Chapter 70 Surgical Pathology of the Parathyroid Glands
![]() This chapter contains additional online-only content, available on expertconsult.com.
This chapter contains additional online-only content, available on expertconsult.com.
Development and Anatomy
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
The parathyroid glands are derivatives of the third and fourth branchial pouches and are first identifiable at 5 to 6 weeks of fetal development as thickenings of the anterodorsal branchial pouch epithelium (seeChapter 2 Applied Embryology of the Thyroid and Parathyroid Glands).1,2 The third pouch gives rise to the lower parathyroids (parathyroid III) and the thymus. Both parathyroid III and the thymus have complex patterns of migration before they assume their final positions caudad to the derivatives of the fourth pouch. Failure of separation of parathyroid III from the thymus results in its appearance in the lower neck within the thymic tongue, the anterior mediastinum, or, more rarely, the posterior mediastinum. Incomplete migration of parathyroid III may result in an ultimate location proximal (i.e., cranial) to parathyroid IV. Because of these patterns of migration, parathyroid III may be present from the angle of the jaw to the pericardium. The upper parathyroids (parathyroid IV) are derived from the fourth branchial pouch together with the ultimobranchial body and are more constant in their final position than is parathyroid III.
Most normal adults have four parathyroid glands, although more than four glands (supernumerary or rudimentary glands) are found in 13% of the population.3 Approximately 3% of normal individuals have been reported to have only three glands, but several studies have suggested that the low glandular weights in such individuals may be related to the fact that not all glands were found. The glands are distributed symmetrically in the neck in 80% of individuals. Grossly the parathyroids are yellow-brown and appear as flattened, bean-shaped structures that measure 4 to 6 mm in length and 2 to 4 mm in width. The average combined glandular weight is 120 ± 3.5 mg for males and 142 ± 5.2 mg for females, with corresponding parenchymal weights of 82.0 ± 2.6 mg and 88.9 ± 3.9 mg, respectively.4
The upper parathyroid glands are present at the junctions of the recurrent laryngeal nerve and the inferior thyroid artery (see Chapters 2, Applied Embryology of the Thyroid and Parathyroid Glands, and 58, Principles in Surgical Management of Primary Hyperparathyroidism).5 Occasionally the upper glands are present within the carotid sheath or in the retroesophageal or retropharyngeal space. In some instances, the glands may be present in the capsule of the thyroid or, more rarely, within the substance of the thyroid gland. Approximately 60% of the lower parathyroids are inferior, posterior, or lateral to the lower pole of the thyroid, whereas 17% are present superiorly, or on the anterior aspect of the thyroid.
The parathyroid glands are encapsulated structures and have a parenchymal component and a fat-rich stromal component.4 The stromal fat cells tend to be concentrated toward the polar regions of the glands. The parenchymal cells are arranged in a cordlike pattern and are separated by stromal elements (Figure 70-1). Parenchymal cells include chief cells, oncocytes, and transitional oncocytes. The chief cells are polyhedral with centrally placed round nuclei with dense chromatin and inconspicuous nucleoli. The cytoplasm of the chief cells is faintly eosinophilic and may occasionally appear clear with moderate numbers of mitochondria and relatively sparse secretory granules at the ultrastructural level.6 Resting or inactive chief cells contain frequent lipid inclusions and prominent deposits of glycogen. The plasma membranes of resting chief cells tend to be straight, whereas increased function is associated with greater tortuosity of the membranes.
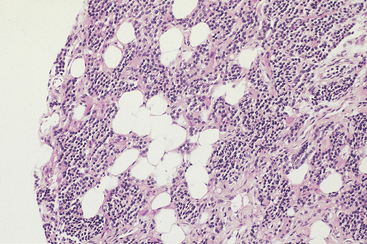
Figure 70-1 Normal parathyroid gland from a young adult, consisting of a mixture of chief cells and stromal fat cells.
Oncocytes are larger than chief cells and have abundant granular eosinophilic cytoplasm filled with mitochondria and nuclei that are larger and more vesicular than those of chief cells (Figure 70-2). These cells appear at puberty and increase in number with advancing age. Oncocytic nodules are found commonly in the parathyroid glands of older individuals. Transitional oncocytes are smaller than mature oncocytes and contain fewer mitochondria.
The amount of stromal fat varies with age and nutritional status. Glands from children contain only very small amounts of stromal fat, but by early adulthood, stromal fat cells make up approximately 20% of the volume of the glands.7 Parathyroid glands from obese individuals generally contain abundant fat, whereas the glands from lean persons or those with severe cachexia are composed almost exclusively of parenchymal cells.
