Chapter 44 Surgical Pathology of the Thyroid Gland
Introduction
In most thyroid lesions that involve surgeons, the presentation of the clinical problem is a single or dominant nodule in the gland. On occasion, the clinical presentation will be an enlarged cervical lymph node.1–4
This chapter concentrates on specific pathologic entities in thyroid nodules and neoplasms. Each major thyroid tumor is discussed in individual chapters in this book, and the reader is referred to those chapters for additional information on these entities (see Chapters 18, Papillary Thyroid Cancer, 19, Papillary Thyroid Microcarcinoma, 20, Follicular Thyroid Cancer, 22, Hurthle Cell Tumors of the Thyroid, 23, Sporadic Medullary Thyroid Carcinoma, 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, 25, Sporadic Medullary Thyroid Microcarcinoma, and 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma).
All neoplasms that arise from thyroid epithelial cells can have some functional capacities. They may respond to TSH and even produce excessive amounts of thyroid hormones or, if medullary carcinoma, may release abnormal quantities of calcitonin or other peptide hormones.1 Immunohistochemical localization of thyroid transcription factor-1 (TTF-1), thyroglobulin, or calcitonin aids in the classification of unusual thyroidal tumors and in providing a definitive identification of metastatic thyroid carcinomas.5,6
Benign Neoplasms: Adenomas and Adenomatous Nodules
The definition of a follicular adenoma is controversial. Many pathologists believe that follicular adenoma is a solitary, encapsulated lesion with a uniform internal architecture that is substantially different from the surrounding thyroidal parenchyma by both growth pattern and cytologic features.1–3
However, we believe that this exact definition of adenoma is too restrictive. In some instances it is difficult to clearly separate hyperplastic/adenomatous nodules arising in the background of goiter or thyroiditis from adenomas; up to 70% of the hyperplastic nodules in goiter are clonal, thus representing neoplastic proliferations.7–9 Therefore, we propose that an adenoma should be defined as a follicle-derived encapsulated nodule with a distinct growth pattern limited to the confines of its capsule that is different from the surrounding thyroid parenchyma. These follicular lesions can be rarely multiple and arise in a background of normal thyroid or in the setting of nodular goiter, toxic goiter, and thyroiditis.10,11
Grossly, adenomas and nodules are well circumscribed and are often demarcated from the adjacent tissue. Their size varies from about 1 mm in diameter to several centimeters. A classic adenoma is fleshy and pale; hemorrhage, fibrosis, and cystic change may be evident.3,12
Microscopically, an adenomatous nodule shows a varied pattern of large and small follicles, usually with abundant colloid. The cells range from flat to cuboidal or columnar with small, round nuclei with even chromatin pattern. The stroma of the nodule often appears edematous. Macrophages, lymphocytes, hemosiderin, fibrosis, and even calcification can be found. Cystic change is common, especially in adenomatous nodules, and is frequently accompanied by the formation of papillae. Occasionally, benign lesions are hyperfunctional, or “hot”; usually this occurs with nodules of nodular goiter rather than with a classic adenoma. In adolescents, especially females, many of the hot, or toxic, nodules contain numerous papillae, often sufficient in number to cause a pathologist to suggest a diagnosis of papillary carcinoma.11–13
In the era of fine-needle aspiration (FNA), some cases of adenomatous nodules and adenomas, especially of oncocytic follicular (Hurthle) cell type, may exhibit prominent post-FNA changes, which include focal hemorrhage, fibrosis, endothelial proliferation, pseudo-vascular and capsular invasion, and even partial or total infarction.14
By immunohistochemistry all adenomatous nodules and adenomas express thyroid transcription factor (TTF-1), thyroglobulin, and show cytokeratin expression similar to normal thyroid parenchyma.15,16
Several studies have suggested the use of different biologic markers to differentiate between follicular adenoma and carcinoma. It has been shown that follicular adenomas usually do not express p53, CA 19–9, PAX-8-peroxisome proliferator-activated receptor-gamma (PPAR-gamma) translocation, or RAS mutation, and they show lower levels of Ki-67 and bcl-2 as compared to follicular carcinoma.17–19 In addition, up to 45% of follicular adenomas in patients with history of radiation exposure can express ret-oncogene rearrangements as compared to 85% of radiation-induced papillary carcinomas.20,21 These above-mentioned studies do highlight some differences between adenomas and carcinomas; however, they are not specific. Therefore, one should rely on morphologic criteria alone to differentiate between follicular adenoma and carcinoma at this time.
