Chapter 65 Surgical Management of Multiglandular Parathyroid Disease
![]() This chapter contains additional online-only content, available on expertconsult.com.
This chapter contains additional online-only content, available on expertconsult.com.
In 1934, Albright et al.1 discovered the unusual entity of parathyroid water-clear–cell hyperplasia and thought this resulted from some form of extrinsic glandular stimulation. The more common parathyroid chief-cell hyperplasia was described in 1958 by Cope et al.2 and was believed to have a similar genesis. However, stimulating agents causing the disease have still not been identified in sporadic parathyroid hyperplasia, in contrast to hyperparathyroidism (HPT) secondary to uremia or long-term lithium therapy (see Chapter 66, Surgical Management of Secondary and Tertiary Hyperparathyroidism). Despite the fact that they represent a minority among HPT patients, ever since the early days of parathyroid surgery cases of parathyroid hyperplasia have continued to intrigue pathologists and parathyroid surgeons (see Chapter 70, Surgical Pathology of the Parathyroid Glands). Hyperplasia is easily overlooked when all parathyroid glands have not been properly visualized at operation, and the results of parathyroid surgery have generally been less satisfactory than with single adenomas. In patients with hyperplasia, ectopic and supernumerary glands may frequently be involved in the pathologic process, and expert knowledge of parathyroid anatomy is essential for the management of this patient group (see Chapter 2, Applied Embryology of the Thyroid and Parathyroid Glands).
Because the distinction between adenoma and hyperplasia has often been equivocal, many physicians have preferred to classify the principal histopathologic entities of HPT as uniglandular and multiglandular disease. This is reasonable because individual glands in adenoma and chief-cell hyperplasia often do not differ grossly or histopathologically and may indeed both represent monoclonal lesions (see Chapters 58, Principles in Surgical Management of Primary Hyperparathyroidism, and 70, Surgical Pathology of the Parathyroid Glands). Disclosure of the genetics behind parathyroid hyperplasia and neoplasia seems increasingly important for discussions of pathogenesis and may in the future possibly provide better criteria for discrimination between the histologic entities. Different genetic abnormalities may cause uniglandular or multiglandular disease or be associated with variable rates of disease progression of either. Genetic analyses can thus be expected to become increasingly important and in the future provide an important basis for decisions about management. A significant proportion of patients with multiglandular parathyroid disease have hereditary disorders requiring special investigations and treatment. They may have associated endocrinopathies as part of multiple endocrine neoplasia (MEN) syndrome, the most common form of which is MEN 1 syndrome, or they may merely have familial forms of HPT (see Chapter 67, Parathyroid Management in the MEN Syndromes). Recently an increasing number of patients with primary HPT have been subjects of unilateral, minimally invasive, or endoscopic parathyroidectomy (see Chapters 59, Standard Bilateral Parathyroid Exploration, and 60, Minimally Invasive Single Gland Parathyroid Exploration). The long-term results of this surgery are presently unknown, but there appears to be general agreement that patients with multiglandular parathyroid disease should undergo complete bilateral neck exploration. The initial neck exploration is the best opportunity to successfully treat a patient with HPT, because repeat parathyroid surgery is generally more difficult and is associated with a lower success rate and higher complication risk (see Chapter 68, Reoperation for Sporadic Primary Hyperparathyroidism).
Histopathology and Surgical Anatomy
Few surgical diseases have such variable anatomy and differing histopathologic features as HPT. A thorough knowledge of parathyroid embryology and a familiarity with the normal anatomy and common location of pathologic glands are essential (see Chapter 2, Applied Embryology of the Thyroid and Parathyroid Glands).
Gross Appearance and Histopathology
Normal adult parathyroid glands have a characteristic light yellowish-brownish color and soft consistency, but a fairly distinct capsule that allows the gland to be separated easily from surrounding fat (see Chapter 70, Surgical Pathology of the Parathyroid Glands).3 Most glands are oval or bean shaped, but they may also be round, elongated, bilobated, or multilobated. Diseased glands are most often round or pear- or kidney-shaped, but may also be bilobated or multilobated, in which case part of a diseased gland may easily be left behind (Figure 65-1). A normal gland usually measures around 5×4×2 mm, and its maximal normal weight is around 60 mg. In infants, the normal parathyroids are gray and transparent; in adolescents, they tend to be more brown-reddish than in adults because of a lower content of fat cells. In histologic sections, the normal adult parathyroids consist of parenchymal cells, mainly chief cells with dispersed fat cells in between them. The content of fat cells increases with age and may be very high in obese persons.3 The normal chief cells characteristically contain numerous cytoplasmic fat droplets, which may be visualized with special staining techniques like the Oil-red O staining.4,5 The cytoplasmic fat droplets are generally absent or markedly reduced in diseased parathyroid glands. Oxyphil parathyroid cells are sparse in young and middle-aged individuals but occur more frequently in older persons or in patients with renal function impairment (see Chapter 66, Surgical Management of Secondary and Tertiary Hyperparathyroidism). Parathyroid adenomas and glands with chief-cell hyperplasia are generally indistinguishable at gross examination. The pathologic glands invariably have increased in size because of tumorous growth of the parenchymal cell component, with a generally concomitant disappearance of fat cells. The increased parenchymal content has generally caused a more reddish-brown color and a distinct consistency that still, however, remains soft and homogenous. The normal and diseased parathyroids differ from lymph nodes mainly by the more yellow-brown or brown-reddish color, their softer consistency, and their more distinct separation from surrounding fat. The thyroid is invariably harder and darker reddish and has more heterogenous consistency, especially on the cut surface.