Hyperparathyroidism
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Hyperparathyroidism (HPT) is a metabolic disorder that is characterized by the increased synthesis and secretion of parathyroid hormone (PTH). Serum calcium levels may be increased, decreased, or normal, depending on renal function and a variety of other factors.8 Hyperparathyroidism is classified into primary, secondary, and tertiary types. Primary HPT is defined by elevation of serum ionized calcium in the setting of an inappropriate elevation of PTH.9 The incidence of primary HPT has increased dramatically over the past several decades, primarily as a result of the introduction of multiphasic biochemical screening programs in the 1970s.10,11 Before that time, most patients with primary HPT presented with renal or bone disease (osteitis fibrosa cystica). Currently, most patients with primary HPT either are asymptomatic or have neuropsychiatric symptoms, gastrointestinal disorders, or relatively mild and nonspecific complaints of lethargy and weakness. In patients with primary HPT, excess PTH originates from neoplastic or hyperplastic proliferations of parathyroid parenchymal cells. High levels of PTH lead to increased renal resorption of calcium, phosphaturia, increased synthesis of 1,24 (OH)2D3, and increased bone resorption.9 As a result, patients have evidence of hypercalcemia, hypophosphatemia, hypercalciuria, increased levels of PTH, and normal levels of PTH-related protein, although occasional patients may be normocalcemic. Rare cases of apparent primary HPT may result from the paraneoplastic production of PTH by nonparathyroid tumors,12 but most cases of malignancy-associated hypercalcemia result from the paraneoplastic production of PTH-related protein.13 Secondary (S)-HPT is an adaptive increase in PTH production associated with chronic renal failure, vitamin D deficiency, or intestinal malabsorptive states, whereas tertiary (T)-HPT is characterized by the development of autonomous parathyroid hyperfunction in patients with established secondary HPT.
Primary Hyperparathyroidism
Parathyroid Adenoma
Parathyroid adenomas are responsible for 80% to 85% of all cases of primary HPT.14 In some series, however, the incidence of adenomas is considerably lower because of differing patterns of patient referral and differences in histologic criteria to distinguish adenomas from hyperplasias. Adenomas can occur at any age, although most become evident clinically in the fourth decade. They occur more commonly in females, with a female-to-male ratio of 3-4:1.9
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Most parathyroid adenomas occur sporadically, although a subset may be associated with the multiple endocrine neoplasia or other heritable syndromes. X-chromosome inactivation studies have revealed that the majority of adenomas are clonal proliferations.15 The multiple endocrine neoplasia (MEN) 1 gene on chromosome 11q13 is a tumor suppressor gene that encodes menin, and mutations of this gene are responsible for the development of MEN 1. Loss of one MEN 1 allele has been observed in up to 40% of sporadic parathyroid adenomas with an inactivating mutation of the second allele occurring in approximately 50% of these cases.16–18 Approximately 5% of adenomas have an activated form of the PRAD-1/CCND1 oncogene, which is a consequence of the juxtaposition of the regulatory region of the PTH gene on chromosome 11 with the coding region of PRAD-1. This rearrangement results in the overexpression of cyclin D-1 in the tumor cells, leading to stimulation of cell growth. Fluorescence in situ hybridization (FISH) analyses have reported similar translocations in up to 8% of parathyroid adenomas.16–18 However, overexpression of cyclin D1 occurs in up to 40% of adenomas, suggesting mechanisms other than translocations for overexpression, including amplification, rearrangements with other enhancers or promoters, and transcriptional activation.
Most adenomas involve a single gland and occur with equal frequency in the upper and lower parathyroids.14,19 The remaining tumors occur in a variety of other sites, including the thymic tongue, thymus gland proper, retroesophageal space, or within the thyroid gland. Rare examples of adenoma have been recorded in the pericardium, vagus nerve, and soft tissues adjacent to the angle of the jaw. Adenomas rarely may involve two glands, although the distinction from hyperplasia in these cases is often problematic.20,21
Parathyroid adenomas range from less than a few millimeters to many centimeters in diameter. In the series reported by Castleman and Roth, adenomas associated with severe bone disease had an average weight of 10 g, whereas smaller tumors were less symptomatic.19 Most adenomas are ovoid and have a smooth surface, but occasional tumors may be multilobate. Tumors of the latter type are at high risk for recurrence because they may be removed incompletely during initial surgery. On cross section, adenomas are encapsulated and soft in consistency, with a color ranging from tan to orange-brown. Foci of cystic change are common, particularly in large adenomas and in patients with the hyperparathyroidism-jaw tumor (HPT-JT) syndrome. Cystic parathyroid tumors may be associated with prominent pericapsular fibrosis. Adherence of a cystic adenoma to the adjacent thyroid or soft tissues may lead to a suspicion of carcinoma at surgical exploration.