Variants of Follicular Adenoma
Atypical Follicular Adenoma
This term is reserved for follicular lesions that exhibit some atypical features including foci of necrosis, excess cellularity, mitoses, and lack of capsular or vascular invasion. However, all cases diagnosed as atypical adenoma have been shown to behave in a benign fashion.22,23
Hyalinizing Trabecular Neoplasm of the Thyroid AKA Hyalinizing Trabecular Adenoma (HTA)/Paraganglioma-Like Adenoma of the Thyroid (PLAT)
The hyalinizing trabecular adenoma is a follicle-derived lesion that has a distinctive histology.24,25 Microscopically, these adenomas grow in nests that are surrounded by dense hyaline stroma. The histology is reminiscent of that seen in paragangliomas; however, the tumor is derived from the follicular epithelium. The nuclear features of the follicular cells are similar to those seen in papillary carcinoma. By immunohistochemistry, the cells of hyalinizing trabecular adenoma stain positive for thyroglobulin and cytokeratin-19, and they stain negative for calcitonin, although the presence of other neuroendocrine markers has been described.26,27
Some authors have proposed that these adenomas actually represent a variant of papillary carcinoma.28 This is due to similar nuclear cytology, immunoprofile, and RET-oncogene rearrangements in both tumors.29 However, a benign behavior has so far been described in all cases of hyalinizing trabecular adenoma that show classical histology. Because of controversy regarding their nature and clinical behavior, these tumors have been designated as hyalinizing trabecular neoplasms by the World Health Organization (WHO) classification schema.30
Signet Ring Cell Follicular Adenoma
This tumor is characterized by the presence of large vacuoles in the cytoplasm of follicular cells, causing peripheral displacement of the nucleus leading to signet ring cell formation. A similar morphologic change also can be observed in hyperplastic nodules arising in the background of nodular goiter. Immunohistochemistry has shown that these vacuoles contain thyroglobulin, which is biochemically and physicochemically altered; in rare instances the cytoplasm may also show mucin positivity.31
Malignant Neoplasms
The most common malignant neoplasms that originate in the thyroid are well-differentiated carcinomas of follicular cell origin; most are papillary carcinomas.3
Most non-neoplastic diseases of the thyroid are not precursors of malignant diseases; autoimmune thyroiditis is an important exception and may predispose to malignant lymphoma.32 Rarely, an adenoma or adenomatous nodule may contain a focus of papillary carcinoma.3,33
Anaplastic carcinomas can arise in goitrous thyroids, and careful examination of the resected tissues may demonstrate benign tumors or well-differentiated carcinomas in close association with the carcinoma. Such findings suggest that the benign tumor or low-grade carcinoma has become “transformed” into the anaplastic carcinoma.34,35
In modern endocrine practice, the initial approach to thyroid nodules is fine-needle aspiration; this technique can triage lesions, which must be removed surgically. However, about 20% of cases will yield an indeterminate diagnosis (the topic of FNA is discussed elsewhere in this book; see Chapter 12, Fine-Needle Aspiration of the Thyroid Gland).36
Frozen sections can be helpful in papillary cancer, but follicular and Hurthle cell tumors cannot be definitively diagnosed by frozen section.37,38
Papillary Carcinoma
About 80% of thyroid carcinomas in countries with sufficient iodine in the diet are papillary carcinomas (PTCs) (see Chapters 18, Papillary Thyroid Cancer, and 19, Papillary Thyroid Microcarcinoma).39 They occur more frequently in women than in men and are rarely familial.39,40 In addition, most thyroid cancers in the pediatric population can be classified as papillary cancers.41 The association of radiation, especially low-dose external radiation, in childhood with the development of adult papillary thyroid cancer is well documented.42,43
Grossly, papillary cancers can present in various sizes and forms. These tumors are predominantly solid, although small cystic foci may be present. Cystic cancers can occur, with one or more cystic spaces occupying most of the neoplasm. Bits of calcified material and crystals may be present in the cyst fluid. Papillae may be so numerous that the cut surface appears granular.2,3,44
Papillary carcinomas can infiltrate the surrounding gland, and their margins often are poorly defined; however, about 10% to 20% of papillary cancers appear grossly encapsulated.9,33 Encapsulation of the primary carcinoma is associated with a lower frequency of lymph node involvement.45,46
Fibrosis is common in and around papillary carcinomas, and it may be distributed in an extremely irregular fashion, grossly and microscopically. Occasionally, fibrosis is so extensive that almost no neoplastic cells can be found. Nonlamellated calcification is also common.47–50
Small papillary carcinomas (microcarcinomas) are defined as 1 cm in diameter or less.30 The prevalence of these lesions in the normal population ranges from 6% to 36% (depending on the population studied and the exact manner in which the thyroid was examined). When visible on gross examination, they present as small, irregular, firm scars; as soft foci of discoloration; or as tiny, calcified lesions. Occasionally, the tumor presents as a metastatic focus, usually as an enlarged cervical lymph node; often the node is cystic and clinically mistaken for a branchial cleft cyst. Microscopically, microcarcinomas contain neoplastic follicles or papillae, with the smallest ones showing a predominance of follicular pattern.51–53 They may be encapsulated or infiltrative.51
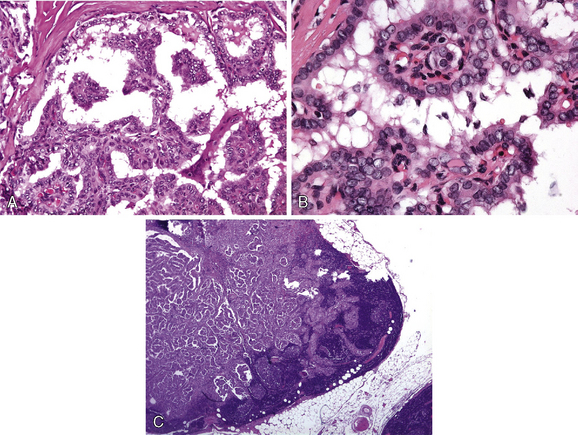
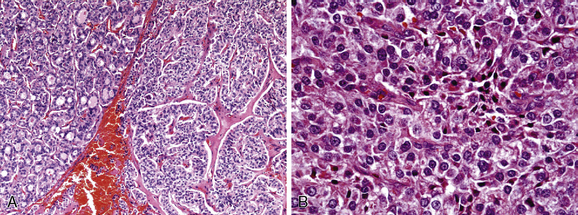
Histologically most papillary cancers contain papillae; however, papillae may constitute only a small part of the neoplasm. Papillary cancer may show a solid pattern, may be composed of follicles (follicular variant), or may be almost entirely papillary (Figure 44-1, A). The papillae contain well-developed fibrovascular cores and are covered with a single layer of the epithelial cells.2,3,54
In papillary carcinoma, the epithelial cell is usually cuboidal to low columnar and contains a distinctive nucleus. The latter is relatively large and irregular in shape, with folds/grooves, indentations and cytoplasmic inclusions (Figure 44-1, B). The nucleolus is often inconspicuous and eccentric in location (lies near the nuclear membrane).54,55 The nuclear heterochromatin tends to be concentrated near the nuclear membrane, causing the central portion of the nucleus to appear relatively pale, empty, or as “ground glass.”55,56 When the cells form papillae or follicles, often most of the cytoplasm is concentrated in the apical or basal portions of the cells, thereby causing neighboring nuclei to appear to overlap one another.57
About one third to one half of papillary carcinomas form laminated calcific spherules, known as psammoma bodies. They measure 5 to 100 mm in diameter, are found in stroma or lymphatics, and probably begin in damaged or dying cells of papillary carcinomas. Anytime a psammoma body is found in normal thyroid tissue, cervical lymph nodes (Figure 44-1, C), or perithyroidal soft tissue, a search should be made for papillary thyroid carcinoma.58,59 Structures resembling psammoma bodies are occasionally found within the colloid of follicles in adenomas or adenomatous nodules, especially those composed of oxyphilic cells, where they seem to arise from calcification of inspissated colloid; these structures should not be equated with psammoma bodies.60,61
Lymphocytic infiltration is often present within and around papillary carcinomas.
Multiple intrathyroidal foci of tumor and metastasis to cervical lymph nodes are common and probably are a direct result of lymphatic invasion by tumor.62–65 Occasionally, cervical nodal enlargement is the presenting sign of metastatic papillary; this is especially found in young males.66 If cervical nodal metastasis is cystic, it must be differentiated from a branchial cleft cyst.67 The nuclear features of papillary carcinoma may not be present in differentiated follicular-patterned metastases.