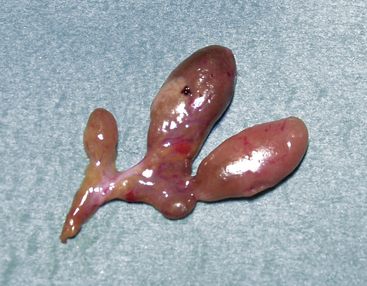
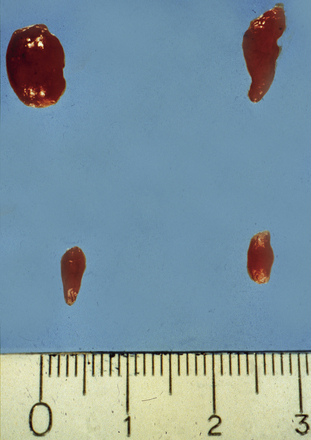
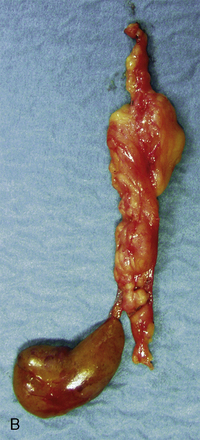
Figure 65-1 Multilobated diseased parathyroid gland. Part of this gland may easily be missed during parathyroid surgery.
Adenomas may exhibit a rim of normal, fat-cell–rich parathyroid tissue from which the adenoma has originated and may occasionally display pronounced nuclear pleomorphism.3,4 Although these features suggest adenomatous disease, similar features may be seen in hyperplastic glands. Some adenomas exhibit distinct nodules of parenchymal cells, indicating their origin from different clones of growing parathyroid cells, whereas other adenomas seem to consist merely of one homogenous cell population. In chief-cell hyperplasia, discrepancy in size between individual glands is generally marked and enlarged obviously abnormal glands will commonly be found to coexist with smaller, apparently normal ones (Figures 65-2 and 65-3). Nodularity and variable growth pattern are generally more obvious in the enlarged hyperplastic glands, where frequently one or several nodules appear to have acquired more pronounced growth advantage or may even occupy the entire gland.5 Cases with modest hypercalcemia commonly exhibit less pronounced glandular enlargement and less variability between glands (see Figure 65-3) but typically still show signs of micronodular hyperplasia in the smaller glands.6 Double adenomas have been reported to occur in 2% to 5% of patients with primary HPT.3,7,8 Some authors have described higher figures and even triple adenomas, but many of these cases are likely to represent only the pronounced asymmetrical glandular involvement of chief-cell hyperplasia. When such glands have occurred asynchronously, causing recurrent HPT in a patient previously operated on with excision of a presumed solitary adenoma, the presence of the MEN 1 syndrome has been common in our experience. The glands of rare water-clear–cell hyperplasia have a characteristic dark to chocolate brown color and are usually markedly and asymmetrically enlarged, with indentations and pseudopod-like extensions.3,4 The water-clear cells are large and polygonal, with cytoplasm that looks empty at low magnification (see Chapter 70, Surgical Pathology of the Parathyroid Glands).
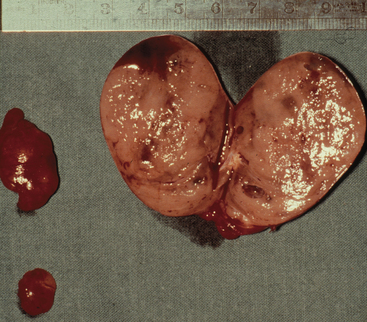
Figure 65-2 Asymmetrical glandular enlargement in a patient with MEN 1–associated HPT (ruler in centimeters).
Secondary parathyroid hyperplasia occurs as an inevitable result of chronic renal insufficiency (see Chapter 66, Surgical Management of Secondary and Tertiary Hyperparathyroidism). The signs of parathyroid stimulation appear early with the renal impairment, and at this stage the parathyroids increase uniformly in size with diffuse hyperplasia. In more severe and prolonged renal insufficiency, the glands have generally attained markedly abnormal morphologic features, with more considerably increased weight and pronounced nodularity comprising both chief and oxyphil cells.3,4 There may also be fibrosis, cysts, and calcifications. Chief cells generally predominate in the secondary hyperplastic glands, but oxyphil and transitional oxyphil cells are often numerous, and their number tends to increase with the severity of renal impairment. During further progression of the parathyroid disease, one or more nodules, with apparently more pronounced growth potential, often become predominant. The term tertiary hyperparathyroidism has been used to define secondary hyperparathyroidism with hypercalcemia persistent after renal transplantation because of the presence of large parathyroid nodules with adenoma-like features.3,4
Number of Parathyroid Glands
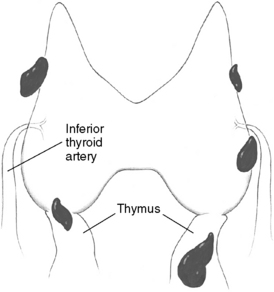
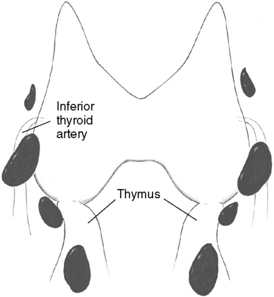
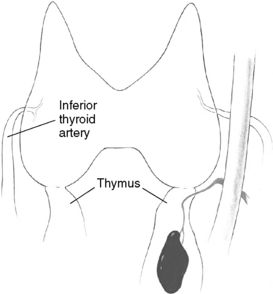
Patients undergoing parathyroid surgery should always be considered to have at least four parathyroid glands.3,9 Supernumerary glands have been demonstrated in nearly 15% of normal individuals and are more likely to be of clinical significance in patients with hyperplasia, especially those with hereditary disease or in HPT secondary to uremia.3,9–12 Proper supernumerary glands occur generally as a fifth gland, most often located in the thymic tongue (Figure 65-4), or occasionally in the vicinity of the thyroid between two other glands (Figure 65-5). Minute pieces of parathyroid tissue or scattered nests of parathyroid cells occur fairly commonly in the thymus and sometimes in fat lobules surrounding a normal parathyroid.9 Such additional glands may be enlarged in hyperplasia and cause synchronous or asynchronously appearing supernumerary glands. Thus, 15% to 40% of uremic patients subjected to long-term dialysis, and thereby continuous parathyroid gland stimulation, have had one or more supernumerary glands retrieved at surgery.12–14 As many as 11 or 12 glands have been encountered occasionally, and when such additional glands have been enlarged they may have appeared as a “string of pearls” along the line of embryologic descent of the parathyroids (Figure 65-6).9
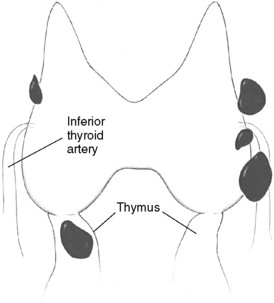
Figure 65-5 Supernumerary parathyroid gland located in the vicinity of the thyroid, between two other glands.