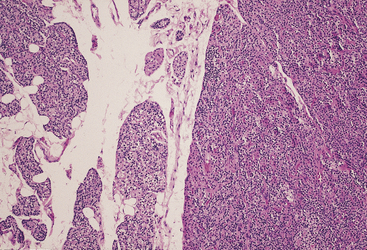
Most adenomas are composed of chief cells arranged in cords and sheetlike arrangements, and foci of glandlike arrangements may be prominent14 (Figures 70-3 and 70-4). Rarely, the tumor cells may have a papillary arrangement.22 The glandlike structures often contain a densely eosinophilic colloid material morphologically similar to that found in the thyroid, and the distinction between parathyroid and thyroid tissue in such instances may be particularly difficult, if not impossible, at frozen section. However, colloid in thyroid tissue frequently contains birefringent oxalate crystals, which are absent from the colloid-like material in parathyroid tumors.23 The colloid in parathyroid often stains with Congo red with green birefringence in polarized light.24 Focal calcification of the luminal colloid may also occur, similar to that seen in oncocytic thyroid neoplasms.
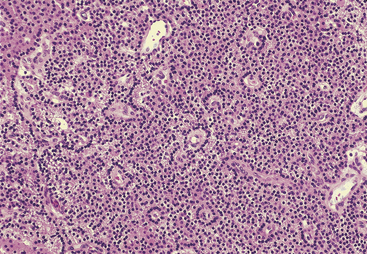
Figure 70-4 Parathyroid adenoma composed of chief cells arranged in palisaded pattern around blood vessels.
Neoplastic chief cells are generally larger than those in the adjacent normal gland.14 The cytoplasm is faintly eosinophilic but may appear clear because of the accumulation of glycogen deposits. In addition to chief cells, adenomas also may contain groups of transitional oncocytes and oncocytes. Adenomas may have a multinodular appearance, with individual nodules being composed of chief cells or oncocytes. The nodules often have a higher proliferative fraction than the non-nodular portions of the adenoma.25
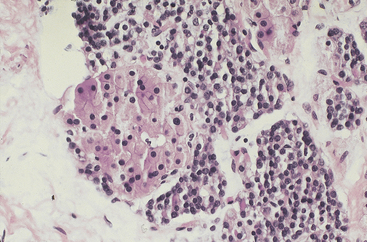
The nuclei of adenoma cells are generally round and central with dense chromatin and occasional small nucleoli (see Figure 70-4). Enlarged hyperchromatic nuclei may be scattered throughout the gland, and occasionally they may form nodular aggregates (Figure 70-5).14 In the absence of other features of malignancy, the presence of such nuclei should not be used as a criterion for the diagnosis of malignancy. Mitotic activity has been considered in the past to be a feature of malignancy.19 However, Snover and Foucar identified mitoses in up to 70% of adenomas.26 Other authors have reported similar findings.27
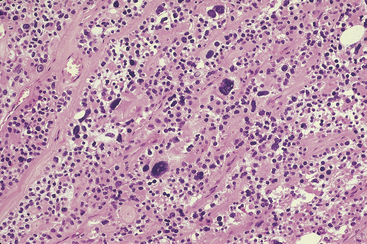
Figure 70-5 Parathyroid chief cell adenoma containing scattered cells with enlarged hyperchromatic nuclei.
A rim of residual normal gland is present in approximately 50% of adenomas (see Figure 70-3). The rim is often separated from the adenomas by a capsule, but in some cases the cells of the rim appear to merge imperceptibly into the adenoma. The probability of finding a rim of normal gland is greater in smaller adenomas than in larger tumors. Typically, the cells within the rim are smaller than the adenoma cells and often contain a single large lipid droplet. If lipid is present within the adenoma cells, it tends to be more finely distributed within the tumor cells. The parathyroid chief cells within the rim generally contain more abundant immunoreactive PTH and messenger RNA than do the adenoma cells.28
Although adenomas have been traditionally described as having no stromal fat, some tumors may contain stromal fat cells distributed diffusely throughout the tumor.14 In some instances, a small biopsy of an adenoma may be misinterpreted as a normal gland because of the abundant fat. Knowledge of the size of the gland would circumvent this potential pitfall. Occasional adenomas may contain abundant fibrous tissue, chronic inflammation, and hemosiderin deposits.
Oncocytic adenomas make up approximately 5% of all parathyroid adenomas.29 At least 90% of the cells within the adenoma should be oncocytes before a lesion is classified as an oncocytic adenoma (Figure 70-6). These tumors are typically orange-tan to dark brown, similar to oncocytomas of other sites. As with chief cell adenomas, the cells may be arranged in cords, nests, tubules, or solid sheets. The cells have abundant granular eosinophilic cytoplasm and vesicular nuclei with a distinct nucleolus. The cytoplasmic granularity corresponds to the presence of numerous mitochondria. Water-clear cell adenomas are exceptionally rare and are characterized by the presence of cells with multiple cytoplasmic vacuoles, similar to those seen in clear cell hyperplasia.30
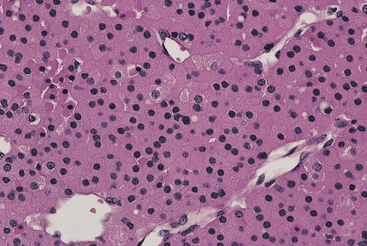
Figure 70-6 Parathyroid oncocytic adenoma composed of large cells with abundant eosinophilic cytoplasm.
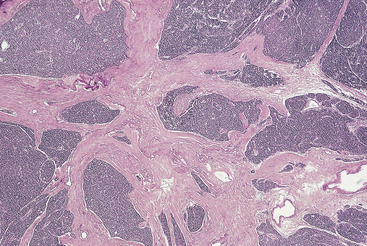
Lipoadenomas (hamartomas) are rare tumors and are characterized by the proliferation of both parenchymal and stromal elements.31 The stromal elements include abundant fibroadipose tissue with varying degrees of myxoid change and variable numbers of lymphocytes (Figure 70-7). The parenchymal components are usually present in the form of compressed cell cords. Microadenomas typically measure less than 0.6 cm in diameter.32,33 They can be so small, in fact, that they may be overlooked on surgical exploration and frozen section. In several reported cases, microadenomas became apparent only after serial sectioning of paraffin-embedded samples. Double adenomas have been reported in less than 2% of patients with primary HPT.20,21 Prior to the development of intraoperative PTH assays, the criteria for the diagnosis of these tumors included the presence of two enlarged glands, each weighing more than 70 mg, and the presence of two normal glands.
Atypical adenomas (equivocal carcinomas) represent a controversial entity. These tumors have some of the features of parathyroid carcinomas but lack unequivocal evidence of invasive growth.34,35 Atypical features include adherence of the tumor to the adjacent soft tissues or thyroid, prominent mitotic activity, trabecular growth pattern, stromal fibrosis, and the presence of tumor cells within the capsule of the gland (Figure 70-8). None of these features, however, is diagnostic of carcinoma. Based on studies reported in the literature and our own observations, most atypical adenomas pursue a benign clinical course.36,37 The molecular phenotype of atypical adenomas appears to be intermediate between that of adenomas and carcinomas. Stojadinovic et al. reported that the phenotype p27(+), bcl-2 (+), Ki-67(−), and mdm(+) was present in 76% of and 29% of typical adenomas and atypical adenomas, respectively, but in no cases of carcinoma.38 However, other workers have failed to confirm these observations.39 Juhlin and coworkers reported a complete loss of adenomatous polyposis coli (APC) protein expression and reduced expression of parafibromin in two of five atypical adenomas.40 In contrast, all adenomas were APC positive, including two cases of HRPT2 mutation-associated adenomas, which were negative for parafibromin. These observations will require further study based on larger case series. From a practical point of view, the diagnosis of atypical adenoma should stimulate regular clinical follow-up with appropriate radiologic studies and measurements of serum calcium and PTH.
Differential Diagnosis
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Adenomas must be distinguished from hyperplasias, and knowledge of the results of ultrasonography, 99mTc-sestamibi scintigraphy, operative findings, and intraoperative PTH (IOPTH) assays is essential for this distinction.41–46 Most adenomas involve a single gland, whereas enlargement of at least two glands is usually apparent on 99mTc-sestamibi scintigraphy in cases of double adenomas and hyperplasias. Multiple studies have confirmed the ability of IOPTH assays to guide surgical decision making by accurately predicting whether or not additional hypersecreting glands remain in the neck after the removal of gland(s) identified by preoperative localization studies.46 The presence of a single enlarged and hypersecreting gland with three normal glands is virtually diagnostic of adenoma. In current practice, the largest gland, as identified by 99mTc-sestamibi scintigraphy, or ultrasonography, is removed and if the level of PTH as determined by IOPTH drops by more than 50% within 10 minutes following excision, the surgical procedure is concluded without having to identify the remaining glands.
In the past, biopsy of a second gland was considered to be essential for accurate classification of the case.14 Most commonly, parathyroid glands associated with adenomas are indistinguishable from those of normal persons, although in many instances the adenoma-associated glands contain cytoplasmic vacuoles within the non-neoplastic chief cells, as discussed later.
In a series of more than 600 cases performed by the traditional four-gland approach or by a minimally invasive procedure, the rates for double adenomas or hyperplasia were 11% and 7%, respectively, for the former approach and 5.1% and 1.6%, respectively, for the latter.46 These findings underscore the fact that a subset of patients had enlarged glands that were not identified by 99mTc-sestamibi scintigraphy, ultrasound, or IOPTH but that would have been discovered with the traditional four-gland approach. The question remains as to whether enlarged glands that are not hypersecreting at initial surgery will cause recurrent hyperparathyroidism in the future. Follow-up studies have demonstrated a cure rate of 98% with the focused approach; however, some studies have questioned the validity of this approach.47
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Parathyroid Carcinoma
Parathyroid carcinomas are rare neoplasms, accounting for less than 1% of all cases of primary HPT in most large series.48,49 Variability in the incidence of this tumor in different parts of the world may represent true geographic differences or variations in the pathologic criteria for the diagnosis of parathyroid carcinoma. Most cases occur in the fourth to sixth decades of life, and in contrast to the female predominance observed for adenomas, there are no sex differences in the incidence of carcinomas.50 Very rarely, these tumors have been reported in the pediatric age group. Most affected patients have serum calcium levels in excess of 14 mg/dL, and metabolic complications are considerably more common than in patients with adenomas. In rare instances, parathyroid carcinoma occurs in the setting of long-standing secondary HPT.51 Parathyroid carcinoma has been reported in up to 15% of patients with the HPT-JT syndrome.52 An increased risk has also been reported in patients with isolated familial HPT,53 but these tumors occur very rarely in association with MEN 1 and 2A.54,55
The tumors are often palpable clinically and appear as ill-defined masses that are densely adherent to the adjacent soft tissues or thyroid gland. Some carcinomas may be completely encapsulated and may be impossible to distinguish from adenoma. In the series of cases reported by Wang and Gaz, the carcinomas had mean diameters and weights of 3 cm and 6.7 g, respectively. Most tumors are firm and gray-white on section (see Chapter 69, Parathyroid Carcinoma).56
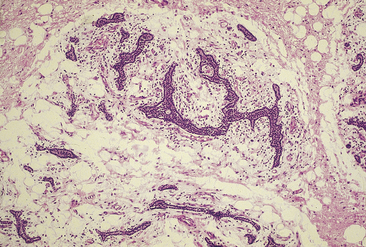
Carcinomas are usually composed of chief cells arranged in solid sheets or trabecular patterns. The component cells may differ minimally from those of adenomas or may show considerable pleomorphism and anaplasia (Figure 70-9). Tumors containing a predominance of clear (vacuolated) chief cells or oncocytes also occur, and carcinomas containing an admixture of the various cell types are not uncommon. As reported by Schantz and Castleman, the principal features for the diagnosis of carcinoma include the presence of thick fibrous bands, mitotic activity, capsular invasion, and vascular invasion57 (Figure 70-10). However, neither mitotic activity nor trabecular growth are specific for malignancy.35 Areas of fibrosis, for example, may be prominent in large adenomas, particularly those with cystic change and in cases of S-HPT.14 Moreover, mitotic activity occurs in a substantial portion of adenomas and hyperplasias, although the presence of abnormal mitoses is characteristic of carcinomas. The presence of collections of neoplastic chief cells in the capsules of parathyroid neoplasms (entrapment) cannot be used as a criterion of malignancy, because this feature occurs commonly in adenomas. However, invasion of the adjacent soft tissues, perineural spaces, or thyroid gland is diagnostic of carcinoma.49
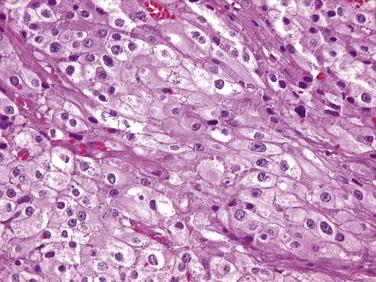
Figure 70-9 Parathyroid carcinoma composed of cells with pleomorphic nuclei containing prominent nucleoli.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree