Chapter 7 Surgery of Cervical and Substernal Goiter
![]() Please go to expertconsult.com to view related video Goiter Surgical Maneuvers.
Please go to expertconsult.com to view related video Goiter Surgical Maneuvers.
The word goiter is derived from “guttur,” the Latin term for throat.2 Surgery for goiter is as complex as it is satisfying. With goiter, the normally complex neck base anatomy is distorted in sometimes predictable and often unpredictable patterns. Size, goiter vascularity, distortion of anatomy, substernal extension, and restrictions imposed by the bony confines of the thoracic inlets can make recurrent laryngeal nerve (RLN) and parathyroid gland identification and preservation challenging. Halsted wrote that “the extirpation of the thyroid gland for goiter better typifies perhaps than other operations, the supreme triumph of the surgeon’s art.”3
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
The operative story of goiter documents the evolution of modern surgical technique. Gunther in 1864 described a case emphasizing the difficulties experienced during early goiter surgery: “After several fruitless attempts at ligation of the arteries, the severe hemorrhage was controlled by compression day and night during eight days by persons alternating with each other at the task” (see Chapter 1, History of Thyroid and Parathyroid Surgery).4
This chapter reviews the patterns of anatomic distortion presented by both cervical and substernal goiter. Substernal goiter represents a distinct subtype of cervical goiter and is discussed separately where appropriate, given its unique challenges. After reviewing key points about goiter definition and clinical evaluation, we shall discuss treatment options, with an emphasis on the surgical approach. The chapter highlights the evaluation of upper airway compromise in patients with goiter, the relationship between the extent of surgery and the likelihood of goiter recurrence, and the predictive risk factors for surgical complications. See also Chapters 8, Approach to the Mediastinum: Transcervical, Transsternal, and Video-Assisted, 9, The Surgical Management of Hyperthyroidism, and 10, Reoperation for Benign Disease.
General Considerations
Goiter Definition
The definition of goiter varies substantially among reports. McHenry suggested 80 g and Russell 100 g, whereas Clark proposed 200 g as the threshold value.5–9 Studies investigating radioiodine treatment for multinodular goiter often define significant goiter as greater than 100 g. Hegedus, Nygaard, and Hansen found that goiter surgical specimens averaged 30 g for unilateral resection and 64 g for bilateral resection.10 In the study by Katlic, Grillo, and Wang, the average weight of substernal goiter was 104 g (range 25 to 357 g), with greatest diameter averaging 9 cm (range 5 to 19 cm).11 In a series of more than 200 cervical and substernal goiters treated at Massachusetts Eye and Ear Infirmary and Massachusetts General Hospital, the mean weight was 143 g and the mean goiter size was 10.5 cm.12
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
The largest goiter we can find documented in the literature is reported by Manoppo, who described a benign, nontoxic goiter that was 75 × 60 × 45 cm. The patient was unable to walk or sit because of the size of the goiter. Treatment was with right hemithyroidectomy under local anesthesia, without major complications.13
The World Health Organization (WHO) 1960 grading system for clinical assessment of goiter defines stage 0 as no enlargement; stages 1 to 3 describe progressive goiter enlargement. Stage 1A includes patients with palpable abnormalities; stage 1B includes patients with palpable and visual abnormalities with the neck in extension. Stage 2 is defined as a goiter that is visible with the neck in neutral position, and Stage 3 as a goiter that is able to be visualized at a considerable distance.14,15 The WHO 1994 goiter classification system is more streamlined. Grade 0 is defined as no palpable or visual abnormality. Grade 1 is defined as a palpable thyroid mass that is not visualized with the neck in neutral position, and grade 2 as a visually apparent mass with the neck in neutral position.16
Substernal Goiter
Definition
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Lahey and Swinton defined substernal goiter as a “gland in which the greatest diameter of the intrathoracic component by x-ray was well below the upper aperture of the thoracic inlet.”17 Crile, in 1939, simply defined substernal goiter as a lesion extending to the aortic arch.18 Lindskog and Goldenberg in 1957 defined substernal goiter as a goiter whose lower border radiographically reaches the transverse process of the fourth thoracic vertebra or lower.19 Katlic, Grillo, and Wang described substernal goiter as when greater than 50% of the goiter is present substernally.11 Sanders et al. defined substernal goiter as that which requires mediastinal exploration and dissection for removal.20 Other definitions have been offered.21–23
Several workers have offered substernal classification schemes. Higgins based his classification scheme on the percentage of goiter in the neck versus the percentage of goiter in the chest, with greater than 50% in the neck being described as substernal, greater than 50% in the chest as partially intrathoracic, and greater than 80% in the chest as completely intrathoracic.24 Cho, Cohen, and Som offered a grading system relating grade to percentage of goiter within the chest. Grade I is defined as 0% to 25% of the goiter within the chest, grade II as 26% to 50%, grade III as 51% to 75%, and grade IV as greater than 75%.25 Shahian offered an interesting and detailed classification scheme. In this classification scheme, type I substernal goiter is associated with the anterior mediastinal extension, type IA involves “isolated” anterior mediastinal disease, whereas type IB involves “extensive” substernal involvement. Type II involves posterior mediastinal involvement, with type IIA being isolated posterior mediastinal goiter, type IIB posterior mediastinal goiter with ipsilateral extension relative to the thyroid lobe of origin, and type IIC contralateral extension relative to the thyroid lobe of origin, with C1 being retrotracheal and C2 being retroesophageal course.26,27
A classification system for substernal goiters is most useful when it takes into account the features of substernal goiters that must be appreciated to extract them safely. We define substernal goiter simply as those goiters that are associated with substernal extension such that the thoracic component requires mediastinal dissection to facilitate extraction. We believe that all substernal goiters require axial computed tomographic (CT) scanning to differentiate between the various subtypes. Such differentiation provides tremendously useful surgical information. We propose the following substernal goiter classification scheme (Table 7-1).