Violation of the capsule of a diseased parathyroid gland during surgery can easily result in implantation of parathyroid cells, which may grow as free grafts into tiny parathyroid nodules and cause recurrent HPT (see Chapter 68, Reoperation for Sporadic Primary Hyperparathyroidism).3,15,16 A condition in which clusters of parathyroid cells have been found diffusely scattered within the adipose tissue of the neck or in the mediastinum has been denoted parathyromatosis; it has generally resulted from implantation of parathyroid cells during operation.15–18 Apparently, similar parathyroid cell nests may occur in the mediastinal thymus in the absence of previous parathyroid surgery.3,17
Vascular Supply
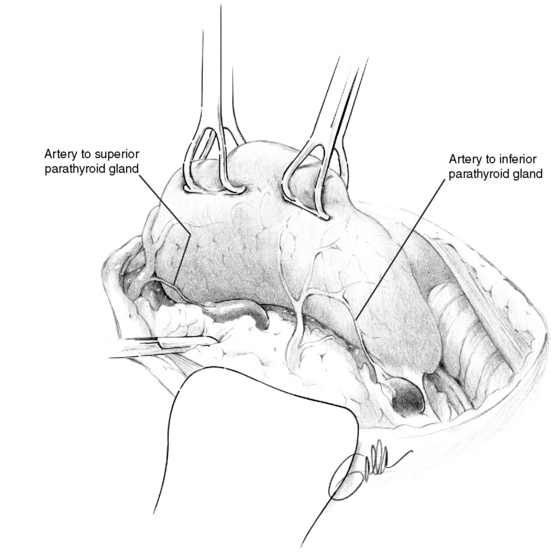
The parathyroid glands receive their principal arterial blood supply from end arteries, generally from the inferior thyroid artery, which, together with the venous effluent, enter the gland via a more or less distinct hilus.3,19 In the diseased gland, a normal glandular remnant may be visible at the hilum. The superior glands are thus most often supplied by arterial branches from the inferior thyroid artery, but in 40% the feeding artery originates from a more or less prominent anastomosing collateral between the superior and inferior arteries, which runs along the posterior margin of the superior thyroid lobe.19 Because the blood supply to the superior parathyroid may depend crucially on this posterior collateral, the posterior branch of the superior thyroid artery should preferably be preserved during parathyroid and thyroid surgery. The artery to a diseased superior gland in the upper posterior mediastinum can be of surprising length and width and may sometimes be followed to deliver the tumor (Figure 65-7). Arterial branches from the inferior thyroid artery almost invariably supply the inferior glands. Also the artery to a diseased inferior gland located below the lower thyroid lobe may be conspicuously large. The artery to a diseased gland in the upper mediastinum may occasionally arise close to the origin of the inferior thyroid artery. This may appear as an additional artery originating from behind the carotid sheath, caudal to the main inferior thyroid artery trunk (Figure 65-8). The supply to an inferior gland in the cervical thymus may come from thymic arteries, whereas the artery to a gland in the anterior mediastinum generally originates from the internal mammary artery.