Blood vessel invasion by papillary carcinoma is uncommon, and metastatic foci in distant sites are unusual (about 5% to 7% of cases), with the lungs most frequently involved.45,68
Variants of Papillary Cancer
Follicular variant of papillary carcinoma (FVPTC) is the most common histologic variant of papillary carcinoma after the usual or classic variant; it is characterized by formation of follicles lined by cells with nuclear features of papillary carcinoma. FVPTC clinically behaves similar to the conventional papillary carcinoma.69,70 At least four distinct variants of FVPTC have been described. The first of these is a tumor that is unencapsulated and infiltrative, thus resembling the growth pattern of classic papillary carcinoma. The difference is that the tumor is totally composed of follicles; these have the appropriate nuclei for the diagnosis of papillary cancer (Figure 44-2). These neoplasms spread and behave as ordinary papillary carcinoma, with multifocal involvement of the gland and lymphatic and nodal metastases. Indeed, in the nodal metastases, a papillary pattern may be found.
The diffuse follicular variant leads to total replacement of thyroid by tumor. Both lymph node and distant metastases (pulmonary) are common in this variant.71 The encapsulated variant usually behaves in an indolent fashion. The encapsulated follicular variant refers to the follicular variant, which is characterized by the presence of a capsule around the lesion and is associated with an excellent prognosis.12,46 In some cases the diagnosis of this particular variant of papillary carcinoma can be difficult due to presence of a multifocal rather than diffuse distribution of nuclear features of papillary thyroid carcinoma.12 Because of this peculiar morphologic presentation, these tumors can be misdiagnosed as adenomatoid nodule or follicular adenoma.12 Some authors have suggested that these tumors be classified as “tumors of undetermined/uncertain malignant potential” because of the excellent prognosis72; however, others have shown that some cases belonging in this category can lead to distant metastasis.73
An occasional FVPTC can be composed almost entirely of distended, colloid-filled follicles of moderately uniform size and shape, thereby closely resembling a common pattern of an adenomatoid nodule; this type of tumor is referred to as a macrofollicular variant of papillary carcinoma. In this variant of FVPTC, rare papillae, an occasional focus of infiltration at the periphery, and the characteristic nuclei confirm the diagnosis.74
Though FVPTC is a variant of papillary carcinoma (based on cytology and clinical behavior), it does share some morphologic and clinical features with follicular carcinoma. These include follicular growth pattern, encapsulation, capsular and vascular invasion, and distant metastases to lung and bone. Gene expression profiling studies have shown that some cases of FVPTC will show similar profile to follicular carcinoma.75 Comparative genomic hybridization analysis showed that the presence and pattern of chromosomal aberration in FVPTC were significantly different from those in classical PTC, but more comparable to follicular adenoma and follicular carcinoma. RAS gene mutations, an abnormality seen in follicular adenoma and carcinoma, are exclusively seen in FVPTC and not in classical PTC;75,76 similarly RET gene translocations and BRAF mutations, which are common in classic PTC, are rare in cases of FVPTC.75–77 Therefore, in view of morphologic features, clinical behavior, and genetic analysis, it is not unreasonable to hypothesize that FVPTC cases, especially of the encapsulated type, may represent a hybrid of papillary carcinoma and follicular adenoma or carcinoma. A reclassification scheme based on this hybrid theory would have significant prognostic and therapeutic implications: encapsulated FVPTC without any capsular and vascular invasion (if the tumor is well sampled) will behave more as a follicular adenoma, and the ones with capsular and vascular invasion will behave as follicular carcinoma.
Papillary carcinoma may be largely or exclusively solid and is termed solid variant. It is not known if this pattern may affect the prognosis in young people. In middle-aged and elderly patients, solid growth pattern may be associated with a loss of differentiation that suggests an aggressive neoplasm. Follicles or papillae may be rare or nonexistent in the primary focus, although they may be present in lymph node metastases.78
Rarely, papillary thyroid carcinoma involves all lymphatic channels of one lobe or of the entire thyroid and is accompanied by severe lymphocytic thyroiditis with interstitial fibrosis. Psammoma bodies are numerous. A primary mass lesion (epicenter) may not be identified. This diffuse sclerosis variant occurs more often in young people, is virtually always accompanied by lymph node metastases, and often has pulmonary metastases.79
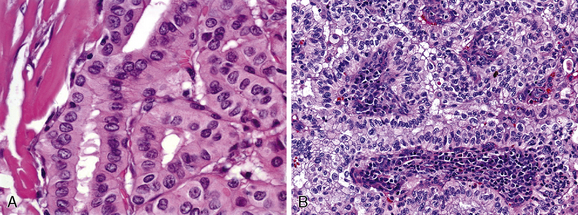
The tall cell variant is an unusual type of papillary carcinoma (about 10%) that appears to be more aggressive than the usual variety (some authors dispute the aggressive behavior of this tumor). The cells in this type are narrow and elongated (three times as long as they are wide) and often are oncocytic (Figure 44-3, A). These oncocytic tumors often show extrathyroidal soft tissue extension and vascular invasion (20% to 25%). Most occur in older persons, on average 20 years older than patients with usual papillary cancer. A mortality rate of 25% is reported.80–82 Columnar cell variant is a rare tumor composed of clear cells with marked nuclear stratification. Although original reports indicated an aggressive clinical behavior, recent studies of tumors confined within the thyroid indicate a more indolent course.83,84
Rarely, a papillary carcinoma is composed of oxyphilic cells (oncocytes, Hurthle cells) and arises in a thyroid altered by lymphocytic thyroiditis. These lesions may show central cystic change. Morphologic resemblance to a benign parotid lesion led to our designating this lesion as Warthin-like papillary carcinoma. This variant appears to have the same spectrum of behavior as the conventional variety85,86 (Figure 44-3, B).
