The surgical management of thyroid disease and its indications continue to evolve. Selecting the appropriate management can often be challenging to both physician and patient. This article describes the surgical management of various thyroid pathologies. Using contemporary guidelines and clinical experience a framework for selecting appropriate surgical treatment is provided.
Surgery of the thyroid gland has evolved in many ways since its modernization by Theodor Kocher in the late nineteenth century. Along with procedural modifications, the surgical indications for benign and malignant disease have also continued to evolve and have often been a source of controversy. Recently, the American Thyroid Association (ATA) and the National Comprehensive Cancer Network have developed task forces aimed at delineating the indications for surgery of both benign and malignant thyroid disease. The algorithms introduced in these guidelines were intended to simplify decisions on the surgical management of controversial issues. The authors at their institution use these guidelines, clinical experience, and informed patient preferences to perform appropriate surgical procedures. This article describes the indications, surgical management, and postoperative care for both benign and malignant processes of the thyroid gland.
Preoperative planning
Before any surgical procedure, a detailed patient history, thyroid function testing, physical examination including laryngoscopy, and appropriate imaging studies should be performed. Patient history including family history, history of prior radiation exposure, and overall health should be solicited. If the patient or family history reveals findings suggestive of multiple endocrine neoplasia (MEN) IIA or IIB, a work-up for pheochromocytoma should be performed preoperatively. To avoid unfavorable outcomes associated with a thyroid storm, preoperative screening thyroid functions tests should be performed. In addition, laryngoscopy should be performed routinely before surgery. Recurrent laryngeal nerve (RLN) compression or invasion may be asymptomatic or slow to progress necessitating visualization to determine vocal fold function. The authors routinely use in-office ultrasound to evaluate lesions for their size and presence of any suspicious sonographic features. Suspicious radiographic findings include microcalcifications, hypoechoic solid nodules, hypervascularity, irregular borders, or a taller rather than wider nodule on transverse imaging. Preoperative awareness of contralateral nodules can also facilitate surgical planning. In cases of cytologically or sonographically suspicious nodules, the extent of disease including lymph node involvement in both the central and lateral neck can be demonstrated with ultrasound. The routine use of preoperative CT scan is not necessary in most cases; however, noncontrast CT scan is beneficial for determining the caudal extent and degree of tracheal compression secondary to large substernal goiters (SSG). Preoperative CT scans may also provide useful insight if there is a concern of laryngotracheal invasion associated with a malignant process. A careful synthesis of the aforementioned tools can help the surgeon optimize the extent of surgery and avoid potential shortcomings in their surgical interventions.
Indications for thyroidectomy
Thyroidectomy can be performed for both benign and malignant disease. The first category includes hyperthyroidism, SSG, and nodular goiter. Hemithyroidectomy may also be of diagnostic use in the case of a suspicious nodule. In this discussion, the malignant category includes well-differentiated thyroid carcinoma (WDTC), medullary thyroid carcinoma (MTC), and anaplastic thyroid carcinoma.
Hyperthyroidism
Hyperthyroidism treatment is aimed at symptomatic relief with control of associated systemic morbidity. Optimal treatment of hyperthyroidism is dependent on the etiology. Hyperthyroidism associated with Hashimoto or de Quervain thyroiditis is self-limited. In general, medical management with the use of β-blockers and antithyroid medications (methimazole or propylthiouracil) achieves good control in most clinical scenarios. Antithyroid treatment may not be ideal long-term therapy in patients with toxic multinodular goiter or toxic adenoma because of the natural history of disease progression. Surgery and radioiodine therapy are definitive management options for toxic nodular disease or Graves disease.
Surgical options for those with hyperthyroidism include hemithyroidectomy for toxic adenoma or total thyroidectomy for toxic multinodular goiter or Graves disease. Patients who may benefit from surgery include those who cannot tolerate or are noncompliant with antithyroid medication, or have absolute or relative contraindications or aversion to radioactive iodine (RAI). Individuals desiring return to normal thyroid function sooner than can be achieved with RAI may also consider thyroidectomy. Shindo recommended that before surgery hyperthyroid patients receive antithyroid medication, propanolol, and potassium iodide especially for patients with Graves disease. The patient should be rendered euthyroid and have a resting heart rate less than 80. Potassium iodide or Lugol solution has the potential benefit of decreasing the thyroid gland vascularity, and minimizing glandular hemorrhage during surgery. This can be administered in 1 to 2 lingual drops 7 to 10 days before surgery. Occasionally, corticosteroids may be necessary in medically refractory Graves disease patients.
SSG
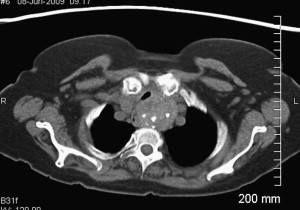
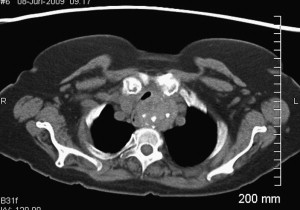
SSG can be challenging for both the patient and surgeon. SSGs generally behave in an indolent manner, following a path of least resistance through the thoracic inlet. Most patients remain asymptomatic until the goiter impedes on adjacent structures of the thoracic cavity. Commonly associated symptoms include supine dyspnea, dysphagia, cough, and hoarseness. Vascular compression of the superior vena cava and vertebral arteries have been reported. The reported malignant potential for SSG is approximately 3% to 21%. Technically, obtaining a fine-needle aspirate for cytologic analysis can be limited by the intrathoracic or retrosternal position of SSGs. If there is not a significant cervical portion, obtaining tissue for diagnosis is contingent on removal of the SSG. Because the single most effective treatment for SSG is surgery, and because of the relative safety associated with this procedure, it is believed that the presence of SSG is an indication for surgery, irrespective of associated clinical manifestations especially in young or middle aged patients. A CT scan can help the surgeon identify its inferior extent and location within the mediastinum (anterior vs posterior), degree of tracheal compression, malignancy, and caudal extent of SSG ( Fig. 1 ).
It is generally accepted that most benign SSGs can be removed through a cervical incision. For the remaining lesions not amenable for removal through a cervical approach, the authors use a ministernotomy, which is performed in conjunction with thoracic surgery. They involve the thoracic surgery service preoperatively for possible sternotomy on patients with posterior mediastinal goiters, those goiters with extension below the level of the aortic arch, and known malignancy in the chest. In addition, Cohen has recommended a sternotomy for the extraction of ectopic goiters. Anatomically, one must recognize the potential anterior displacement of the RLN by a posterior mediastinal goiter. Prior knowledge of this allows the surgeon to anticipate the location of the RLN during surgical dissection. Additional surgical exposure is gained by positioning the patients’ head in neck extension allowing for a cephalad displacement of the SSG. This facilitates improved delivery and visualization of the goiter.
Nodular Goiter
Thyroid nodules are a common finding, and often a source of diagnostic and management dilemmas. The low cost and availability of ultrasound have made detection of thyroid nodules very easy and has led to the 10 times increased detection rates. Coupled with the incidental detection of nodules during imaging for nonthyroid processes clinicians are seeing an epidemic. Although most of the nodules are benign, 5% to 10% of nodules are malignant. Surgeons should familiarize themselves with the sonographic characteristics of thyroid nodule pathology and their appropriate management.
Appropriate treatment of thyroid nodules relies on diagnostic cytology obtained through fine-needle aspiration biopsy (FNAB). The risk of malignancy is the same for patients with a solitary nodule or multiple nodules. Sonographically benign-appearing nodules larger than 1.5 cm or sonographically or clinically suspicious nodules should be considered for biopsy. The results of FNAB combined with ATA published treatment guidelines help direct the management of thyroid nodules. FNAB results are classified as benign (70%); indeterminate (10%, “suspicious for malignancy” or possible follicular neoplasm); malignant (5%); and nondiagnostic (15%).
Ultrasound-guided FNAB greatly improves diagnostic accuracy over palpation with reduction in false-negative and nondiagnostic rates. Management of cytologically benign thyroid nodules includes observation with interval follow-up caused by the FNAB false-negative rates of up to 5%. A significant increase in nodule size greater than 20% to 50% or development of suspicious sonographic features warrants repeat ultrasound-guided FNAB or surgical excision.
Nodules with FNAB demonstrating indeterminate cells (“suspicious,” “follicular,” or Hurthle cell neoplasm) carry approximately a 10% to 20% risk of malignancy. Solid or complex nodules yielding persistent nondiagnostic findings may demonstrate a 5% to 10% risk of malignancy. For these patients with associated risk factors, the ATA suggests thyroid lobectomy as an initial treatment. Following lobectomy, intraoperative frozen histopathologic analysis positive for carcinoma requires a total thyroidectomy and possible central node dissection if gross nodal metastasis is identified. If the pathologist defers diagnosis until permanent sectioning, all patients with malignancy except those with a single focus or subcentimeter disease should undergo completion thyroidectomy. Completion thyroidectomy is usually performed in the first month following the initial procedure. Scenarios that may benefit from a total thyroidectomy rather than lobectomy include (1) individuals with tumors greater than 4 cm and cytologic atypia, (2) FNAB “suspicious for papillary carcinoma,” (3) patients with family history of thyroid carcinoma, and (4) childhood radiation exposure caused by increased risk of malignancy in these clinical settings.
Malignancy
Optimal surgical management of thyroid malignancy is dependent on the type and extent of malignancy: WDTC, papillary thyroid carcinoma (PTC), follicular thyroid carcinoma (FTC), Hurthle cell carcinoma (HCC), MTC, or anaplastic thyroid carcinoma.
Several variations of thyroidectomy exist but total or near total thyroidectomy is indicated for most thyroid malignancies. It is necessary to become familiar with the definitions, to communicate effectively with various members of the team. Partial lobectomy involves removing a portion of a single thyroid lobe and is not indicated in diagnosis or treatment of malignancy. Thyroid lobectomy or hemithyroidectomy involves resection of one thyroid lobe and may be sufficient for an isolated, subcentimeter FTC or PTC. Near total thyroidectomy is the resection of all grossly visible thyroid tissue, leaving a small fragment (1 g) of residual thyroid gland near the insertion of the RLN and ligament of Berry. A subtotal thyroidectomy involves leaving residual thyroid tissue greater than 1 g or with the ipsilateral posterior capsule intact. This is not an oncologically acceptable maneuver in the treatment of thyroid carcinoma. Total thyroidectomy implies complete removal of the thyroid gland and is the procedure of choice for PTC, FTC, HCC, and MTC. Completion thyroidectomy refers to the removal of the contralateral residual thyroid lobe following a prior hemithyroidectomy when the diagnosis is made postoperatively.
PTC represents approximately 80% of thyroid malignancies. It may display multicentricity in the ipsilateral or contralateral lobe in a high proportion of cases. This multicentricity poses the predicament of hemithyroidectomy versus total thyroidectomy as definitive surgical therapy. At the Mayo Clinic, Hay and colleagues compared patients treated with ipsilateral thyroid lobectomy plus isthmusectomy with bilateral resection (total thyroidectomy, bilateral subtotal, or near total thyroidectomy) for PTC. This study analyzed long-term 20-year results of these procedures. The data revealed local recurrence rates of 14% and 2% for thyroid lobectomy and bilateral resection, respectively. Also, rates for nodal metastasis were less with bilateral resection compared with thyroid lobectomy, 6% and 19%, respectively. Although near-total–total thyroidectomy is considered the definitive surgical modality for PTC, there are certain clinical circumstances in which hemithyroidectomy may be performed. The ATA states that hemithyroidectomy may be adequate for less than 10-mm, low-risk, isolated, intrathyroidal papillary carcinomas without evidence of cervical nodal disease.
PTC can manifest with cervical nodal metastasis in 20% to 90% of cases. Some authors have demonstrated the presence of central and lateral neck metastases with rates of 62.2% and 25.6%, respectively. Preoperative ultrasound is a sensitive tool in detecting the presence of cervical nodal disease, but may be more sensitive for the lateral rather than central compartment. The central compartment or level VI is defined by the carotid arteries laterally, hyoid bone superiorly, and suprasternal notch or brachiocephalic vessels inferiorly. Therapeutic central compartment dissection should be considered in patients with PTC and nodal metastasis. For those with lateral nodal metastasis a functional level II to V neck dissection should be included.
FTC can be categorized as either minimally or widely invasive. Minimally invasive is the more common of the two and has no extension through the thyroid capsule with or without vascular invasion. Patients with lesions less than 1 to 1.5 cm may undergo hemithyroidectomy. For tumors described as minimally invasive with vascular invasion and widely invasive FTC near total or total thyroidectomy without central neck dissection is acceptable. Unlike PTC and HCC that spread through lymphatics, FTC primarily spreads hematogenously or by direct extension. This behavior eliminates the need for elective or prophylactic neck dissections.
HCC is considered a variant of follicular carcinoma. This rare tumor comprises a small percentage of thyroid cancers, in the neighborhood of 2% to 3%. They tend to behave aggressively, and can readily demonstrate lymphatic spread. Near-total–total thyroidectomy is the recommended intervention. Like PTC, therapeutic central compartment node dissection should be performed in the presence of nodal metastasis. Evidence of lateral compartment nodal disease requires a functional neck dissection of levels II to V.
MTC is derived from parafollicular C cells of the thyroid gland and is embryologically distinct and differs in its behavior from WDTC. It is unresponsive to RAI, and surgery is the accepted basis of treatment. MTCs are multifocal and metastatic in most patients, and total thyroidectomy with elective or therapeutic central compartment neck dissection is the procedure of choice. This has been shown to achieve 80% biochemical cure rate, with a return to normocalcitonemia. If there is evidence of levels II to V nodal disease, a therapeutic lateral neck dissection is suggested. Patients with MTC require genetic testing for somatic RET mutations associated with MEN IIA and IIB. Al-Rawi and Wheeler suggest that despite total thyroidectomy and selective neck dissection 20% of patients recur or have residual disease. MTC association with MEN IIA and MEN IIB requires all first-degree relatives including children be tested for RET mutations. It is generally accepted that children with MTC and those with MEN IIA should undergo a thyroidectomy by age 6 years. Those with the more aggressive MEN IIB should have surgery within the first year of life. In addition, one can use microdissection effectively in this population.
Indications for thyroidectomy
Thyroidectomy can be performed for both benign and malignant disease. The first category includes hyperthyroidism, SSG, and nodular goiter. Hemithyroidectomy may also be of diagnostic use in the case of a suspicious nodule. In this discussion, the malignant category includes well-differentiated thyroid carcinoma (WDTC), medullary thyroid carcinoma (MTC), and anaplastic thyroid carcinoma.
Hyperthyroidism
Hyperthyroidism treatment is aimed at symptomatic relief with control of associated systemic morbidity. Optimal treatment of hyperthyroidism is dependent on the etiology. Hyperthyroidism associated with Hashimoto or de Quervain thyroiditis is self-limited. In general, medical management with the use of β-blockers and antithyroid medications (methimazole or propylthiouracil) achieves good control in most clinical scenarios. Antithyroid treatment may not be ideal long-term therapy in patients with toxic multinodular goiter or toxic adenoma because of the natural history of disease progression. Surgery and radioiodine therapy are definitive management options for toxic nodular disease or Graves disease.
Surgical options for those with hyperthyroidism include hemithyroidectomy for toxic adenoma or total thyroidectomy for toxic multinodular goiter or Graves disease. Patients who may benefit from surgery include those who cannot tolerate or are noncompliant with antithyroid medication, or have absolute or relative contraindications or aversion to radioactive iodine (RAI). Individuals desiring return to normal thyroid function sooner than can be achieved with RAI may also consider thyroidectomy. Shindo recommended that before surgery hyperthyroid patients receive antithyroid medication, propanolol, and potassium iodide especially for patients with Graves disease. The patient should be rendered euthyroid and have a resting heart rate less than 80. Potassium iodide or Lugol solution has the potential benefit of decreasing the thyroid gland vascularity, and minimizing glandular hemorrhage during surgery. This can be administered in 1 to 2 lingual drops 7 to 10 days before surgery. Occasionally, corticosteroids may be necessary in medically refractory Graves disease patients.
SSG
SSG can be challenging for both the patient and surgeon. SSGs generally behave in an indolent manner, following a path of least resistance through the thoracic inlet. Most patients remain asymptomatic until the goiter impedes on adjacent structures of the thoracic cavity. Commonly associated symptoms include supine dyspnea, dysphagia, cough, and hoarseness. Vascular compression of the superior vena cava and vertebral arteries have been reported. The reported malignant potential for SSG is approximately 3% to 21%. Technically, obtaining a fine-needle aspirate for cytologic analysis can be limited by the intrathoracic or retrosternal position of SSGs. If there is not a significant cervical portion, obtaining tissue for diagnosis is contingent on removal of the SSG. Because the single most effective treatment for SSG is surgery, and because of the relative safety associated with this procedure, it is believed that the presence of SSG is an indication for surgery, irrespective of associated clinical manifestations especially in young or middle aged patients. A CT scan can help the surgeon identify its inferior extent and location within the mediastinum (anterior vs posterior), degree of tracheal compression, malignancy, and caudal extent of SSG ( Fig. 1 ).