Purpose
To characterize and quantify Bruch membrane opening (BMO)-based optic nerve head (ONH) parameters in a large, young and healthy, predominantly white population.
Design
Cross-sectional study and reliability analysis.
Methods
The ONH of 1344 predominantly white subjects were imaged with spectral-domain optical coherence tomography (SD-OCT). A customized script, coded in Matlab, was used to manually segment and measure multiple BMO-based parameters of the ONH. Measurements were compared to those obtained with confocal scanning laser ophthalmoscopy (Heidelberg Retina Tomograph; HRT). Regression analysis was performed to assess the relationship between BMO parameters and other ocular and demographic variables.
Results
Mean BMO disc and neuroretinal rim (NRR) areas ranged from 0.94 to 4.06 mm 2 (mean 1.77 ± 0.38 mm 2 ) and 0.94 to 3.99 mm 2 (mean 1.56 ± 0.31 mm 2 ), respectively. When compared to the equivalent HRT measurements, SD-OCT-derived measures differed significantly for all comparable ONH parameters ( P < .001). The limits of agreement computed from Bland-Altman plots comparing SD-OCT and HRT measurements showed suboptimal agreement between the techniques. Linear regression analysis demonstrated an effect of ethnicity, axial length, and refractive error on BMO-based parameters.
Conclusions
We have quantified BMO-based parameters in a large cohort of young adults using SD-OCT. These data will be informative in constructing normative profiles for clinical and research purposes in glaucoma diagnosis and management.
The optic nerve head (ONH) is the definitive site of ocular damage in glaucoma. To date, the vast majority of studies evaluating normal and disease-related structural variation in the ONH have used 2-dimensional techniques (eg, photo-based planimetry), which provide information about the surface morphology. However, recent work has shown that not only are conventional measurement techniques based on the clinically observable disc margin (DM), which is imprecise, but the underlying anatomy of the ONH also influences the risk of developing glaucoma. The rationale for using the clinically defined DM in ONH assessment is now being questioned because of advances in imaging technology that allow visualization of the subsurface architecture.
One of the primary assumptions employed in studies of ONH morphology is that the clinically perceived contour of the DM represents an actual anatomic structure determined by an underlying “scleral” connective tissue. Recent research has shown that this conventional clinical knowledge may be flawed for two reasons. DM anatomy is complex and involves the Bruch membrane, the end point of the Bruch membrane (Bruch membrane opening [BMO]), and the border tissues of Elschnig—not just the sclera. Consequently, it has been suggested that current neuroretinal rim (NRR) measurements lack a solid anatomic foundation because (1) the observed DM does not represent the anatomic structure that defines the border of the rim tissues as they pass through the ONH, and (2) neural tissue orientation within the ONH is not considered. The new generation of spectral-domain optical coherence tomography (SD-OCT) instrumentation overcomes these measurement limitations by providing anatomically and geometrically accurate BMO-based measurements of the NRR and related structures, thus enabling enhanced detection of open-angle glaucoma.
Few data are currently available regarding BMO-based ONH characteristics in normal populations. The primary aim of this study was to measure and describe these parameters in a large, healthy young adult Australian population. Establishing normative profiles of BMO-based ONH anatomy will ultimately be essential in assisting the clinician in the diagnosis and monitoring of ONH disease, including glaucoma.