Papillary carcinoma on occasion can demonstrate easily found mitotic figures, enlarged or hyperchromatic nuclei, abnormal DNA content, necrosis, and regions of nondescript neoplastic cells often growing in sheets (loss of differentiation). These characteristics may indicate that more aggressive behavior is likely.87 These features occur most often in the cancers diagnosed in older people and in those tumors that are extrathyroidal.87,88
Immunohistochemistry of Papillary Carcinoma
Almost all papillary cancers express TTF-1 and thyroglobulin. Papillary cancers show a different cytokeratin profile than the normal thyroid and other follicular lesions. It has been shown that immunostains for cytokeratin-19 can be helpful in the diagnosis of papillary carcinoma. A mesothelium-derived marker HBME-1 can be expressed in up to 68% of the papillary thyroid carcinomas.15,16 The other antigen studied in papillary cancers include Galectin-3, S-100, neuroendocrine markers, CD44, CD57, CA-125, and so on.15,16,89 However, despite these extensive studies the morphology remains the single most important tool for the diagnosis of this tumor.15,16,89
Molecular Pathology of Papillary Carcinoma
Recent literature on the thyroid has focused mainly on the role of various biologic events and genetic determinants in the pathogenesis of various thyroid tumors (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia). Rearrangements of the RET gene, known as RET/PTC, have been identified in papillary carcinoma of the thyroid.90–92 In normal thyroid, wild-type RET is only expressed in C cells and not follicular cells.91 RET/PTC seen in papillary carcinomas occurs because of fusion of tyrosine kinase domain of RET to the 5′ portion of the various genes.93,94 To date, at least 10 novel types of rearrangements have been described in papillary carcinoma. RET/PTC 1 and 3 are the most common forms that occur in sporadic papillary carcinoma. The prevalence of RET/PTC in papillary carcinoma varies significantly among various geographic regions94–96; in the United States it ranges from 11% to 43%.94 In sporadic tumors, RET/PTC1 is the most common form of rearrangement (60% to 70%) followed by RET/PTC3 (20% to 30%). The other rare forms of RET/PTC rearrangements have been mainly found in radiation-induced papillary carcinomas. Several studies have shown a strong association between radiation-induced papillary carcinoma and the expression of RET/PTC; in papillary carcinoma found in children affected by the Chernobyl nuclear accident, RET/PTC3 was found to be the most common form of rearrangement, followed by RET/PTC1.43,94,97
Recently it has been shown that RET/PTC expression can also occur in some benign lesions. These include hyalinizing trabecular neoplasm/adenoma,29 hyperplastic nodules (especially the ones that are seen in patients with a history of external radiation), and follicular adenoma.98,99 Some investigators have shown a high prevalence of RET/PTC in histologically benign thyroid tissue affected by Hashimoto’s thyroiditis; these studies concluded that thyroiditic glands harbor multiple foci of papillary carcinoma, which are not identified by histologic examination only.100,101 However, others have disputed these findings and failed to reproduce these results.102
It is well known that the RAF/MEK/ERK pathway is a significant contributor to the malignant phenotype associated with deregulated RAS signaling.103 An activating mutation in BRAF was described in human papillary thyroid cancers. BRAF activating mutations in thyroid cancer are almost exclusively the BRAF V600E mutation and have been found in 29% to 69% of papillary thyroid cancers, 13% of poorly differentiated cancers, and 10% of anaplastic cancers.104–106 Other rare BRAF mutations found in papillary carcinoma are BRAF K601E, AKAP9-BRAF, BRAF V599ins BRAF V600E+K601del, and V600D+FGLAT601–605ins.107 Interestingly, presence of BRAFV600E correlates with variants of PTC. There is a gradual increase in the presence of the mutations from the follicular variant of PTC to the classic variant, tall cell, and columnar variant. More recent studies consisting of large cohorts of patients have shown a strong correlation of BRAF mutation with nonfavorable clinicopathologic features.7,108
There is practically no concordance between papillary carcinoma with RET/PTC translocations and BRAF or RAS mutations. BRAF mutational analysis of FNA samples has been shown to be of value in the preoperative diagnosis of papillary thyroid carcinoma in cases diagnosed as indeterminate or suspicious for malignancy.109,110
Follicular Carcinoma
Follicular carcinoma accounts for about 5% or less of all thyroid carcinomas in the United States (see Chapter 20, Follicular Thyroid Cancer). It is more common in women than in men and occurs more frequently in patients over 30 years of age.2,111 This tumor is more often found where iodine deficiency occurs.112,113
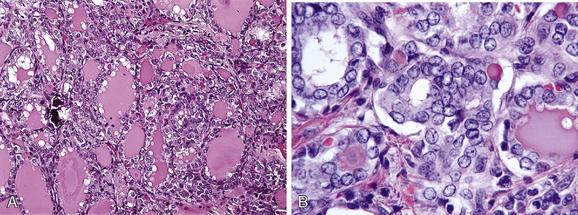
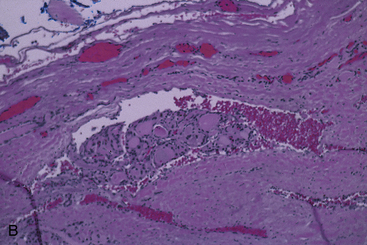
Follicular carcinoma is an expansile neoplasm that nearly always is encapsulated and grossly resembles follicular adenoma. It presents as a fleshy, solid, encapsulated mass, sometimes with focal fibrosis and calcification. The capsule is usually well developed, but in some examples, extensions beyond the capsule may be noted. Sometimes invasion of veins can be appreciated in or outside of the tumor capsule (see Figure 44-4).2,111,114
On microscopic examination, follicular carcinomas most often have a microfollicular pattern and resemble a cellular follicular adenoma; rarely, the tumor may exhibit a macrofollicular growth pattern. Trabecular or solid patterns are also fairly common. The cells of follicular carcinoma are slightly to moderately larger than those present in most adenomas and adenomatous nodules, but otherwise they are similar. Mitotic figures range from rare to easily identified.70,111,115
Follicular carcinomas have been divided traditionally into (1) localized, minimally invasive cancers and (2) more widely invasive cancers.70,116 Because follicular carcinomas are nearly always encapsulated, the distinction between adenoma and minimally invasive carcinoma may be difficult.12,111 Carcinoma is recognized by its penetration into and through the capsule that surrounds it, by its extension into vessels at its periphery, and (occasionally) by the presence of distant metastasis. Even a minimally invasive carcinoma can present as a metastatic lesion.117–119 Multiple sections of the periphery of the neoplasm may be necessary to find the invasion, although most follicular cancers are diagnosed on examination of 10 different sections of the capsule-tumor interface. Whether the number of invaded veins can be correlated with outcome is unclear.111,120,121
The minimally invasive neoplasms rarely recur or spread to distant sites, so the outlook for most patients is good.126–129 However, data on long-term prognosis for minimally invasive follicular carcinoma are not available, because much of the clinical literature fails to differentiate FVPTC from follicular carcinoma. Follicular carcinoma has little tendency to invade lymphatic vessels and to spread to lymph nodes. Hematogenous spread to the skeleton, lungs, brain, liver, and other tissues occurs.122,123
Some follicular carcinomas that are not localized or minimally invasive have been called widely invasive; these include examples in which multiple fingers of neoplastic cells extend into the surrounding thyroid or in which there is extensive replacement of the thyroid gland and soft tissues of the neck noted grossly.123
Post-FNA effect may mimic capsular “invasion” and “penetration.” Histologic clues to differentiating between true invasion and pseudo-invasion include the geographic linear pattern of FNA-induced pseudo-invasion and the presence of granulation tissue, macrophages (many containing hemosiderin), cholesterol clefts, and, rarely, well-formed granulomas.124
Molecular Pathology of Follicular Carcinoma
A specific translocation t (2;3) leads to the expression of PAX-8-peroxisome proliferator-activated receptor-gamma (PPAR-gamma) chimeric protein; initial studies by Kroll et al. demonstrated this translocation is specific to follicular carcinoma (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia).17 However, follow-up studies employing immunohistochemistry and molecular biology have shown that PPAR-gamma expression can occur in some cases of follicular adenoma, follicular variant of papillary thyroid carcinoma, and even benign thyroid parenchyma.19,125 RAS mutations are more frequent in follicular carcinoma as compared to follicular adenoma; some authors have found an association between RAS mutations and clinically aggressive follicular carcinomas.21,126,127 Loss of heterozygosity on chromosome 10q and 3p can be seen in follicular carcinoma, suggesting a role of tumor suppressor genes in its pathogenesis.128,129