Posterior Mediastinal Goiters (Substernal Goiter Type II)
Most surgical and radiographic series suggest that substernal goiters affect the anterior mediastinum in approximately 85% of patients and the posterior mediastinum in approximately 15% (see Table 7-1).26–29 Extension into the anterior mediastinum brings the mass anterior to the subclavian and innominate vessels and anterior to the RLN. The relationship of the anterior mediastinal goiter to the RLN is as in the normal cervical gland—that is, that the nerve is deep. When substernal goiter expands to the posterior mediastinum, it excavates the region posterior to the trachea, pushing the trachea anteriorly and splaying the great vessels anteriorly. The mass then comes to rest in a space posterior to the innominate vein, carotid sheath contents, innominate and subclavian arteries, RLN, and inferior thyroid artery.26,27,30 Of importance, the relationship of the mass and the RLN is reversed as compared with the normal cervical orthotopic gland-RLN relationship. The RLN is ventral to the inferior component of the mass and, if not recognized early on, can be stretched or cut by even the most meticulous thyroid surgeon. The nerve can also be entrapped between components of the posterior mediastinal goiter; even in these circumstances, a portion of the goiter will be deep to the RLN. Such posterior mediastinal goiters can come to rest in a space bounded inferiorly by the azygous vein, posteriorly by the vertebral column, laterally by the first rib, medially by the trachea and esophagus, and anteriorly by the carotid sheath, subclavian and innominate vessels, superior vena cava, and phrenic and recurrent laryngeal nerves.26,27
![]() Please see the Expert Consult website for more discussion of this topic, including Figure 7-1.
Please see the Expert Consult website for more discussion of this topic, including Figure 7-1.
Posterior mediastinal goiter (type IIA) can occur ipsilateral to the cervical gland of origin or may come to rest through retrotracheal extension in the contralateral thorax (substernal goiter type IIB) (see Table 7-1). Extension to the right thorax is more commonly seen as a result of aortic arch and associated branch vessels obstructing the left posterior mediastinal descent pathway.31,32 Contralateral thoracic extension in the posterior mediastinum may occur either behind the trachea and esophagus (IIB1) or between trachea and esophagus (IIB2). Axial CT scanning and barium swallow help to determine this pattern. Generally the right chest caudal extension is limited at the level of the azygous arch (Figure 7-1).33
Isolated Mediastinal Goiter (Substernal Goiter Type III)
Although rare, thyroid masses within the mediastinum may exist without connection to the normal cervical orthotopic gland. Such purely isolated mediastinal goiters represent only 0.2% to 3% of all goiters requiring surgical treatment.11,24,34,35 Such lesions are important to recognize because unlike all other types of substernal goiters, blood supply of the isolated mediastinal goiter may be through purely mediastinal arteries (including the aorta, subclavian, internal mammary, thyrocervical trunk, and innominate) and veins. This is extremely important in planning their surgical resection.11,24,36–40 This entity is best termed isolated mediastinal goiter. Other terms have been used, including aberrant mediastinal and ectopic mediastinal goiter.
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Three explanations exist for isolated mediastinal goiter. Embryologic fragmentation of thyroid anlagen with hyperdescent, likely associated with cardiac and great vessel descent, may explain some cases of isolated mediastinal goiter (see Chapters 6, Thyroglossal Duct Cysts and Ectopic Thyroid Tissue, and 10, Reoperation for Benign Disease).41,42 Alternatively, isolated mediastinal goiter may form as an exophytic nodule, through progressive attenuation of the nodule-thyroid stalk.41,43,44 Finally, the isolated mediastinal goiter may form as a parasitic nodule representing a thyroid tissue fragment implant in the upper mediastinum from past goiter surgery. We have seen such implants also within the perithyroid area and posterior to the upper cervical segment of the carotid artery.
Prevalence, Pathogenesis, and Natural History
Prevalence
Multinodular goiter affects 4% of the U.S. population and up to 10% of the British population.45 New thyroid nodular disease occurs in from 0.1% to 1.5% of the general population per year.46,47 Globally, iodine deficiency contributes to the vast majority of cases of multinodular goiter and is estimated to affect 1.5 billion people, or nearly 30% of the world’s population in 1990.48 Further, it is estimated that approximately 655 million people in 118 countries are affected by endemic goiter. Endemic goiter regions are defined as iodine-deficient regions in which at least 5% to 10% of the population is affected by goiter. In certain iodine-deficient regions, higher goiter rates occur. In 1994, in Bangladesh, approximately 47% of the population was affected by endemic goiter.49 The majority of the natural iodine supply exists as iodide in the world’s oceans. It is therefore mainly noncoastal mountainous and lowland regions—where iodine is leached from the soil by flooding, heavy rainfall, and deforestation—that are at risk for endemic goiter. Sporadic forms of multinodular goiter do occur in iodine-replete regions with lesser prevalence.16,48 Prevalence estimates of sporadic goiters vary between authors, ranging from less than 4% (clinical evaluation series) and between 16% and 67% (ultrasound series).50,51
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Based on tuberculosis screening radiography in Australia and in the United States, substernal goiter has been estimated to be present in 0.02% of general population and 0.05% of females older than 40.52,53 The incidence of substernal goiter was found to significantly increase with age, with 60% of substernal goiters occurring in patients older than age 60.52 Rates of substernal goiter in the past several decades appear to be decreasing, perhaps related to the introduction of iodized salt, thyroid hormone suppressive therapy, radioiodine use in selective cases, and perhaps more sensitive detection and earlier intervention.2,54 Substernal goiter, as a percentage of patients undergoing thyroidectomy, ranges depending on the series from less than 1% to greater than 20%, with most suggesting a rate of approximately 10%.11,17,18,27,55–59 Substernal goiters represent approximately 5% of all mediastinal tumors.27,60
Pathogenesis
The pathogenesis of goiter formation has classically relied on the notion that iodine deficiency promotes persistent elevated thyroid-stimulating hormone (TSH) levels, inducing diffuse thyroid enlargement through thyrocyte proliferation. Nodules form in the enlarged thyroid as the patient ages, eventually giving rise to multinodular goiter.61,62 Since the early 2000s, this classic model has been challenged by the view that the thyroid gland has an intrinsic propensity to form nodules over time. In this new model, low iodine levels and elevated TSH are considered additional factors, exacerbating the innate process of nodule formation.63,64
The shift in conceptualization of goiter pathogenesis has stemmed from new cellular models, first proposed by Studer and Derwahl.63–66 These authors have argued that the development of multinodular goiter, at least in the later stages, is independent of TSH levels. Nodules arise because thyroid follicles are embryologically derived from polyclonal progenitors and have thus a heterogeneous sensitivity to TSH signaling.67 Thyroid follicles have similarly a differential growth response to continuous exposure to goitrogens, explaining the multinodular pattern of goiters. In addition, thyrocytes may undergo somatic mutations and acquire a distinct growth capacity.68,69 The contribution of somatic mutations in multinodular goitrogenesis remains, however, controversial. This is in contrast with rare growth-promoting germline mutations of the TSH receptor gene, which are believed to be pivotal in congenital diffuse goiter with hyperthyroidism.70 Importantly, activating mutations in the TSH receptor gene have not been associated with an increased risk of malignancy.71
Natural History
The natural history of untreated, sporadic, nontoxic goiter is not completely understood, but slow growth appears to be the general predictable pattern. Berghout et al. suggested a steady volume increase of up to 10% to 20% per year.72 Pregnancy, iodine deficiency, consumption of goitrogens, and alteration in suppressive or antithyroid medical regimens can result in goiter progression. Hemorrhage into a preexisting nodule can also result in the development of acute, regional, and airway symptoms.73 In patients presenting with diffuse goiter, there is a general tendency toward nodule formation and progressive autonomy, with hyperthyroidism ultimately developing in up to 10% of patients (see Chapters 5, Hyperthyroidism: Toxic Nodular Goiter and Graves’ Disease, and 9, The Surgical Management of Hyperthyroidism).74
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Most substernal goiters arise in the setting of preexisting cervical goiter. It is of note, however, that some patients with substernal goiter have no significant cervical goiter component. Substernal goiters virtually always have a connection to the cervical orthotopic gland. Lahey, in his vast experience of approximately 24,000 goiter surgeries, believed that all substernal goiters arise from the cervical gland and maintain their cervical blood supply.75 Even in cases of extreme substernal goiter extending to the diaphragm, the mediastinal component has been found to contain connections to the cervical gland and a blood supply from the inferior thyroid artery.76–78 Connection to the cervical gland may be robust or attenuated, but it virtually always exists. The work of Torre, based on an impressive series of 237 substernal goiters, suggests that substernal goiters arise 10 years after cervical goiter presentation, suggesting that substernal goiter evolves from preexisting cervical goiter in most cases.8
The inferior extension of cervical goiter and formation of substernal goiters is poorly understood. The inferior descent relates in part to the pattern of nodular disease within the cervical gland. Inferior progression results from a limitation of the strap muscles anteriorly, trachea medially, and vertebral column posteriorly. As Lahey and Swinton have described, the neck is “a space with no bottom.”17 The repetitive forces of deglutition, respiratory dynamics, negative intrathoracic pressure, and gravitational forces in the setting of permissive mediastinal and neck base fascial planes facilitate the downward extension of cervical goiter. Typically, anterior mediastinal extension (substernal goiter type I) occurs from the ipsilateral lobe’s inferior expansion. Descent associated with significant retrotracheal posterior mediastinal extension may arise from more posterior elements of the thyroid gland such as posterior tubercles of Zuckerkandel. (see Table 7-1).
Clinical Presentation
Cervical Goiter
History
The history of goitrous growth and associated symptoms is critical for determining surgical candidacy. This history should be obtained not only from the patient but also from his or her family. Regional symptoms should be addressed relating to respiration, phonation, swallowing, and presence of globus (lump sensation). As Pemberton emphasized in 1921, symptoms associated with goiter may be positionally induced.38 Positions that may provoke goiter regional symptomatology include being supine, arms raised (as when reaching for an upper cabinet), extreme neck extension, extreme neck flexion (as with reading a book in bed), and turning the head to the extreme left or right. Patients thus need to be questioned about positional provocation of regional symptoms. In addition, the family needs to be questioned about nocturnal symptoms, as symptoms may manifest initially in the setting of recumbency and upper airway relaxation during sleep. Symptoms may also be associated with exercise and increased oxygen demands. A history of preceding upper respiratory tract infection may produce dyspnea in a patient with long-standing tracheal obstruction secondary to goiter through new laryngotracheal mucosal edema. Patients with cervical or substernal goiter may present with cough, dyspnea, foreign-body sensation, neck tightness, change in collar size, or wheezing and may come to the head and neck surgeon with a misdiagnosis of asthma or chronic obstructive pulmonary disease (COPD). In our series of patients with large cervical and substernal goiter, we found that 25% of patients were preoperatively asymptomatic.12
Symptoms of hypothyroidism and hyperthyroidism should be reviewed. Hyperthyroidism may slowly evolve in patients with multinodular goiter or may develop acutely in response to significant iodine load such as with CT scan contrast (Jod-Basedow phenomenon) or with the introduction of iodized salt in endemic goiter regions.79 A history of migration from an area of endemic goiter should be obtained, as well as a history of exposure to known goitrogens, notably iodine and lithium. A family history of thyroid disease should be obtained.
Physical Examination
After documentation of thyroid size, the examiner should note the consistency and fixation of the mass, especially with respect to the larynx and trachea. Estimation of goiter size by physical examination is clearly an inaccurate method of assessment. Jarlov et al. found substantial errors in the clinical assessment of thyroid size as compared with ultrasonographic assessment.80 Estimated weight based on the physical examination generally underestimates multinodular goiter weight by 25 to 50 g.46 The larynx (landmarks include thyroid notch and cricoid anterior arch) and trachea should be examined for deviation from the midline. Typically, cervical goiter will deviate the larynx and trachea to the contralateral side. The neck must be examined for adenopathy as well as scarring from past thyroid and other neck surgery. Jugular distention and subcutaneous venous redistribution should be noted. Although this may be present with large benign cervical or substernal goiter, true superior vena cava syndrome is generally due to malignant thyroid disease and warrants careful scanning and evaluation.11
It is imperative in all patients with goiter that the larynx be examined. In our series, we have found that 2% of patients with goiter presented with vocal cord paralysis in the setting of benign disease and no prior neck surgery, and 3.5% of preoperative patients presented with goiter overall.12 Vocal cord paralysis without a history of past thyroid surgery implies invasive thyroid malignancy until proved otherwise. It should be noted, however, that benign goiter has also been associated with vocal cord paralysis, presumably through stretch, which may recover postoperatively (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve).81 Certainly, such a preoperative finding focuses the surgeon’s attention on the extreme importance of preserving the contralateral RLN. The laryngeal examination in patients with large cervical goiter can be difficult if there is edematous or redundant supraglottic mucosa, laryngeal compression and deviation, and hypopharyngeal crowding resulting from goitrous extrinsic compression. Symptomatic assessment of the voice, like symptomatic assessment of the airway, does not predict objective findings in patients with goiter and should not replace the laryngeal exam. In our patient series, voice change was reported preoperatively in 12.8%, but vocal cord paralysis was present in only 3.5%, consistent with the work of Michel, emphasizing that glottic function cannot be predicted by voice assessment.82,83 Michel noted in his series of substernal goiters that although hoarseness was described in 26%, vocal cord paralysis was only found in 3%.89 We thus reiterate our recommendation that all patients with goiter undergo preoperative laryngeal examination.
Substernal Goiter
In many ways the history and physical examination for patients with substernal goiter overlaps significantly with those for patients with cervical goiter. In our series, we have found that as cervical goiter progresses substernally, given the restriction of the bony confines of the thoracic inlet, it increasingly compromises the airway. In short, substernal goiter evolution is strongly correlated with tracheal deviation, the development of regional airway symptoms, and radiographic airway compression.12 Buckley and Stark noted that although the maximum tracheal deviation with substernal goiter usually occurs at the thoracic inlet, it may occasionally occur farther inferiorly.33 Larger surgical series of substernal goiter show 70% to 80% of substernal goiter patients are symptomatic at presentation. Cervical mass is noted in from 69% to 97%, respiratory symptoms in 42% to 96%, dysphagia in 26% to 60%, and acute airway presentation in from 1% to 5%.2,8,11,19,20,25,55,84–88 Interestingly, these series show that 10% to 30% of substernal goiter patients, as previously noted, have no significant palpable cervical abnormality, 3% to 7% present with vocal cord paralysis, and 4% to 50% are asymptomatic at presentation. Wax and Briant have noted that, with careful questioning, up to one third of patients who are “asymptomatic” admit to symptoms.88
Pemberton’s sign is described as the development of head and neck venous engorgement with facial congestion, plethora, and venous distention with arms raised over the head. It is sometimes expanded to include the development of transient respiratory insufficiency. Pemberton’s sign is thought to indicate goiter extension into the thoracic inlet, with secondary relative venous and airway obstruction.38,58,90 Our series of large cervical and substernal goiters suggests that Pemberton’s sign is insensitive in the evaluation of substernal goiter, as only 4.4% of patients presented with a positive Pemberton’s sign.12 Substernal goiters can also present with neck and upper chest pain and have rarely been associated with hematemesis secondary to downhill esophageal varices (without signs of portal hypertension), abscess formation, Horner’s syndrome, chylothorax (secondary to thoracic duct obstruction), transient ischemic attacks through “thyroid steal syndrome,” venous thrombosis, and intubation injuries, especially to the posterior tracheal membranous wall.56,91
Laryngeal shift to the side of a dominant cervical goiter suggests contralateral substernal goiter and requires axial imaging of the neck and chest. Similarly, laryngeal shift without any palpable cervical findings suggests substernal goiter and similarly requires axial neck and chest imaging.11,19,79,84,92 Finally, substernal goiter is suspected when the clavicle intervenes before the inferior extent of the thyroid mass can be palpated.
Goiter Workup
In the workup of patients presenting with a goiter, the clinician may address the following three important issues: (1) the existence or the potential development of airway compression, (2) the risk of malignancy, and (3) the presence of hyperthyroidism (Boxes 7-1 and 7-2).
Box 7-1 Airway Imaging, Flow Volume Loops, and Goiter Symptoms: Summary
• The presence of preoperative shortness of breath correlates with goiter size, but it is of limited value as a screening tool for tracheal abnormalities.
• Dysphagia correlates with radiographic findings of esophageal deviation and compression. In the absence of dysphagia, patients do not require further esophageal imaging.
• Symptomatic assessment of voice does not predict objective findings in patients with goiter and should not replace the laryngeal exam.
• Flow volume loop studies most accurately document airway obstruction in the setting of significant airway compression. However, they correlate poorly with goiter weight and upper airway symptoms. We do not recommend flow volume loop studies as part of the routine work-up for patients with goiter.
• Thus, symptomatic flow volume loop and plain film assessment of goiter is insensitive, in distinction to axial CT scanning. The finding of tracheal compression on axial CT scanning correlates significantly with the presence of shortness of breath; therefore, we consider the finding of CT scan tracheal compression to be an appropriate surgical indication given its symptomatic respiratory correlate.
Box 7-2 Workup for Benign Goiter
CT, Computed tomography; MRI, magnetic resonance imaging.
Acute Airway Compromise
![]() Please see the Expert Consult website for more discussion of this topic, including Figure 7-2.
Please see the Expert Consult website for more discussion of this topic, including Figure 7-2.
Acute Airway Compromise
The frequency of development of acute airway compromise in cervical and substernal goiter varies depending on the population studied. Unfortunately, most of the information available is from surgical series. Allo and Thompson have estimated that from 1% to 3% of patients with untreated mediastinal goiter die of respiratory obstruction.55
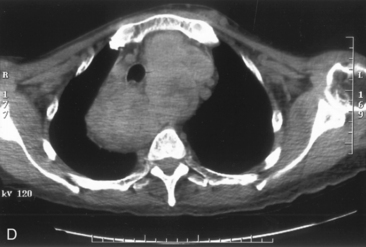
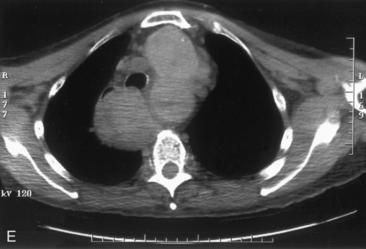
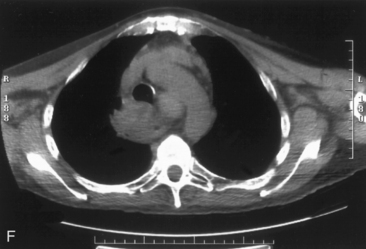
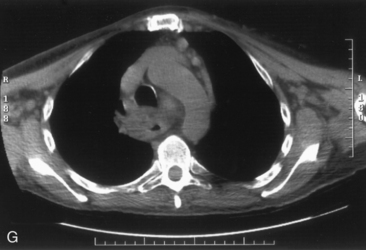
The airway can be affected by goiter through a number of mechanisms, including, most typically, ongoing slowly progressive airway deviation and compression. Acute events can also precipitate airway compromise, including hemorrhage into a nodule of multinodular goiter. Georgiadis has described acute stridor caused by hemorrhage within a nodule after neck trauma.93 Pulli and Coniglio have described a patient whose goiter increased in size after a fall on ice.2 Torres et al. have presented a number of cases showing acute life-threatening tracheal obstruction in patients with long-standing, intrathoracic goiter with subsequent histology showing multiple foci of recent hemorrhage.94 Hemorrhage as a mechanism of goiter size increase is supported by the work of Bodon and Piccoli.95 We have seen acute tracheal obstruction with magnetic resonance imaging (MRI) scan evidence of central nodular hemorrhage in what was ultimately found to be a large benign follicular adenoma (see Figure 7-2). Sudden airway symptoms may also arise as a result of cystic degeneration, malignant degeneration, upper respiratory tract infection, or pregnancy.96 Rare mechanisms for respiratory distress in patients with large goiters include decompensated right heart failure, pleural effusion, and pulmonary hypoperfusion secondary to compression of the pulmonary arteries.90 We agree with Cougard et al. that a patient with goiter and acute airway decompensation requiring endotracheal tube intubation should be brought to surgery when stable, while intubated. Ultimately, after thyroid surgery, laryngeal examination should be performed to rule out laryngeal edema and assess vocal cord function before extubation.97
Miller et al., in a nonsurgical series of 400 patients with goiter, evaluated patients with pulmonary function tests and flow volume loops. Nearly one third of such patients had flow volume evidence of upper airway obstruction.9 Gittoes et al., in a study of 153 goiter patients followed in a medical/endocrine clinic, also found that 33% of such patients had evidence of upper airway obstruction on flow volume loop analysis.45 Thus, two large nonsurgical series suggest that up to one third of patients with large goiters followed in endocrine clinics have evidence of upper airway obstruction as defined on flow volume loops. Limited data are available within this population as to the rate of development of acute airway symptoms. Alfonso found, in a surgical series of 91 patients with benign goiters with either radiographic or symptomatic evidence of upper aerodigestive tract compression, that approximately 9% of patients presented with acute upper airway obstruction.98 Reeve, Rubenstein, and Rundle, in a large screening radiographic study that identified patients with significant thyromegaly as defined by plain radiographs, found 7.6% of such patients presented with “profound respiratory obstruction.”52 In a surgical series of patients with substernal goiter, reports vary, with 1.3% to 5% of such patients presenting with acute airway insufficiency.11,25,55,84,85,92,99,100 Higher rates of acute airway insufficiency have been reported in other surgical series.86,101
Thus, approximately one third of patients with goiter in medical series have upper airway obstruction as defined by flow volume loops, and from 1.3% to 9% of such patients may present with acute airway symptomatology. Unfortunately, a long, chronic, stable history does not preclude spontaneous acute airway insufficiency.55 Warren has beautifully documented cases of acute respiratory failure secondary to goiter in elderly patients. All presented with acute respiratory collapse without a history of respiratory symptoms 48 hours before the respiratory failure.100 Cho, Cohen, and Som also emphasized, in a review of 70 patients with substernal goiter, the potential for sudden and unpredictable respiratory distress.25
There is sometimes a tendency for patients with large compressive goiters to be followed with flow volume loops and symptomatic assessment. There is no evidence to suggest this is a rational approach. It is known that flow volume loops begin to detect airway obstruction when tracheal diameter is reduced to an extremely limiting 5 mm.102 Symptoms of acute airway insufficiency may not occur until up to 75% of the tracheal lumen is obstructed.94,103 At our institution, serial axial CTs are recommended to determine surgical candidacy, most typically by documenting substernal extension or tracheal compression.12 We have found that regional signs and symptoms, such as upper airway obstruction or radiographic evidence of tracheal or esophageal compression, are more likely with masses larger than 5 cm. Also, fine-needle aspiration (FNA) represents a less accurate assessment with lesions of this size as compared with thyroid nodules between 1 and 3 cm.
Evaluation of Upper Airway Compromise and Other Regional Symptoms
Regional Symptomatic Assessment
Upper airway obstruction is a common finding in patients with goiter, highlighting the importance of optimal airway evaluation. Common presenting symptoms in our series of patients with goiter included shortness of breath (approximately 50%) and dysphagia (approximately 50%), emphasizing the effect of cervical and substernal goiter on the adjacent cervical viscera. Although earnest symptomatic assessment at presentation is crucial in patients with goiter, clinical experience suggests subjective symptomatic assessment of the upper aerodigestive tract compressive symptoms in patients with goiter can be quite problematic. Although we found that the presence of shortness of breath correlates with goiter size, shortness of breath as a screening tool for tracheal deviation or compression is of limited value. This is despite the fact that the presence of shortness of breath is significantly related to the imaging finding of tracheal compression, consistent with the work of Mackle.104 For the airway, symptomatic assessment alone may therefore be inadequate in goiter patients. In the setting of large cervical or substernal goiter, one cannot purely rely on the presence or absence of shortness of breath to assess true tracheal compromise, without routine axial CT scanning assessment for tracheal compression. In our series there was no significant difference in the percentage of patients with airway symptoms between patients with purely cervical and patients with substernal goiter. However, in our series substernal goiter was highly associated with tracheal deviation and compression.12
Our series also demonstrated a positive correlation between thyroid size and globus sensation and symptoms of hyperthyroidism. No correlation was established between goiter size and presence of dysphagia, local discomfort, change in voice, hemoptysis, or symptoms of hypothyroidism. There was a significant positive correlation between preoperative dysphagia and the presence of esophageal compression and deviation.12
Flow Volume Loop Analysis
Although symptomatic assessment is important in patients with cervical and substernal goiter, the relationship between such symptoms at presentation and pulmonary function evaluation is problematic. Flow volume loops have been utilized to define airway compromise in goiter patients. In our centers, flow volume loop analysis is not part of the routine workup for goiter. Goiter weight is not well correlated with flow volume loop results.9,45 Bonnema has also reported that overall flow volume loop results do not correlate well with tracheal caliber on axial CT.105 Only 30% to 40% of those with flow volume loop abnormalities have symptoms, and among those with normal flow volume loops, up to 60% may have airway symptoms.
This discordance between flow volume loop results and symptoms or objective axial CT scan findings in goiter patients should not be surprising. It is well known that lung function indices are inconsistently related to tracheal size in normal subjects.106,107 Poiseuille’s law (flow proportional to radius) implies that with a significant reduction in the tracheal airway, small changes in tracheal caliber will be reflected in significant changes in flow volume loop characteristics, but with lesser degrees of tracheal narrowing, the two variables are more weakly related. In fact, it is known clinically that tracheal compression of up to 75% of the tracheal lumen may occur without clinical manifestation.9,94 Flow volume loop studies can detect tracheal stenosis when tracheal diameter is less than 5 mm, but it may not be affected in less severely narrowed airways.102 Flow volume peak inspiratory flow of less than 1.5 L has been associated with a high risk of acute respiratory failure and therefore has been used as an indication for urgent thyroidectomy.9,94,98
Imaging
Radiographic Evaluation and Regional Symptomatology
A number of studies question the strength of the correlation between regional symptoms and imaging study findings. In particular, the functional impact of airway narrowing diagnosed via CT scan remains controversial. Alfonso et al. found that two thirds of surgical goiter patients had preoperative radiographic evidence of compression. Almost half of those patients with evidence of compression had no symptoms. Those patients with airway symptoms who had old radiographs available for comparison were found to have compression up to 3 to 4 months before the onset of airway symptoms.98 Jauregui found in a series of asymptomatic euthyroid goiter patients that 25% had radiographic evidence of tracheal obstruction and 60% had evidence of airway obstruction by flow volume loops.108 Cooper et al. found that tracheal diameter and airway symptoms are weakly related.109 Melissant and others have found little correlation between lung function and the CT scan–defined degree of tracheal obstruction.105,110
Conclusions from our series stand in contrast with the above literature. As noted earlier, we found a significant correlation between the presence of shortness of breath and the objective CT scan radiographic finding of tracheal compression.12 Barker et al. similarly noted that if CT scan showed greater than or equal to 50% tracheal diameter narrowing, symptoms should be expected.111 We therefore consider tracheal compression to be an important radiographic finding and an appropriate surgical indication in these patients (Boxes 7-1 and 7-2).
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
The relationship between esophageal symptoms and abnormal barium swallows is less contested. Alfonso et al. found all 25 patients with dysphagia also had abnormal barium swallows.98 In our series, dysphagia was significantly associated with both radiographic esophageal deviation and compression, as evaluated with barium swallow studies. The absence of dysphagia was associated in our series with a negative predictive value of 96% for esophageal compression.12 We therefore consider that patients without dysphagia likely do not have esophageal involvement and do not require further esophageal imaging. (See Box 7-1.)
Chest Radiography and Barium Swallow Study
![]() Please see the Expert Consult website for more discussion of this topic.
Please see the Expert Consult website for more discussion of this topic.
Chest Radiography and Barium Swallow Study
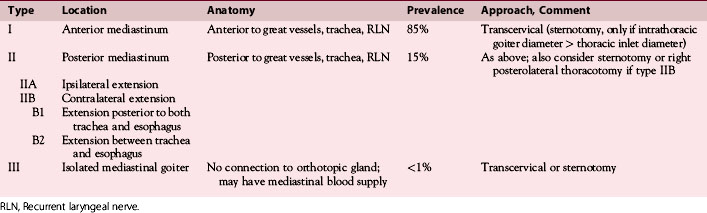
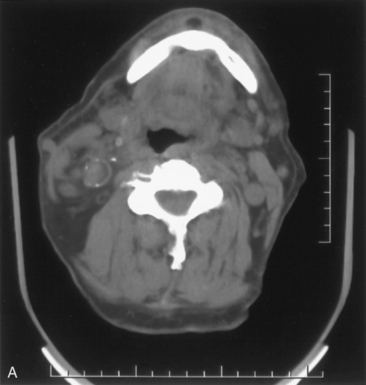
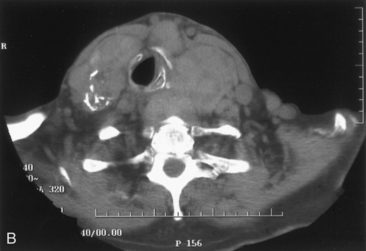
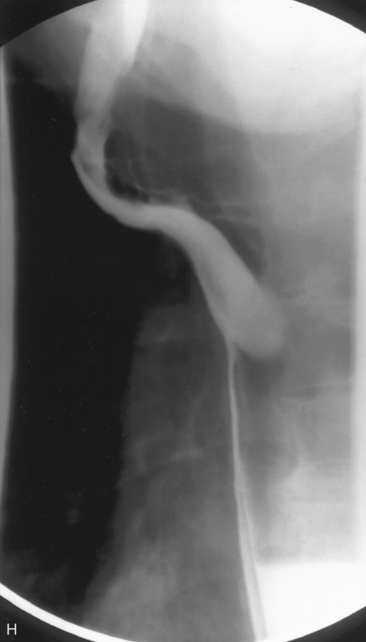
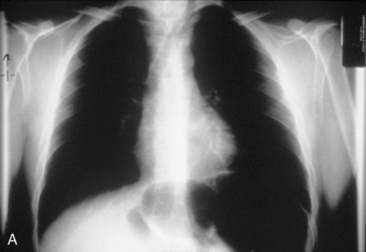
Chest radiography preoperatively provides limited information regarding tracheal air column and may allow detection of macronodular pulmonary metastasis in patients with thyroid enlargement. Chest radiographs may reveal cervical or mediastinal densities with or without calcification and may, with substernal extension, identify a plane of reflection of the mediastinal pleura. Chest radiograph is read as abnormal in up to 40% to 90% of patients with substernal goiter.11,22,56,83,87,112 Several workers have noted that tracheal diameter as estimated from plain films is significantly greater than tracheal diameter as measured on CT scan, axial studies, or in cadaveric studies.111,113 Melissant et al. found that plain films missed significant tracheal obstruction in 50% of patients.110 Cooper et al. also found in review of cervical and substernal goiter patients that plain films were misleading in 48% of patients, resulting in both over- and underestimation of tracheal narrowing as compared with CT axial scanning.109 Plain chest radiograph can be especially misleading in the rare posterior mediastinal goiter with contralateral extension retrotracheally with respect to the lobe of origin (substernal goiter type IIB). Tracheal deviation in this complex circumstance may be toward the size of the mass as seen on plain radiographs.26,27,114 We have also found that chest radiograph generally gives reasonable information regarding the degree of tracheal air column deviation, but it does significantly underestimate tracheal compression (see Figure 7-2).12 In addition, when a goiter is present bilaterally, it may symmetrically compress the airway without any deviation. In such circumstances, axial CT scanning may reveal a significantly compressed airway, yet plain chest radiograph may fail to show any distortion of the tracheal air column.
Barium swallow, although not generally helpful in the preoperative evaluation of patients with cervical and substernal goiter, may be helpful in posterior mediastinal goiter. Michel found that sensitivity for the identification of substernal goiter by chest radiograph was 59%, by thyroid scanning it was 77%, and with barium swallow it was 71%, emphasizing the inaccuracies of these modalities in goiter evaluation (Box 7-2).83 Our recommendation is to not perform barium swallow studies in the absence of dysphagia, as the presence of esophageal compression is unlikely in asymptomatic patients.12
Axial CT Scanning
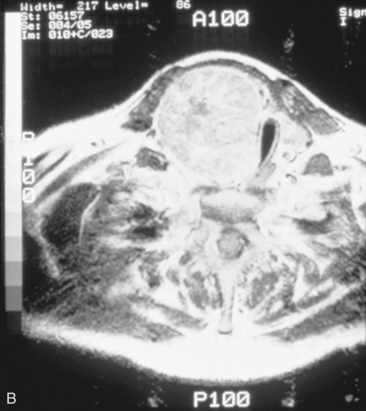
We have found routine CT scanning to be very helpful in preoperative assessment of patients with large cervical or substernal goiters.12 CT can be performed safely (i.e., without the Jod-Basedow phenomenon) in patients with thyroid disease, if TSH is not found to be suppressed. CT scanning shows the margin of benign goiter to be smooth and may often delineate gross calcification (which may be punctate, linear, eggshell, amorphous, or nodular), versus the fine stippled microcalcification that may be present in papillary or medullary carcinoma (see Chapter 13, Ultrasound of the Thyroid and Parathyroid Glands). In patients with substernal goiter, continuity of the mediastinal mass and cervical orthotopic gland can be identified. Precontrast attenuation of thyroid tissue exceeds adjacent neck musculature by at least 15 Hounsfield units or greater and by more than 25 Hounsfield units after contrast enhancement. In patients with substernal goiter, this high attenuation, which is uncharacteristic in other types of mediastinal disease such as lymphoma or thymoma, can be helpful diagnostically.33,115 With CT scanning, the extent of cervical and substernal extension can be accurately defined, and the exact relationship of the goiter to the trachea, esophagus, and great vessels can be determined. The presence of nodal disease can also be established through axial scanning. Generally, benign goiter shows inhomogeneous density with discrete nonenhancing low-density areas. Malignancy may be considered in the setting of the radiographic findings of irregular/infiltrative margins, vocal cord paralysis, and nodal enlargement, especially if the nodes are calcified, cystic, or enhancing.116–118 Contrary to McHenry’s view that preoperative radiographic evaluation does not alter intraoperative management, we believe that CT scanning is essential for all patients with large cervical and substernal goiters.7 We are more likely to obtain CT if the clinical exam suggests that the goiter is large, symptomatic, bilateral, or substernal or if malignancy is suspected based on vocal cord paralysis or regional lymphadenopathy (see Box 7-2). CT scanning ideally shows the relationship of the goiter to surrounding cervical viscera, including the airway. These relationships can affect not only surgery but also approach to intubation. Axial CT provides objective, reproducible measures of tracheal caliber. A patient’s surgical candidacy may derive from information obtained from axial CT scanning, especially given a lack of sensitivity of symptomatic, flow volume loop analysis, and plain radiographic assessment. At our institutions, documentation of substernal extension or tracheal compression on CT is an appropriate surgical indication.12 CT scanning also provides helpful information to exclude invasive malignancy. It is true that CT scanning does not differentiate well between fibrosis and tumor, although this distinction is generally not a significant clinical issue for routine goiter patients. Finally, enhanced CT scanning is essential in determining the mediastinal relationships to allow safe operative management for large posterior mediastinal goiters. Identification of significant retrotracheal extension of either cervical or substernal goiter helps to preoperatively predict that the RLN will be in a ventral position. Preoperative information of ventral nerve displacement is tremendously helpful in the offering of safe cervical and substernal goiter surgery (Box 7-3).
Box 7-3 Imaging: Summary
• Plain chest radiography may detect macronodular metastatic disease but generally offers limited information about the tracheal air column. Airway compression is underestimated and bilateral goiter with circumferential tracheal compression may not be well seen on plain radiographs.
• We recommend CT scanning in all patients with large cervical and substernal goiters. Axial CT scanning has the advantage of being readily available and easily interpreted by the surgeon. CT scanning is used to judge a patient’s surgical candidacy by accurately defining the degree of tracheal impact. In patients with substernal goiter, CT scanning informs surgical planning regarding sternotomy and potential thoracic surgical involvement. CT scanning is helpful in identifying the radiographic correlates of malignancy. It is also of tremendous importance in accurately defining the anatomic relationships, especially in predicting when an RLN will be ventral through retrotracheal and posterior mediastinal extension. Contrast should be avoided if the patient’s thyroid functional status is unknown or if the patient is subclinically hyperthyroid.
• Advantages of MRI scanning are excellent soft-tissue delineation, excellent definition of the goiter’s relationship to mediastinal vessels, and sagittal and coronal display. Disadvantages of MRI scanning include cost, patient claustrophobia in nonopen MRI scanning suites, and poor definition of calcification patterns.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree