Figure 2 , Top middle and right) to fit with the recipient bed prepared previously (the same size of the recipient bed was used both for the anterior and the posterior lamellae). To prepare a donor lamella with a thickness similar to that of the excised one, a 200-μm head was used, basing this selection on previous published data showing that microkeratome-assisted dissection creates a donor lamella, the thickness of which in general is thicker (10% to 20%) than the intended value, based on the width of the head slit.
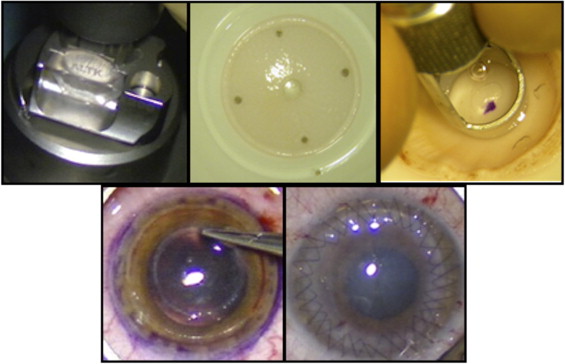
The donor stem (endothelium and deep stroma) was fitted into the central hole of the recipient bed without sutures ( Figure 2 , Bottom left). The 9-mm donor anterior lamella then was placed on top of the posterior one and was sutured into position with 4 cardinal 10-0 nylon stitches. Surgery was completed with a double-running 10−0 nylon suture ( Figure 2 , Bottom right). The bites of the sutures were passed only through the anterior lamella, leaving the posterior one free to adapt. Finally, the anterior chamber was filled with balanced salt solution injected with a 30-gauge needle through a limbal puncture.
Based on treatment regimens previously used for patients at high risk for immunologic rejection, steroids were given systemically (1 mg/kg body weight prednisolone tapered off over a 3-month period) and topically (dexamethasone 0.1% eye drops every 2 hours and tapered off to daily over a 5-month period). Systemic acyclovir (400 mg twice daily) was given for at least 1 year to all patients with herpetic keratitis. In all cases, all sutures were removed within 12 months after surgery.
Results
Thirty-one phakic or pseudophakic eyes of 31 patients were included in this series. The average age at surgery in the study was 38.3 ± 18.4 years (mean ± standard deviation), ranging from 7 to 76 years. Eighteen patients were males (58.0%) and 13 patients were females (42.0%).
Sixteen scars (51.6%) were of herpetic origin ( Figure 3 , Top left and right), 10 scars (32.2%) of bacterial origin ( Figure 3 , Middle left and right), and 5 scars (16.1%) resulted from acanthamebic infection ( Figure 3 , Bottom left and right); in all eyes, corneal deep vascularization was present, with 3 clock hours or more involvement. In 2 (6.4%) eyes with cataract, a mushroom keratoplasty was combined with an open-sky extracapsular cataract extraction and intraocular lens implantation. All procedures were uneventful, no recurrence of infection occurred in patients with microbial keratitis, and all grafts but 3 were clear at the final examination. In 2 eyes, progressive lens opacification developed and a standard phacoemulsification with intraocular lens implantation in the capsular bag was performed between 6 months and 1 year after mushroom keratoplasty; in one of them, the surgical trauma caused severe endothelial cell loss requiring regrafting. One patient experienced an episode of allograft rejection revealed by central corneal edema and endothelial precipitates; treatment with topical and systemic steroids failed to control the rejection and the graft decompensated. One eye with glaucoma unresponsive to maximal medical treatment underwent repeat cyclocryocoagulation, but phthisis bulbi eventually occurred.
Visual results are summarized in Figures 4 and 5 . Thirty-six months after surgery, best spectacle-corrected visual acuity was 20/40 or better in 26 (83.8%) eyes, ranging between 20/200 and 20/20. The mean refractive astigmatic error was within 4.50 diopters in 26 of 31 cases, averaging 3.10 ± 1.59 diopters.