Normal Parathyroid Anatomy
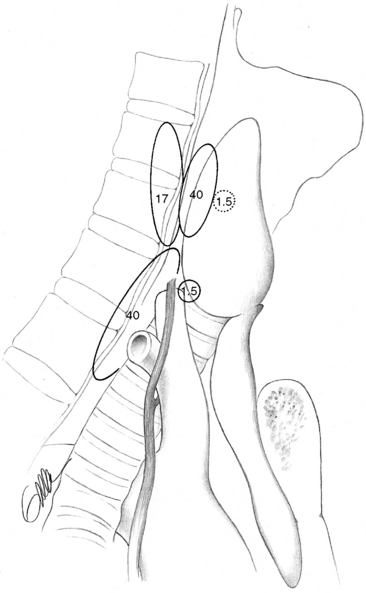
The superior parathyroid glands are most commonly found adjacent to the posterior thyroid capsule, approximately 1 cm above the intersection between the recurrent laryngeal nerve and the inferior thyroid artery, generally at a level corresponding to the cricoid cartilage (Figure 65-9, A).3,9 The glands may be situated at the posterior edge of the upper thyroid lobe or on its ventrolateral surface, more occasionally medially, hidden behind the thyroid in this area. Rarely are superior glands located at the top or above the superior thyroid pole. They can also lie more caudally, along the posterior or lateral thyroid surfaces, where branches of the inferior thyroid artery or the recurrent laryngeal nerve or the nerve-artery intersection may obscure them. A gland can also commonly be disguised behind the common dorsal protrusion of the thyroid in this area, the thyroid tubercle of Zuckerkandl. When superior glands occupy an ectopic location, they have a tendency to lie more posterior in the neck, in the para- or retroesophageal area. The more caudal they are in location, the more posterior their position generally is.
The inferior parathyroid glands are most often situated around the lower thyroid pole, at its caudal lateroposterior or ventral surfaces, often within the thyrothymic ligament or the upper parts of the cervical thymus (Figure 65-9, B).3,9 They can also be found somewhat more cranially, lateral to the thyroid, up to the recurrent laryngeal nerve–inferior thyroid artery intersection, but occasionally even above this point. Inferior glands located on the lateroventral aspects of the lower thyroid lobe may be engulfed within the outer layers of the surgical thyroid capsule, but generally they remain outside the thyroid and the inner layers of this capsule. Such glands are often flattened out but may nevertheless generally be dissected with retained vascular supply. On rare occasions, inferior parathyroids are truly intrathyroidal, deeply hidden in the middle or lower parts of the thyroid.20 Occasionally, inferior parathyroids have descended farther, together with the thymus, to occupy a position in the mediastinal thymus; rarely are they found in the parathymic tissue of the upper mediastinum around the large vessels or attached to the pleura or the pericardium (see Chapter 2, Applied Embryology of the Thyroid and Parathyroid Glands). Incomplete descent may leave an inferior gland high up in the neck, at the level of the angle of the mandible or the hyoid bone, often surrounded by undescended thymic tissue and parathymic fat. This aberrant glandular location, named the parathymus,21 should be suspected, especially if no thymic tissue is found below the thyroid lobe. The glands have most often been located medial to the carotid artery and the jugular vein. Aberrantly descended glands have sometimes also been included within the carotid sheath, generally at the level of the carotid artery bifurcation, but occasionally also lower in the neck.3,22–24
Glandular Symmetry
Although the location of individual glands can vary considerably, there is often marked symmetry of the four parathyroid glands, and if this symmetry is considered, parathyroid operation can often be made easier. The superior glands are thus symmetrically located on contralateral sides of the neck in 80% of cases, whereas such symmetry may be noted for inferior glands in 70%.3,9 In most cases, the superior glands are situated above and the inferior glands below the recurrent laryngeal nerve–inferior thyroid artery intersection. Less commonly, the pairs of superior and inferior glands on one or both sides are located either below or above the nerve-artery intersection, and it may then be difficult to judge which glands constitute the true superior or inferior ones. The most common asymmetrical positioning is one in which only one of the inferior glands is situated lower down in the cervical thymus. Another asymmetrical positioning is when both glands on only one side are located above or below the nerve-artery intersection.9
Technique of Parathyroid Exploration
Positioning of the Patient and Skin Incision
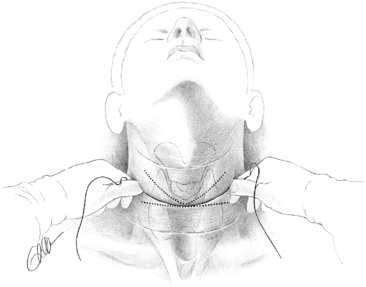
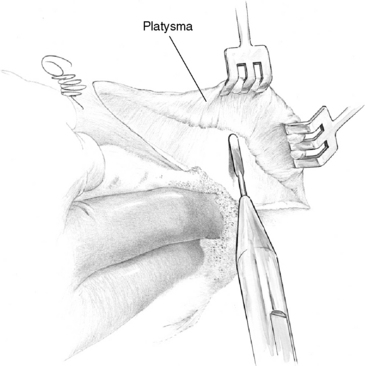
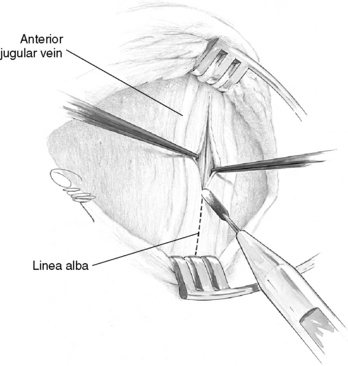
Optimal exposure of the operative field in parathyroid operations is obtained with pillow support under the shoulders and with neck extension, which should lift the thyroid up from the mediastinum. The neck is opened by a collar skin incision that, if placed approximately two fingerbreadths above the sternal notch, will leave a scar in the jugulum when the neck is not extended. For easy access with smaller incision the skin incision may be placed slightly higher. To achieve symmetry and possibly conceal the scar in a skin crease, the surgeon preferably marks the site of the incision before surgery, with the patient in a sitting position and the head kept straight (Figure 65-10). A generous platysma-skin flap is dissected, generally by electrocautery (Figure 65-11), after which the strap muscles are separated in the midline from the sternal notch to the upper part of the thyroid cartilage (Figure 65-12) and retracted laterally. If better exposure is required, as in patients with concomitant goiter, the strap muscles may without apparent postoperative inconvenience be divided or the thyroid cartilage insertion of the sternothyroid muscle may simply be released in its tendinous part. Generally, however, the operation is efficiently performed with the strap muscles intact.
Fascial Layers
Neck exploration is markedly facilitated if the appropriate cleavage planes between the different fascial layers of the neck are utilized during the exposure.3,23 This generally results in minimal distortion of the anatomy and also minimizes the risk to cause bleeding, which will easily obscure the anatomic details and the characteristic consistency and color of the normal and diseased parathyroid glands. The thyroid is covered by the superficial cervical fascia, but under that is also enveloped in the pretracheal fascia. The pretracheal fascia engulfs the superior thyroid lobe in two layers, which join laterally to form the vascular sheath. More caudally, the dorsal fascia covers a dorsal compartment containing the recurrent laryngeal nerve. The pretracheal fascia is distinct around the superior thyroid lobe, but in adults is generally more friable at the middle and lower parts of the thyroid.3 Early in the exploration, the superficial cervical fascia layers are separated from the vascular sheath for mobilization of the thyroid, and transection of this fascia is performed close to the dorsal aspects of the upper thyroid lobe. On the right side, care is taken to avoid injury to a nonrecurrent laryngeal nerve. During this exposure, the middle thyroid vein is divided and ligated early to allow dissection in the “cotton-wool” plane lateral to the thyroid. Traction is applied for adequate rotation of the thyroid lobe and appropriate visualization of its posterior aspects (Figure 65-13). In the lower neck, the fascia is transected more laterally to ensure that the central neck structures, including the thymus, the inferior parathyroid, and the recurrent laryngeal nerve, can be pushed safely toward the midline by blunt dissection. When the thyroid lobe has been mobilized efficiently and safely by this maneuver, the ventral layer of the pretracheal fascia generally still has to be detached from the surgical thyroid capsule along the superior-posterior thyroid border to expose the superior parathyroid gland. Thereafter, the more friable part of this fascia covering the inferior thyroid pole and the cervical thymus is transected, and dissection of the thyreothymic ligament and the cervical thymus can be undertaken. If the recurrent laryngeal nerve needs to be exposed, a thin dorsal layer of the pretracheal fascia generally has to be cut longitudinally along the direction of the nerve (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve). In children, this dorsal layer of the pretracheal fascia, covering the recurrent laryngeal nerve, is often very distinct.
Exposure of the Parathyroids
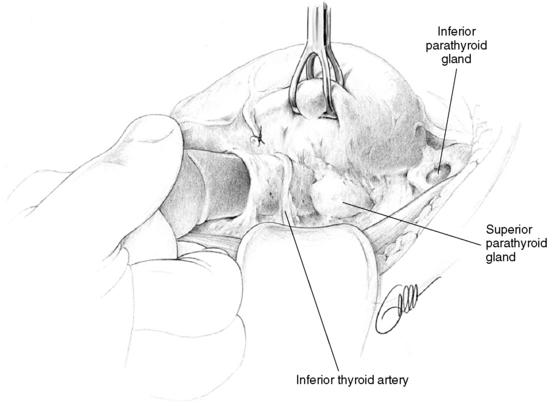
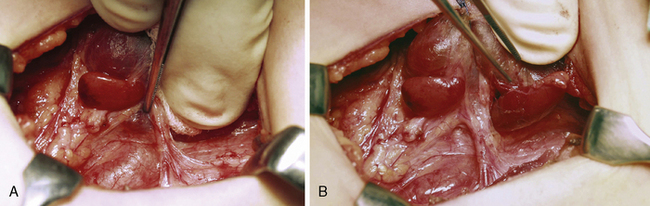
On the side that is explored first, the thyroid lobe is mobilized and the fascial layers are divided as described earlier, closely adjacent to the thyroid capsule, to initially expose the most common locations of the superior and inferior parathyroid glands, respectively (see Figure 65-9). The pertinent site to find a superior parathyroid is the lateroposterior aspect of the superior thyroid lobe, just above the nerve-artery intersection, whereas inferior glands are most often found adjacent to the thyroid insertion of the thyreothymic ligament (see Figure 65-9). The deeper fascial layers of the recurrent laryngeal nerve generally need not be explored, especially not at this stage, because too much and possibly incautious dissection will easily obscure the characteristic features and color of the parathyroids. Interest during this initial phase of the operation is focused just as much on exposure of possible normal-sized glands. It is crucial to visualize these glands before selecting an appropriate strategy of parathyroidectomy in the presence of multiple gland involvement. The principle has been expressed as “not removing anything before seeing everything.”25 The normal-sized glands are generally also the most difficult to visualize and need to be detected early, before even minimal bleeding or tissue edema has disturbed the vision. The surgeon should initially carefully inspect the dissected areas to recognize the typical brownish color of the normal parathyroids or the more reddish color and somewhat firmer but still soft and homogenous consistency and size discrepancy of diseased glands (Figures 65-14, A and B, and 65-13).