Well-Differentiated Follicular “Tumors of Undetermined/Uncertain Malignant Potential”
This designation has been proposed in thyroid pathology for follicle-patterned encapsulated tumors that have been controversial and difficult to diagnose because of (1) questionable or minimal nuclear features of papillary thyroid carcinoma or (2) questionable or one focus of capsular invasion that is confined to tumor capsule and does not traverse the entire thickness of the capsule and lacks any nuclear features of papillary thyroid carcinoma.72
This terminology may be extremely helpful to pathologists in the diagnoses of certain follicular patterned lesions; however, these terms are proposed on the basis of data that lack complete clinical follow-up. Therefore, clinicians may find it problematic to establish treatment strategies.12
Oncocytic (Hurthle Cell) Tumors
Oncocytic (Hurthle cell) neoplasms are still the subject of debate (see Chapter 22, Hurthle Cell Tumors of the Thyroid).130 However, many studies from numerous institutions throughout the world have shown that oncocytic or Hurthle cell tumors can be divided into benign and malignant categories by careful adherence to strict pathologic criteria. More important, these pathologic distinctions predict clinical behavior.30,131,132
Most oncocytic (Hurthle cell) neoplasms behave as follicular carcinomas—that is, pathologically the capsule or vessels should be assessed for invasion.131,132 However, some papillary carcinomas show oncocytic cytology and appropriate nuclear features; these behave as usual papillary cancers.133
Oncocytic follicular (Hurthle cell) carcinomas should be separated as a category of thyroid neoplasms that is different from true follicular cancers. They can metastasize to regional lymph nodes as well as spread hematogenously; in addition, histologic evidence of invasive characteristics is found more commonly in oncocytic cancers than in nononcocytic follicular tumors.23 Approximately one third of oncocytic thyroid tumors show invasion (i.e., are cancers) as compared with 2% to 5% of nononcocytic follicular tumors. Hence, the finding of oncocytic (Hurthle cell) cytology in a fine-needle aspiration sample of a thyroid nodule should lead to surgical resection of the lesion to assess malignancy.134 Size is an important feature; large oncocytic (Hurthle cell) tumors (4 cm or greater) have an 80% risk of malignancy.131,134,135
It has been shown that oncocytic follicular (Hurthle cell) tumors are biologically different than other follicular-derived tumors. H-RAS mutations are more frequent in oncocytic follicular (Hurthle cell) carcinoma than follicular carcinoma.136,137 Maximo et al. studied the relationship between mitochondrial DNA alterations and thyroid tumorigenesis. This study showed that oncocytic follicular (Hurthle cell) tumors display a relatively higher percentage of common deletions of mitochondrial DNA as compared to other follicular tumors. In addition, oncocytic follicular (Hurthle cell) tumors also showed germline polymorphisms of ATPase 6 gene, which is required for the maintenance of mitochondrial DNA.138
Insular Carcinoma
Most authors classify insular carcinoma as merely a form of poorly differentiated follicular carcinoma.139 Histologically the tumor is composed of islands and nests of small cells within a richly vascular stroma. Necrosis, high mitotic rate, and lymphovascular invasion are common.139,140
Many examples show differentiated areas, including papillary carcinoma or its follicular variant. If insular carcinoma occurs as a recurrence of another thyroid tumor, the latter is usually papillary.139,141,142 Hence, these associations with papillary cancer and the ability for regional nodal metastasis should place insular cancer in a category different from true follicular carcinoma.
Clinically these tumors are aggressive with regional and distant (hematogenous) metastasis in over 50% of cases and a 5-year mortality rate of about 50% to 60%.143
Poorly Differentiated Carcinoma
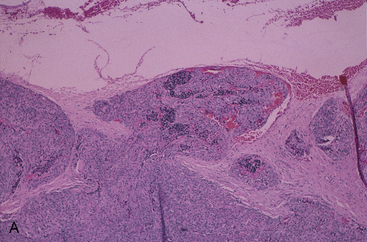
Poorly differentiated thyroid carcinomas are a heterogeneous group of tumors that take their place between well-differentiated follicular/papillary and anaplastic carcinomas. They have been included in the 2004 WHO classification of thyroid tumors; however, a controversy still exists regarding their nature, morphologic diagnostic features, clinical significance, and management.141,144 Studies of thyroid carcinomas classified as “poorly differentiated carcinoma” have often included examples of tumors that are recognizable as originating from follicular epithelium (often with evidence of coexistent papillary or follicular carcinoma), but with some notable differences: moderate to high rates of mitotic activity, composed of solid masses or trabeculae of relatively uniform epithelial cells, tiny follicles present in varying numbers, regions of acute necrosis, and more aggressive than usual well-differentiated carcinomas145 (Figure 44-5). Included among these lesions are insular carcinoma; columnar cell, tall cell, and trabecular types of papillary cancer; and “poorly differentiated” carcinoma of Sakamoto.145 These tumors generally lack the usual histologic features and exceptional aggressiveness of anaplastic carcinomas, but they are neither typical follicular nor papillary carcinomas.146,147 At present most experts agree that the common pathologic features of poorly differentiated carcinomas are solid/trabecular/insular growth, large size, frequent extrathyroidal extension, extensive vascular invasion, presence of necrosis, and increased mitotic activity. They may be associated with well differentiated components, of either follicular or papillary type, and less frequently with anaplastic carcinoma.87,141A recent study from the Memorial Cancer Center in New York identified a group of encapsulated tumors with features of poorly differentiated carcinoma; in this small subset, the survival was better than expected for poorly differentiated thyroid cancer. This suggests that gland-confined encapsulated lesions may not harbor the serious prognosis of those tumors with extrathyroidal extension even in the presence of aggressive morphologic features.148 Literature data, although limited, have shown a distinct molecular pathway in poorly differentiated carcinomas, almost exclusively involving RAS gene alterations.78
Anaplastic Carcinoma
Fewer than 5% of thyroid carcinomas may be classified as anaplastic or undifferentiated (see Chapter 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma).149 They are most common in regions of the world where iodine is deficient. Traditionally, these tumors include spindle-cell and giant-cell cancers and the rare small-cell carcinomas. However, most lesions originally classified as small-cell anaplastic carcinomas represent either medullary carcinoma, insular carcinoma, or small-cell malignant lymphoma.149,150 The possibility of a metastatic carcinoma from another organ always has to be considered.
Most anaplastic thyroid carcinomas are spindle-cell and giant-cell tumors.151 These aggressive neoplasms usually occur in elderly people, more often women. The patient may have a history of a thyroid nodule that, after many years of stability, suddenly begins to grow rapidly. Some patients are known to have had low-grade thyroid carcinoma; others have low-grade thyroid carcinoma discovered at the time of diagnosis of the anaplastic tumor.149,152
Careful pathologic examination of thyroids that contain anaplastic carcinomas has demonstrated a high (50% to 70%) incidence of remnants of well-differentiated follicular or papillary carcinoma or sometimes an adenoma or adenomatoid nodules confirming the clinical impression that anaplastic carcinomas arise out of tumors of low grade.153
Grossly, a hard, pale, infiltrative mass with soft foci of necrosis and hemorrhage is seen. These tumors invade the cervical soft tissues and involve the regional lymph nodes, often by direct extension. Microscopic examination reveals varied histologic patterns, many mitotic figures, and regions of tumor necrosis.154
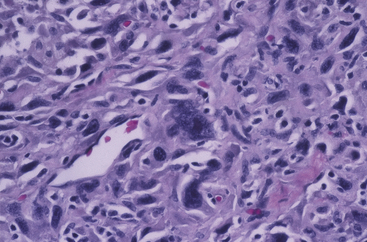
Anaplastic carcinomas are pleomorphic and can be composed of medium-sized to large cells with a vaguely epithelial appearance. There may even be squamous cell differentiation (or a tendency toward this pattern).155 Others appear sarcomatous, resembling malignant fibrous histiocytoma, fibrosarcoma, and angiosarcoma. The giant-cell carcinomas often have bizarre giant cells, frequently multinucleated, and containing abnormal mitotic figures. Less commonly, some giant cells resemble osteoclasts156 (Figure 44-6).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree