Gamma Knife stereotactic radiosurgery (GKS) has become an important management strategy for an increasing number of patients with skull base tumors. For select patients with lateral skull base disorders, given the proximity to sensitive critical structures such as the brainstem, cranial nerves, and cochlea, this technology has emerged as a first-line treatment to achieve the paramount goals of long-term tumor control and maintenance of existing neurologic function. This article reviews the indications, technique, and results of GKS for the treatment of vestibular schwannoma and glomus jugulare tumors, and highlights our experience in treating these tumors at the Mayo Clinic.
Key points
- •
Gamma knife radiosurgery provides excellent functional preservation and tumor control in long-term follow-up for select patients with vestibular schwannomas or glomus jugulare tumors.
- •
Excellent pretreatment hearing is the most reliable predictor of long-term hearing preservation following stereotactic radiosurgery.
- •
Long-term follow-up is necessary to critically assess outcome after stereotactic radiosurgery.
Introduction and technique
Gamma Knife stereotactic radiosurgery (GKS) was first instituted at the Mayo Clinic in 1990, being the third Gamma Knife unit to be installed in the United States, resulting in extensive experience in treating lateral skull base tumors over the past 24 years. The Leksell Gamma Knife (Elekta Instruments, Norcoss, GA) uses either 201 (models U, B, C, 4-C) or 192 (model Perfexion) fixed cobalt 60 radiation sources that can be collimated to radiation beams of 4-mm, 8-mm, 14-mm, or 18-mm ovoids with the earlier models. The current Perfexion model allows the radiation to be collimated to 4-mm, 8-mm, or 16-mm ovoids of radiation and in addition each shot is divided into 8 sections of 24 beams of radiation that can potentially be completely blocked or collimated to the 4-mm, 8-mm, or 16-mm sizes to allow more complex shapes than just the original ovoids. This ability also has the advantage of allowing blocking patterns to potentially protect critical radiation-sensitive structures such as the cochlea or optic nerves and chiasm.
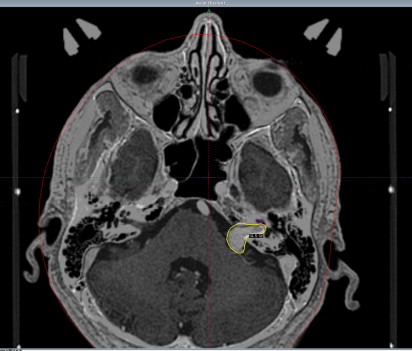
A 4-point stereotactic head frame is applied after infiltration of the pin sites with local anesthetic. Detailed stereotactic imaging of the tumor and adjacent critical temporal bone structures is then performed using MRI and computed tomography (CT). A postgadolinium axial spoiled gradient recalled (SPGR) acquisition in the steady state MRI with 1-mm thick, volumetrically acquired slices (28–60 images) is performed to encompass the tumor and surrounding pertinent anatomy. The patient is then taken to the CT scanner where a noncontrast axial CT scan of the temporal bones is obtained with 1-mm thick slices. This technique allows the user to contour the bony cochlea and determine an accurate volumetric dose to the cochlea. In addition, this enables clinicians to minimize the image distortion of stereotactic MRI. It also allows better delineation of tumor involvement into the temporal bone in patients with, for example, paragangliomas. This imaging is directly transferred to a computer workstation. MRI and CT can also be fused using the Gamma Plan software (Elekta Instruments, Norcross, GA). A conformal radiation dose plan is then developed using a combination of isocenters that are derived by way of collimators of various sizes and differential weighting, after which the treatment is performed ( Fig. 1 ). Patients are dismissed following the procedure from the outpatient observation ward without restrictions. We obtain follow-up audiograms and MRI scans at 6-month intervals for the first year, then yearly for the next several years, eventually moving to MRI scans every 3 to 4 years if no evidence of tumor growth is detected.
Introduction and technique
Gamma Knife stereotactic radiosurgery (GKS) was first instituted at the Mayo Clinic in 1990, being the third Gamma Knife unit to be installed in the United States, resulting in extensive experience in treating lateral skull base tumors over the past 24 years. The Leksell Gamma Knife (Elekta Instruments, Norcoss, GA) uses either 201 (models U, B, C, 4-C) or 192 (model Perfexion) fixed cobalt 60 radiation sources that can be collimated to radiation beams of 4-mm, 8-mm, 14-mm, or 18-mm ovoids with the earlier models. The current Perfexion model allows the radiation to be collimated to 4-mm, 8-mm, or 16-mm ovoids of radiation and in addition each shot is divided into 8 sections of 24 beams of radiation that can potentially be completely blocked or collimated to the 4-mm, 8-mm, or 16-mm sizes to allow more complex shapes than just the original ovoids. This ability also has the advantage of allowing blocking patterns to potentially protect critical radiation-sensitive structures such as the cochlea or optic nerves and chiasm.
A 4-point stereotactic head frame is applied after infiltration of the pin sites with local anesthetic. Detailed stereotactic imaging of the tumor and adjacent critical temporal bone structures is then performed using MRI and computed tomography (CT). A postgadolinium axial spoiled gradient recalled (SPGR) acquisition in the steady state MRI with 1-mm thick, volumetrically acquired slices (28–60 images) is performed to encompass the tumor and surrounding pertinent anatomy. The patient is then taken to the CT scanner where a noncontrast axial CT scan of the temporal bones is obtained with 1-mm thick slices. This technique allows the user to contour the bony cochlea and determine an accurate volumetric dose to the cochlea. In addition, this enables clinicians to minimize the image distortion of stereotactic MRI. It also allows better delineation of tumor involvement into the temporal bone in patients with, for example, paragangliomas. This imaging is directly transferred to a computer workstation. MRI and CT can also be fused using the Gamma Plan software (Elekta Instruments, Norcross, GA). A conformal radiation dose plan is then developed using a combination of isocenters that are derived by way of collimators of various sizes and differential weighting, after which the treatment is performed ( Fig. 1 ). Patients are dismissed following the procedure from the outpatient observation ward without restrictions. We obtain follow-up audiograms and MRI scans at 6-month intervals for the first year, then yearly for the next several years, eventually moving to MRI scans every 3 to 4 years if no evidence of tumor growth is detected.
Vestibular schwannoma treatment
Dose
Dose planning and treatment of vestibular schwannoma (VS) have evolved significantly over the last several years, accounting for improved cranial nerve morbidity without compromising tumor control. During the early years of VS treatment at the Mayo Clinic, the mean tumor margin dose was 18 Gy (range, 16–20 Gy). Although tumor control was excellent, the rate of new facial weakness and the incidence of trigeminal neuropathy were high. Among 44 VSs treated between 1990 and 1993 the incidence of new facial weakness and facial paresthesias was 21% and 36% respectively. Moreover, the rate of serviceable hearing preservation during this time period was only 25%. In an effort to reduce cranial nerve morbidity, the prescribed tumor margin dose was reduced in 1994 and then again in 1996. At present, we typically prescribe 12 to 13 Gy for patients with serviceable hearing and 13 to 14 Gy for patients with poor pretreatment hearing. We treat using the 50% isodose line, which maintains a high intratumoral dose with steep radiation falloff.
Nonaudiologic Cranial Nerve Morbidity and Tumor Control
Between 1997 and 2000, 84 patients were treated with a mean tumor margin dose of 13 Gy at Mayo Clinic. Over this interval, there were no new cases of facial weakness, trigeminal symptoms occurred in less than 4% of patients, and the short-term serviceable hearing preservation rate improved to 77% compared with earlier treatment protocols. In a prospective observational outcome analysis of 46 patients at our institution, comparing microsurgery with GKS for sporadic VS, we saw facial weakness in 2 patients (4%) and 1 patient (2%) developed a new trigeminal neuralgia after treatment, requiring medication. Sixty-three percent of patients maintained serviceable hearing (pure tone average <50 dB; speech recognition score >50%) at last follow-up. These patients also completed questionnaires evaluating dizziness, tinnitus, headache, and general quality of life. The GKS group had no decline on any component of the health status questionnaire compared with before treatment. Moreover, there was no difference in tinnitus or headache at any time comparing microsurgery with GKS. The final tumor control rate at last follow-up was 96%. As of 2013, more than 700 patients have been treated with GKS for VS in our practice.
A comprehensive review of the literature, including both retrospective and prospective studies, shows similar favorable results, with the incidence of a new facial nerve weakness ranging from 0.0% to 6.1%, hearing preservation rates of 43% to 74%, and radiosurgical failure rates ranging from 1.4% to 10.8%. Hasegawa and colleagues reviewed their long-term outcomes (median follow-up, 12.5 years) in 440 patients who underwent GKS for VS with a median tumor margin dose of 12.8 Gy. The actuarial progression-free survival at 5 and greater than or equal to 10 years was 93% and 92%, respectively. No patient developed treatment failure more than 10 years after treatment. Of 287 patients treated at a dose of less than or equal to 13 Gy, 3 (1%) developed facial palsy, and 3 (1%) developed facial numbness. Collectively, based on our own experience and that in the literature, we counsel patients with sporadic VS that single-fraction GKS with a tumor margin dose of 12 to 13 Gy has a 92% to 95% chance of long-term tumor control with a very low risk of adverse facial or trigeminal nerve outcome.
Hearing Preservation
In 2010 Yang and colleagues published a systematic review of the literature evaluating hearing preservation following GKS for VS. Among 4234 patients with a mean follow-up of 44.4 months (median 35 months) the overall percentage of patients with serviceable hearing was 51%. Similarly, Hasegawa and colleagues reported hearing outcomes at a median follow-up of 38 months among 117 patients treated with a mean marginal dose of 12.4 Gy. These investigators found an actuarial hearing preservation rate of 55% at 3 years and 34% at 8 years.
Long-term audiometric follow-up is particularly relevant for these patients because hearing deterioration seems to follow a protracted course compared with other cranial nerve morbidity. Nevertheless, studies following GKS and reporting median audiometric follow-up data beyond 5 years are rare, and this is further complicated by discrepancies in how hearing outcome is reported. We recently reviewed our long-term outcome in 44 patients with serviceable pretreatment hearing (defined as American Association of Otolaryngology – Head and Neck Surgery class A or B). All patients underwent GKS for sporadic VS treated with 12 or 13 Gy to the 50% isodose line, and the mean duration of audiometric follow-up was 9.3 years. We controlled for age-related decline over this long follow-up by using the nontumor contralateral ear as a control. The Kaplan-Meier estimated rates of serviceable hearing at 1, 3, 5, 7, and 10 years following GKS were 80%, 55%, 48%, 38%, and 23%, respectively.
This observation is important because data from studies with inadequate follow-up may lead to the inaccurate assumption that hearing loss following GKS for VS is exclusively an early phenomenon that levels off after the first several years. Our observations show that hearing loss following GKS continues to decline even 10 years after treatment.
Several factors, such as young patient age, low radiation dose to the cochlea, and small tumor size, have been favorably associated with hearing outcome following GKS. However, the only factor that has been consistently shown to be critical in durable hearing preservation after GKS is the presence of excellent pretreatment hearing.
Dose to the Cochlea in Hearing Preservation
Recently, significant attention has been paid to cochlear radiation dose as a critical and potentially modifiable prognostic factor for hearing preservation following GKS. Several reports have identified thresholds ranging from 3.0 Gy to 5.3 Gy, beyond which there is a correlation with loss of serviceable hearing. However, from a practical standpoint, if the tumor extends out to the fundus of the internal auditory canal, it is often impossible to completely cover the tumor with the prescribed radiation dose while maintaining the cochlear dose below these thresholds. We recently evaluated the relevance of cochlear radiation dose in 59 patients undergoing GKS with serviceable pretreatment hearing using more accurate CT-based volumetric cochlear dose calculations. Following multivariate analysis, again, only excellent pretreatment hearing was most predictive of preserved serviceable hearing despite the cochlear dose. Although reducing the radiation dose to the cochlea may play some role in improving hearing outcomes after GKS, this may necessitate reducing the margin dose or intentionally undertreating the lateral portion of the tumor. However, these strategies may run the risk of compromising long-term tumor control.
Radiographic Follow-up after Gamma Knife Stereotactic Radiosurgery
A positive treatment response after GKS typically shows clearing of the central enhancement of the target ( Fig. 2 ). This clearing can typically be seen approximately 6 months after the GKS procedure. Tumor control following GKS is defined as absence of tumor growth or a reduction in tumor volume after treatment. The imaging changes observed after radiosurgery can be variable and must be interpreted correctly to ensure that the procedure is not prematurely deemed a failure. Tumor expansion after VS radiosurgery is common and rarely denotes a failed procedure, and most patients only require further imaging. Approximately 5% of tumors enlarge relative to their pretreatment tumor size after radiosurgery, but do not show sequential growth on follow-up imaging. As such, additional tumor treatment should be reserved only for patients who show progressive tumor enlargement on serial imaging.
Gamma Knife Stereotactic Radiosurgery for Large Vestibular Schwannomas
Typically, GKS is reserved for tumors smaller than 3 cm in linear posterior fossa diameter. Several studies have documented increased morbidity following GKS for larger tumors, but most of these tumors were treated with higher tumor margin doses (16 Gy) than is now generally used. We recently reviewed our experience treating patients with VSs larger than 2.5 cm in posterior fossa diameter and identified 22 patients treated between 1997 and 2006. The median tumor margin dose was 12 Gy. No patient had symptomatic brainstem compression at the time of GKS. The median treated tumor volume was 9.4 cm 3 (range, 5.3–19.1 cm 3 ) and the median maximum posterior fossa diameter was 2.8 cm (range, 2.5–3.8 cm). The 5-year actuarial rate of tumor control, freedom from new facial neuropathy, and preservation of functional hearing after GKS were 82%, 85%, and 28%, respectively in this subset.
The median GKS treatment volume in the 4 patients who met the criteria for GKS failure was 9.6 cm 3 (range, 8.2–19.1 cm 3 ). One death occurred during follow-up. This 59-year-old man had a rapidly enlarging tumor 7 years after GKS. The patient underwent microsurgical debulking, and pathologic analysis identified the mass as a pleomorphic sarcoma; the patient died 3 months from the time of microsurgery. The other 3 patients who fulfilled the criteria for GKS failure required ventriculoperitoneal shunt placement within 1 year of surgery, and the tumor in each case subsequently failed on radiographic criteria as well. To date, just 1 lesion has required microsurgical removal, and 2 others continue to be radiographically monitored. Corticosteroid therapy for symptomatic radiation-induced changes in the brainstem and/or cerebellum was required in 3 patients (14%) for a median of 12 weeks. However, multivariate analysis did not identify a specific feature predictive of treatment failure for these larger tumors treated with GKS.
Neurofibromatosis Type 2–associated Vestibular Schwannoma and Gamma Knife Stereotactic Radiosurgery
The natural history of neurofibromatosis type 2 (NF2)–associated VS is poorly understood. Previous series have reported variable outcomes, depending on the definition of tumor control and length of follow-up. Rowe and colleagues, from Sheffield, United Kingdom, evaluated 122 NF2-related VSs (96 patients) and estimated that, at 8 years after treatment, 50% of VSs lacked discernible growth. Phi and colleagues reported an actuarial tumor control rate of 66% and a hearing preservation rate of 33% at 5 years.
We recently published our experience on 26 patients with 32 NF2-related VSs who underwent GKS. Eighty-four percent of NF2-associated VSs showed growth arrest or tumor regression after GKS at a median follow-up of 7.6 years for the entire series. A notable association was found between marginal dose and tumor control. Specifically, the median marginal dose for tumors decreasing in size after GKS was 15.5 Gy compared with 13 Gy for tumors that enlarged after treatment, suggesting that NF2-associated VS may be best managed with higher marginal doses than those used for sporadic VS. Despite favorable tumor control, the useful hearing preservation rate was only 25% in the treated ear. These data corroborate earlier studies suggesting that GKS provides tumor control in approximately 80% of NF2-associated VSs at 5 years; however, hearing preservation was seen in less than 40% of treated tumors.
Risk of Malignant Transformation after Gamma Knife Stereotactic Radiosurgery for Neurofibromatosis Type 2–associated Vestibular Schwannoma
Great concern has been raised regarding the possibility of malignant tumor transformation after radiation in patients with NF2 given their lack of a tumor suppressor gene. There have been 9 reported cases of malignant transformation of VS and 29 reported cases of malignant peripheral nerve sheath tumor (MPNST) after radiation therapy for VS (both radiosurgery and conventional radiotherapy). Of the 29 cases of MPNST that occurred after radiation treatment, 11 of 26 patients (40.7%) in whom there was available information had neurofibromatosis (NF). An independent study in patients with NF revealed that, in a population of 1348 patients with NF2, 106 would have received radiosurgery. Malignant transformation occurred in 5 of these cases, corresponding with a risk of 4717 × 10 −5 ; a greater risk than that found in patients who do not have NF2. However, among 14 potential cases of histologically verified intracranial malignancy arising in a stereotactically irradiated field, only 4 patients had NF2. At present, although the available information suggests that there is a risk of malignant transformation caused by radiosurgery, patients should be reassured that the risk is very low.
Cochlear Implantation after Gamma Knife Stereotactic Radiosurgery for Neurofibromatosis Type 2–associated Vestibular Schwannoma
An important and unique potential of using GKS to treat NF2-associated VSs is the ability to maintain an anatomically intact cochlear nerve, thus enabling them to become candidates for cochlear implantation. We have taken advantage of this concept and have placed ipsilateral cochlear implants in 3 patients with NF2 after GKS for VS. The mean age of these patients was 68 years, and they had been deaf in the implanted ear for up to 10 years before cochlear implantation. All patients experienced significant restoration of hearing after cochlear implantation, achieving greater than 85% correction on hearing in noise-sensitive testing. Although microsurgery followed by auditory brainstem implantation remains our preferred method in managing VS in patients with NF2 with tumors greater than 2.5 cm, GKS followed by cochlear implantation remains an effective strategy in select patients with NF2 with smaller growing tumors.
Cystic Tumors
Cystic VS may represent a unique subtype of tumor. Although the mechanism of cyst formation is still unclear, it is postulated that recurrent microbleeding or osmosis-induced expansion of cerebrospinal fluid in trapped arachnoid may be part of the pathogenesis. One important difficulty in evaluating outcomes of these tumors after GKS is trying to decide what percentage of the tumor needs to be cystic for it to be considered a truly cystic tumor. Furthermore, there is also some speculation that radiation can induce delayed cyst formation with radiation-induced repeat microbleeding, increased vascular permeability, or by scarring of arachnoid adhesions in irradiated tumors. Pendl and colleagues noted that 3 of 6 cystic VSs had rapid and significant expansion of the cysts that required surgery after GKS. Delsanti and colleagues reported on 54 cystic VSs, of which 6.4% required additional treatment; approximately 3 times what they report for noncystic tumors. In our experience, and in the small numbers in the literature, there does not seem to be any increased cranial nerve morbidity in treating cystic VS in select patients, and tumor control seems to be favorable. Nonetheless, taking into account a potentially higher failure rate of GKS for cystic VSs, our preference is to manage predominantly cystic tumors with microsurgery.
Repeat Radiosurgery
Once a patient has failed GKS, further tumor-directed therapy is warranted. Overall, we do not favor retreating a patient with GKS after failing GKS as an initial treatment. Although our experience, and that of other surgeons, acknowledges a higher morbidity with microsurgery of VS after failed GKS, that is our recommended strategy in patients who have failed primary GKS. However, there are patients who, because of significant perioperative morbidity, may be candidates for repeat GKS. To date, there are limited data on repeating GKS to the same tumor after treatment failure. Overall, it seems that repeat GKS may be a safe and effective option in select patients for small tumors using current marginal doses (12–13 Gy).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