If not already identified by this early inspection, the superior parathyroid gland, whose position is the most consistent, is first searched for more thoroughly at the posterior edge of the upper thyroid pole. If the thyroid capsule has been exposed appropriately, a normal parathyroid may be seen or indeed sometimes palpated with a pincette, because it is often “floating” (i.e., appears movable in between the two layers of the pretracheal fascia). Transection of the attachments of the outer layer of this fascia frequently makes the gland pop out or extrude from a lump of fat in this area (see Figure 65-13). If the superior gland has still not been identified, the dissection continues around the ventral and lateroposterior thyroid surfaces, where the gland may be concealed in a cleft of the thyroid or obscured by a prominent thyroid tubercle. By medially rotating the upper thyroid lobe, if necessary with holding sutures or tissue graspers, the surgeon can bring its posterior aspects into better view. Further dissection may require division of the most distal branches of the inferior thyroid artery so that the recurrent laryngeal nerve and the nerve-artery intersection can be completely visualized. The superior gland may be deeply hidden in this area, under the nerve or the nerve-artery intersection, or sometimes even disguised within the ligament of Berry.
If the superior parathyroid gland has not been found after this exploration, the search will also have to be directed toward the common ectopic positions, the tracheoesophageal groove where 2% to 4% of superior glands can be even below the thyroid artery, inferior or a position lateral to or behind the esophagus in the middle or lower neck, or the superior, posterior mediastinum (see Figure 65-7). Division of the posterior sheath of the pretracheal fascia from the level of the inferior thyroid artery to the upper thyroid pole may efficiently expose these glands, after which a finger can help explore the sites lateral to and behind the esophagus, directly on the vertebral fascia, down toward the superior mediastinum (see Figures 65-7 and 65-14, A and B). Only occasionally is the superior parathyroid intrathyroidal, and hemithyroidectomy is therefore rarely indicated when such a gland is missing. If the superior gland has been difficult to find after an extended search, the superior thyroid artery may occasionally be divided for better exposure of the dorsal aspects of the upper thyroid lobe.
The search for the inferior parathyroid gland begins around the inferior, low lateral, and ventral aspects of the lower thyroid pole and includes the thyreothymic ligament and upper parts of the thymus (Figures 65-15, A and B, and 65-9). The cervical thymus is dissected laterally from surrounding fat but kept intact to allow inspection on all sides, before its fascia is cut open over any suspicious lesion. If an inferior gland is not readily identified, the dissection will have to proceed cranially along the lateral and posterior border of the thyroid, between the branches of the inferior thyroid artery, even above the nerve-artery intersection. The surface of the thyroid is also carefully inspected, because an inferior parathyroid may frequently be located within the surgical thyroid capsule at the ventrolateral surface of the lower thyroid pole. The thyroid is also palpated, and any nodularity on its surface is dissected and possibly excised. If the thyroid is completely normal, it is unlikely to contain a diseased parathyroid gland, but if it is enlarged and nodular, an incision or partial resection of the lower thyroid pole may be indicated.20 These measures are preferred to hemithyroidectomy, which may be a serious disadvantage if reoperative surgery should later become necessary. An alternative and even more attractive approach is to utilize intraoperative ultrasound to visualize an enlarged intrathyroidal inferior parathyroid gland. Finally, with the inferior gland still missing, as much as possible of the thymus is pulled up from the anterior mediastinum by blunt dissection and removed (see Figure 65-15, A and B).
Ectopic Glands in Patients with HPT
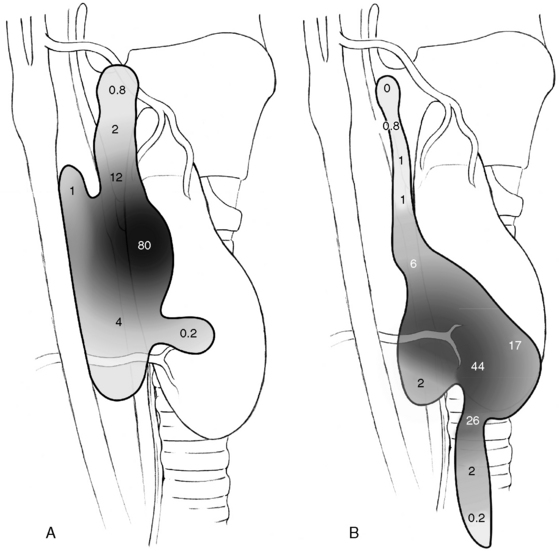
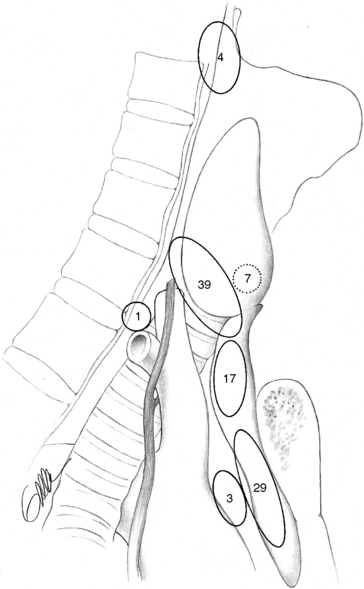
Patients undergoing surgery for HPT have been reported to have more frequent distribution of glands in ectopic positions than is known from autopsy studies of normal parathyroid gland anatomy. This may depend on selection bias associated with patient referral, but it also depends also on different opinions about what really constitutes an ectopic location. It has been suggested that especially enlarged superior glands may more frequently achieve an aberrant location. Thompson et al. found 30% to 40% of superior gland adenomas displaced to a paraesophageal, retropharyngeal, or retroesophageal position, which contrasts to the 1% to 4% incidence of normal superior parathyroids in this location.3,22–24 The same authors have suggested that a superior parathyroid gland adenoma, because of the enlargement, could be pushed by swallowing and migrate in a plane between the pretracheal sheath and the prevertebral fascia to a common position beside or behind the esophagus, caudal and posterior to the inferior thyroid artery. From here they believe it could descend more caudally into the posterior mediastinum, or medially into the tracheoesophageal groove, where it, in contrast to inferior glands in this location, always should be posterior or lateral to the recurrent laryngeal nerve (see Figures 65-7 and 65-9, A and B). This apparent migration may possibly be particularly valid for the more markedly enlarged superior glands (see Figures 65-7 and 65-9, B). Nearly 20% of enlarged superior glands have been retrieved at reoperation from the tracheoesophageal groove or the retropharyngeal or retroesophageal space and 30% to 40% from the superior posterior mediastinum3,22–24,26–32 (Figure 65-16). Unusual superior glands are located higher up around the superior thyroid pole or on the medial surface of the thyroid lobe. Uncommon superior glands have occurred higher, in a parapharyngeal position up to 2 cm above the thyroid pole, sometimes covered by pharyngeal muscles.24 These glands are much rarer than undescended inferior glands at this level, which have more lateral location within the carotid sheath. The superior parathyroid glands are rarely intrathyroidal; more commonly they may be hidden within a groove of the thyroid, especially if the patient has nodular goiter, sometimes reported to occur in 30% to 40% of parathyroid operations.
Inferior parathyroid gland adenomas seem to lack a similar tendency for displacement with enlargement, but they are still more variably distributed and often more difficult to find than the superior adenomas. Like normal inferior glands, the enlarged ones with this origin are most commonly found around the inferior thyroid pole, in the thyreothymic ligament, or within the cervical thymus. Fewer than 2% of inferior diseased glands appear in the superior mediastinum, intimately related to the aortic arch or other large vessels in this area, out of reach from the cervical incision. Rare glands occur deeper in the middle mediastinum or at the aortopulmonary window, although this was a rather common mediastinal location in Tominaga’s extraordinary series of more than 1000 patients with secondary HPT.12 The incidence of intrathyroidal inferior glands has varied, possibly because of the difficulty of distinguishing glands that are entrapped within the surgical thyroid capsule or engulfed within nodular thyroid tissue. True intrathyroidal glands occur in our experience in no more than 1% or less of the patients with HPT, but they have been reported as common in reoperative series.35 Undescended inferior glands are uncommon (< 1%) and can occur above the thyroid often together with thymic tissue, in which case thymus may typically be lacking below the thyroid. Inferior glands may also be aberrantly descended and occur in the vascular sheath often together with thymic tissue. A cord of fibrous thymic tissue can be seen as a track (“yellow-brick road”) to the tumor, crossing laterally from the thyreothymic ligament and sometimes pointing to the tumor.24 The vascular sheath tumors can occur from the level of the clavicle to the carotid bifurcation. Rare inferior glands have been buried in the esophageal wall or located in the lateral triangle of the neck. Glands with location inside the pericardium, within the vagal nerve, or in the mucosa of the pyriform sinus have been occasionally described but are rare.3,22–24 In reoperative series, 30% of inferior diseased glands have been found deep in the mediastinal thymus, 3% have been located outside the thymus around the great vessels in the mediastinum, and 4% have been undescended (Figure 65-16).3,26–35 Occasionally, both superior and inferior glands have been located in the carotid sheath.
Palpation
A diseased parathyroid located behind or within the thyroid or in a retroesophageal position can sometimes be identified by palpation. The sensation that may help identify the gland is that of a circumscribed, sometimes movable tumor with typical soft consistency in contrast to the harder and nodular thyroid (see Figure 65-7).
Glandular Excision
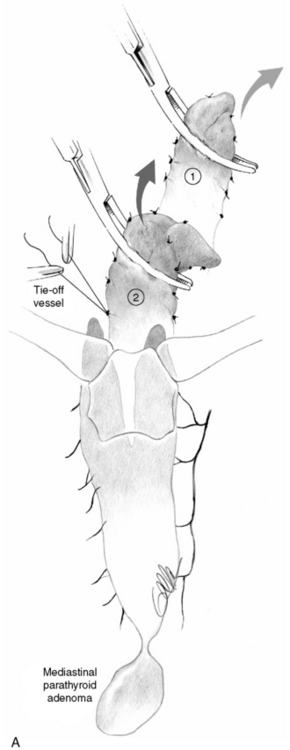
Dissection of a diseased parathyroid gland should appreciate its vascular supply. Delivery of a diseased superior parathyroid is achieved by dissection close to the surface of the gland, beginning at its ventral and dorsal parts. The parathyroid artery and vein, with origin at the posterior aspect of the upper thyroid lobe, remains to be transected last (see Figure 65-13).36 Dissection of a pathologic inferior parathyroid begins at its caudal end, because the vascular hilus generally is located cranially (see Figure 65-13). The possibility of bilobation or multilobation of the gland should be emphasized so that no part of the gland is left behind (see Figure 65-1). The surgeon takes care to avoid rupture of the tumor capsule by holding the gland only in its vascular stalk or in connective tissue around it, whereas layers of surrounding tissue are cut or teased off. If a normal-sized parathyroid is devitalized, it may be cut into small fragments and reimplanted; in cases with multiglandular disease generally in the brachioradial muscle of the forearm.36 The recurrent laryngeal nerve is not routinely exposed during parathyroid surgery except when dissection requires identification or is extensive or at reoperation. One should emphasize that the nerve may be closely associated with the anterior or medial parts of an enlarged upper parathyroid or the dorsal parts of an inferior diseased gland.36
Thymectomy and Central Compartment Dissection
The incidence of clinically significant supernumerary glands is increased in patients with multiglandular involvement, especially in patients with MEN 1–associated HPT or HPT secondary to uremia. It is therefore necessary to explore carefully possible sites of supernumerary glands when one is operating on patients in whom parathyroid tissue has been either genetically triggered or stimulated through renal insufficiency. Cervical thymectomy and excision of fat in the central neck compartment, especially fat pads around the proper parathyroids, is therefore often a part of the operative procedure.9,12 This is now a recommended routine in patients with MEN 1 syndrome or patients with HPT secondary to uremia (see Chapters 66, Surgical Management of Secondary and Tertiary Hyperparathyroidism, and 67, Parathyroid Management in the MEN Syndromes).