Methods
Participants
Participants were identified from the Western Australian Pregnancy Cohort (Raine) Study, a longitudinal prospective study established in Perth, Western Australia in 1989. Prenatal information (eg, diet, exercise, work, and health) and ultrasound imaging were acquired at regular intervals during the pregnancies of 2900 women enrolled until 1991. Subsequently, periodic follow-ups of their children were conducted and various clinical and general health (eg, childhood milestones, height, weight, behavior, and illnesses) data recorded. Ocular health examinations at the 20-year follow-up of the cohort were conducted from 2010 to 2012. All participants had a comprehensive ocular examination that included visual acuity assessment, anterior segment examination, corneal pachymetry, intraocular pressure (IOP) measurement, autorefraction, and a mydriatic optic disc assessment. No participants had signs of glaucoma or other optic neuropathies. ONH imaging was performed using both SD-OCT (Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany) and Heidelberg Retina Tomograph (HRT; Heidelberg Engineering) instrumentation to allow for comparison of anatomic parameters. Autorefraction was conducted following cycloplegia in all subjects using Nidek ARK-510A (NIDEK Co Ltd, Tokyo, Japan). The study adhered to the tenets of the Declaration of Helsinki. Ethics approvals were obtained from the Human Research Ethics Committee of the University of Western Australia. Informed consent was obtained from all subjects after providing them with a detailed explanation of the study.
Spectral-Domain Optical Coherence Technology Imaging and Image Processing
SD-OCT provides 3-dimensional imaging, denser sampling, and enhanced imaging reproducibility compared to traditional time-domain OCT. SD-OCT uses frequency-based image acquisition, enabling 2- to 3-fold improved axial resolution and up to 50-fold increases in scan speed. For all participants, each eye was imaged using the Spectralis OCT with a standardized scanning protocol. A 49-line raster scan at 30 μm (15 × 10 degrees) centered on the optic disc (with tracking on) was performed in each eye, with 100 frames averaged to improve the image quality. Refraction and keratometry data were entered for all participants to correct for interindividual ocular magnification effects. All scans were subjectively assessed for good signal strength and minimal motion artifacts. ONH scans were exported from Heidelberg Eye Explorer as PNG files with the y-scale set as 1:1 μm for analysis. We developed a customized script, coded in Matlab (Mathworks, Inc, Natick, Massachusetts, USA), to enable measurement of BMO-related parameters. The user interface allowed the trained observer (P.S.) to visualize and manually demarcate the positions of the BMO (2 points per B-scan) and optic cup (if defined; 2 points per B-scan) ( Figure 1 ). For each scan the innermost termination of the BMO was defined as the DM, with the cup margin bound by an approximately horizontal line connecting the 2 points and intersecting with the internal limiting membrane (ILM). In this way a reference plane was constructed such that structures above the plane and within the DM were defined as NRR, and those below the plane as optic cup. The BMO-based horizontal rim area (BMO-HRA) was defined as BMO disc area – cup area. The BMO-HRA was set to equal the BMO disc area in eyes wherein the cup was so shallow that the ILM did not cross the BMO reference plane. The BMO and ILM topographic outlines were reconstructed by calculating the position of the marked points on the x-axis of the OCT image as a percentage of the total image width, and mapping them on the corresponding scan line of the fundus image. Once all the required B-scans were marked, the inbuilt Matlab function “roipoly” was used to delineate the BMO disc and cup for area measurements. Linear dimensions were defined as the maximum horizontal and vertical diameters of the BMO disc and cup boundary. Parameters were adjusted for image magnification effects using the OCT x scaling (expressed as μm/pixel) found in the image information tab of Heidelberg Eye Explorer, such that area measurements were in mm 2 (BMO, cup, and NRR) and linear measurements (horizontal and vertical BMO and cup) in mm.
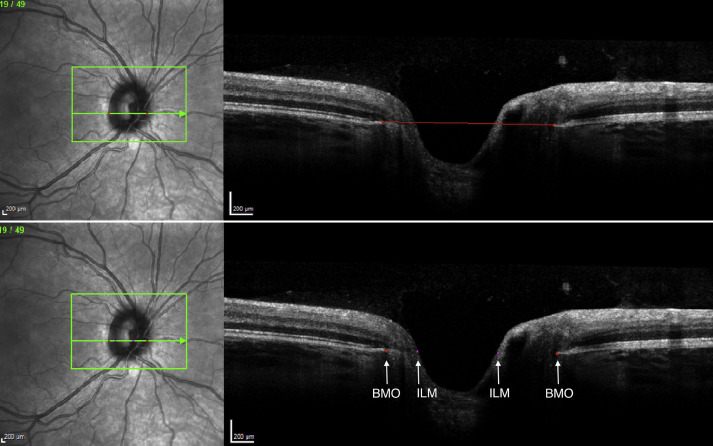
Statistical Analysis
Data management and statistical tests were performed in the R statistical environment (R Development Core Team, 2014; R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were described as the mean ± standard deviation (SD). We used the paired t test and intraclass correlation to compare the similarity of sample distributions between right and left eyes. Linear regression models were used to estimate the effect of different variables (age, sex, ethnicity, height, mean axial length, mean spherical equivalent [SEQ], and BMO-HRA) on BMO area in both univariate and multivariable analyses. Unless otherwise specified, all statistical tests were 2-sided.
The interobserver and intraobserver reproducibility of measuring BMO and NRR areas were evaluated by computing the intraclass correlation coefficient (ICC) from a randomly drawn sample of 50 images manually delineated twice. The agreement between the SD-OCT-derived and HRT-generated ONH area parameters were assessed by Bland-Altman analysis. The Bland-Altman plot allows comparison of SD-OCT and HRT by plotting the difference in the ONH area parameter from the 2 imaging instruments (vertical axis) against the average of the differences (horizontal axis).
Results
Data were available for 1344 subjects (2688 eyes), comprising 652 female (48.5%) and 692 male subjects (51.5%) ( Table 1 ). Participants were aged 18–22 years (mean, 20 years) with the vast majority (85.5%) being white (5.6% Chinese, 8.9% Other). There was no difference in age between male and female subjects (confidence interval [CI] for difference in means [−0.091 to 0.005], 2-sample t test t (1342) = −1.77, P = .076). BMO-based ONH parameter measurements are shown in Table 2 . The interocular ICCs were generally high (>0.80), indicating that parameter measurements between eyes were concordant and utilization of mean values in subsequent analyses were appropriate. Intragrader repeatability was excellent, with ICCs of 0.98 (0.97–0.99, P < .001) and 0.96 (0.92–0.98, P < .001) and the agreement between graders computed as 0.98 (0.96–0.99, P < .001) and 0.94 (0.90–0.97, P < .001) for BMO disc and NRR areas, respectively.
| Variable | Mean (Male, Female) | Standard Deviation (Male, Female) | Range |
|---|---|---|---|
| Sex, n | 692, 652 | ||
| Age (y) | 20.1 (20.1, 20.0) | 0.5 (0.5, 0.4) | 18.3–22.1 |
| Height (cm) | 172.6 (179.0, 165.8) | 9.6 (7.3, 6.5) | 145.0–199.0 |
| Weight (kg) | 73.1 (78.8, 16.4) | 17.1 (67.1, 15.8) | 40.8–176.5 |
| BMI (kg/m 2 ) | 25.8 (25.4, 26.2) | 11.0 (9.3, 12.6) | 15.4–51.7 |
| Axial length (mm) | 23.6 (23.8, 23.4) | 0.9 (0.9, 0.9) | 20.4–27.9 |
| Spherical equivalent (D) | −0.1 (−0.1, −0.2) | 1.6 (1.5, 1.6) | −11.0 to 8.1 |
| Parameter | Right | Left | P Difference in Means a | ICC [95% CI] | SDOCT R/L Mean [Range] | HRT R/L Mean [Range] | P Difference in Means a |
|---|---|---|---|---|---|---|---|
| BMO area (mm 2 ) | 1.77 ± 0.37 | 1.78 ± 0.41 | .162 | 0.85 [0.83–0.87] | 1.77 ± 0.38 [0.94–4.06] | 1.92 ± 0.50 [0.86–4.17] | <.001 |
| Cup area (mm 2 ) | 0.21 ± 0.24 | 0.21 ± 0.26 | .568 | 0.85 [0.84–0.87] | 0.21 ± 0.24 [0.00–1.41] | 0.40 ± 0.31 [0.00–1.73] | <.001 |
| Rim area (mm 2 ) | 1.56 ± 0.31 | 1.57 ± 0.33 | .175 | 0.87 [0.86–0.88] | 1.56 ± 0.31 [0.94–3.99] | 1.51 ± 0.37 [0.76–4.13] | <.001 |
| C/D area ratio | 0.11 ± 0.11 | 0.11 ± 0.11 | .715 | 0.89 [0.88–0.90] | 0.11 ± 0.11 [0.00–0.53] | 0.20 ± 0.11 [0.00–0.57] | <.001 |
| C/D ratio | 0.25 ± 0.20 | 0.25 ± 0.20 | .026 | 0.85 [0.83–0.86] | 0.25 ± 0.19 [0.00–0.70] | 0.28 ± 0.20 [0.00–0.79] | <.001 |
| BMO vertical diameter (mm) | 1.53 ± 0.17 | 1.54 ± 0.18 | .121 | 0.80 [0.78–0.82] | 1.53 ± 0.17 [1.06–2.33] | ||
| BMO horizontal diameter (mm) | 1.53 ± 0.17 | 1.53 ± 0.18 | .148 | 0.83 [0.82–0.85] | 1.53 ± 0.17 [1.14–2.38] | ||
| Cup vertical diameter (mm) | 0.40 ± 0.33 | 0.39 ± 0.34 | .159 | 0.85 [0.83–0.86] | 0.40 ± 0.32 [0.00–1.31] | ||
| Cup horizontal diameter (mm) | 0.46 ± 0.36 | 0.45 ± 0.37 | .170 | 0.85 [0.83–0.86] | 0.46 ± 0.35 [0.00–1.54] | ||
| BMO shape factor (V/H) | 1.00 ± 0.08 | 1.01 ± 0.08 | .002 | 0.64 [0.60–0.67] | 1.01 ± 0.08 [0.77–1.22] | ||
| Cup shape factor (V/H) | 0.85 ± 0.21 | 0.86 ± 0.21 | .912 | 1.33 [0.27–0.39] | 1.86 ± 0.16 [0.38–1.50] |
Mean BMO disc and NRR areas ranged from 0.94 to 4.06 mm 2 (mean, 1.77 ± 0.38 mm 2 ) and 0.94 to 3.99 mm 2 (mean, 1.56 ± 0.31 mm 2 ), respectively. The optic cup was defined in 1993 sets of images (74.1%) with the area ranging from 0.00 to 1.41 mm 2 (mean, 0.21 ± 0.24 mm 2 ). Mean values for cup-to-disc (C/D) ratio and C/D area ratio were noted as 0.25 ± 0.19 and 0.11 ± 0.11, respectively. When compared to the equivalent HRT measurements, SD-OCT-derived measures differed significantly for all 5 ONH parameters ( Table 2 ). Figure 2 shows the Bland-Altman plots comparing SD-OCT and HRT measurements of mean BMO disc (Top), NRR (Middle), and cup (Bottom) area. The limits of agreement (mean difference) were calculated as −0.95 mm 2 to 0.66 mm 2 (−0.14 mm 2 ), −0.72 mm 2 to 0.82 mm 2 (0.05 mm 2 ), and −0.44 mm 2 to 0.06 mm 2 (−0.20 mm 2 ) for the disc, NRR, and cup areas, respectively.
Figure 3 shows the mean area distributions (in 0.25 mm 2 increments) for BMO disc area (Top), NRR area (Middle), and cup area (Bottom), grouped by sex. All distributions demonstrated some degree of positive skewness, being most marked for mean cup area. The Shapiro-Wilks test indicated each distribution departed from normality ( P < .001). Indeed, while this may be partly attributable to our large sample size, in this case the central limit theorem works to advantage in making the parametric tests we have employed in the current study ( t test and linear regression) more robust to violations of this assumption. Regression residuals (eg, Q-Q plots) were also assessed for normality.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