The surgeon performs the thymectomy by dissecting close to the thymic capsule, exploring first the cervical portion of the gland (see Figure 65-15, A and B). Traction is then applied with the aid of two surgical forceps grasping the tissue further and further down, and with mainly blunt dissection under the sternum and careful ligation of veins draining into the innominate vein, as much as possible of the thymus is delivered, until it tends to rupture, with a featherlike ending. Clearance of fat in the central part of the neck should be done cautiously, to avoid jeopardizing the circulation of normal parathyroids.
Glandular Biopsies
It has often been recommended that the identity of normal parathyroid glands should be verified by glandular biopsy or that normal parathyroids should be excised to secure the histopathologic diagnosis. However, parathyroid gland biopsies can easily result in devascularization of the glands or tissue implantation, and therefore routine biopsy should be avoided as much as possible.37 The identity of a normal gland is usually evident from size, consistency, and color. If biopsy is needed to determine whether a recognized lesion really is a parathyroid, it should be taken with great caution. Excision of normal-sized glands is sometimes used to support the histopathologic diagnosis but should be avoided as much as possible, because these glands are the ones that should be selected as remnant tissue.
Subtotal versus Total Parathyroidectomy
In 1958, when first describing parathyroid chief-cell hyperplasia, Cope et al. suggested that this entity should be treated by subtotal parathyroid resection.2 Subsequently, subtotal parathyroidectomy was recommended for treatment of patients with hyperplasia secondary to renal insufficiency.38 Because of higher recurrence rates in patients with secondary HPT, it was later suggested that these patients should be subjected to total parathyroidectomy without autotransplantation.35 However, this strategy was soon abandoned, because the aparathyroid condition was claimed to result in adynamic bone, low turnover bone disease with occasionally increased bone pain, and increased risk of hypercalcemia. It was later demonstrated that parathyroid glands after resection retained their function when autotransplanted to the sternocleidomastoid muscle in the treatment of patients with thyroid carcinoma, and total parathyroidectomy with autotransplantation was recommended in patients with primary parathyroid hyperplasia.39 Wells et al. subsequently refined the autotransplantation technique and proposed that the forearm muscle be used as the transplantation site.38,39 By sampling blood for parathyroid hormone (PTH) determination proximal to the transplantation site, one could then document whether the transplanted tissue was functioning.40 Others have suggested that the abdominal or presternal subcutaneous fat should provide a more natural implantation site for the parathyroid,41,42 but most surgeons would still use the forearm musculature because of easier hormone sampling for verification of graft function. It has also been demonstrated that cryopreserved parathyroid tissue could be used for transplantation in patients with deficient parathyroid function after parathyroid operation.43–45 However, the cryopreserved tissue seems to function in only about 60% of patients, in contrast to the primarily autotransplanted parathyroid tissue that has proved function in more than 90% of patients.
Subtotal parathyroidectomy has been reported to provide long-term cure in 85% to 90% of patients who have sporadic primary parathyroid hyperplasia.46 Failure resulting in persistent disease has been related mainly to the presence of overlooked supernumerary glands, whereas remnant recurrence generally has been more rare or developed later during follow-up.46 Remnant recurrence should result from intrinsic properties within the remaining parathyroid tissue, because it has also been difficult to prevent with more radical resection.47 Subtotal parathyroidectomy is preferable if one or two normal-sized glands occur together with the abnormal ones, because normal parathyroids are less suitable for autotransplantation but in our experience serve excellently as remnants.
Total parathyroidectomy with autotransplantation appears advantageous, especially in patients with four markedly enlarged and diseased glands or in the reoperative situation, because tissue that could be selected as remnant has already acquired marked growth potential. Recurrence in forearm grafted tissue should be possible to remove under local anesthesia and therefore is easier to manage than remnant recurrence in the central neck compartment.38,39
Recurrence of hypercalcemia has been encountered in up to one third of patients undergoing total parathyroidectomy and parathyroid autotransplantation.3,14,48 This has often resulted from the presence of overlooked supernumerary glands, but in a proportion of patients with generally mild hypercalcemia it has been caused by graft hyperfunction related to the transplantation of diseased tissue.3,12,49–51
Occasionally, remarkable growth and even infiltration of grafted tissue has occurred, probably because of the inappropriate selection of tissue for transplantation.49–51 A state of prolonged hypocalcemia immediately after surgery is another general problem with this procedure. Regardless, subtotal parathyroidectomy and total parathyroidectomy combined with autotransplantation are probably more or less equally efficient in curing parathyroid hyperplasia, provided that the procedures are adequately performed. Comparison of the two methods is difficult because most failures have resulted from the presence of ectopic or supernumerary glands.3,25,38
Whatever procedure is undertaken, it should be essential to choose the smallest parathyroid gland without macroscopic nodules as the designated remnant in performing a subtotal resection or for grafting in association with total parathyroidectomy.52–54
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree