17 Sleep Disturbance in the Geriatric Population
 Introduction
Introduction
With an aging population and obesity epidemic, interest in snoring and obstructive sleep apnea has increased substantially over the past 10 to 15 years. Obstructive sleep apnea (OSA) is a prevalent disorder characterized by intermittent cessation of airflow during sleep that results in excessive daytime sleepiness. Sleep disturbance prevalence is ~ 60% in elderly adults and affects nearly 67% of institutionalized elderly adults.1,2
The most common presenting symptoms, and the reason many patients seek treatment, are habitual snoring, bed partner/family complaints, and excessive daytime somnolence (EDS). Snoring affects 40% of men and 20% of women3 and frequently is the first sign of more serious underlying OSA. Geriatric patients also more frequently complain of insomnia and poor sleep quality.
Other associated symptoms that may suggest a sleep disorder include witnessed apnea, nighttime arousal, daytime sleepiness or fatigue, motor vehicle accidents, poor sleep quality, morning headaches, and mood disorders or memory concerns. Due to this wide array of symptoms and manifestations, sleep disorder management requires a multidisciplinary approach. Sleep medicine is the new medical discipline dedicated to treating this condition. Sleep medicine teams frequently involve geriatricians, pulmonologists, otolaryngologists, neurologists, psychiatrists, oral/maxillofacial surgeons, dentists, behavioral psychologists, and nutritionists working together to care for patients with sleep disorders.
This chapter describes the evaluation and management of sleep disorders in elderly patients.
 Classification of Obstructive Sleep-Related Breathing Disorders
Classification of Obstructive Sleep-Related Breathing Disorders
Sleep-related breathing disorders range in severity from airway narrowing with increased airway effort due to upper airway resistance, to partial (hypopnea) or complete (apnea) airway collapse with respiratory effort (Tables 17.1 and 17.2).4
Snoring is generated by primarily pharyngeal soft tissue vibration. The sound is typically louder on inspiration when negative pressure and increased tissue collapse are more likely. It can occur without hypopnea or apnea; however, nightly snorers have at least a 50% probability of having sleep apnea on overnight polysomnography (PSG). Therefore testing is recommended in nightly snorers, especially if there is associated daytime sleepiness or fatigue. If testing is performed and the overall apnea-hypopnea index (AHI) is fewer than five events per hour, the diagnosis is primary snoring. Patients with primary snoring rarely have EDS and feel well rested after a full night of sleep. Patients who have fatigue and snoring but PSG testing negative for OSA should be screened for sleep deprivation, restless leg syndrome, hypothyroidism, depression, anemia (especially iron deficiency), Addison disease, collagen vascular diseases, and medication side effects. Polypharmacy is especially prevalent in the geriatric population, and each patients’ medication list should be monitored and minimized at every opportunity and clinical encounter.
Upper airway resistance syndrome (UARS) refers to patients who do not meet PSG criteria for OSA but have daytime somnolence or other debilitating somatic complaints.5 In this condition, the PSG demonstrates progressively increased respiratory effort (abnormally negative intrathoracic pressure, with snoring and increased diaphragmatic electromyographic [EMG] activity) terminating in an arousal, commonly referred to as respiratory effort–related arousals (RERAs).6
Table 17.1 Respiratory event definitions and types
Respiratory event | Definition |
Apnea | A cessation of airflow for at least 10 seconds |
Hypopnea | A reduction in airflow (> 30%) at least 10 seconds with > 4% oxyhemoglobin desaturation or electroencephalogram (EEG) arousal |
Respiratory effort–related arousal (RERA) | Sequence of breaths for at least 10 seconds with increasing respiratory effort of flattening of the nasal pressure waveform leading to an arousal from sleep when the sequence of breaths does not meet criteria for an apnea or hypopnea |
Obstructive | Continued thoracoabdominal effort in the setting of partial or complete airflow cessation |
Central | The lack of thoracoabdominal effort in the setting of partial or complete airflow cessation |
Mixed | A respiratory event with both obstructive and central features; generally begin as central events and end with thoracoabdominal effort without airflow |
Used with permission from Kushida CA, Littner MR, Morgenthaler T, et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep 2005;28(4):499–521. | |
The diagnosis of OSA is established if the patient has both an AHI of ≥ 5 with evidence of excessive daytime sleepiness or fatigue. Each episode must last a minimum of 10 seconds and be associated with reduction in oxygen saturation of 3 to 4% and terminated by brief, often unconscious, arousals from sleep. The United States Medicare guidelines diagnose OSA if the AHI is > 15 or an AHI of 5 with two medical comorbidities and/or excessive daytime sleepiness. The American Academy of Sleep Medicine defines mild OSA as AHI between 5 and 15, moderate between 15 and 30, and severe if > 30.7 Practitioners should be cognizant that variations in published standards of hypopnea definition may result in differences in AHI.8
 Epidemiology of OSA in Older Adults
Epidemiology of OSA in Older Adults
Population-based studies estimate that 2 to 3% of female adults and 4 to 5% of male adults have OSA.9,10 OSA, however, is even more prevalent in elderly patients. In a large study of randomly selected community-dwelling elderly, 65 to 95 years of age, Ancoli-Israel et al reported prevalence rates of 62% for an AHI > 10, 44% for an AHI > 20, and 24% for an AHI > 40.2 She found that with 18 year follow-up in this population, the AHI remained relatively stable and changed only with associated changes in body mass index (BMI).11 The Sleep Heart Health Study showed similar findings of increased prevalence of OSA in elderly patients compared with the general population.12 Patients age 60 to 69 had 51% prevalence of mild to moderate OSA, with increases minimally (54% age 70–79; 56% age 80–89) in prevalence with increasing age. Disease severity was not observed to increase significantly with age.
Table 17.2 Indices of sleep disordered breathing
Indices | Definition |
Apnea index | Number of apneas per hour of total sleep time |
Hypopnea index | Number of hypopneas per hour of total sleep time |
Apnea-hypopnea index (AHI) | Number of apneas and hypopneas per hour of total sleep time |
Respiratory effort–related arousals (RERAs) index | Number of RERAs per hour of sleep time |
Respiratory disturbance index (RDI) | Number of apneas, hypopneas, and RERAs per hour of total sleep time |
Central apnea index | Number of central apneas per hour of total sleep time |
Mixed apnea index | Number of mixed apneas per hour of total sleep time |
Used with permission from Kushida CA, Littner MR, Morgenthaler T, et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep 2005;28(4):499–521. | |
Other studies estimate that the rate of mild OSA is 50 to 60% in older men and 30 to 40% in older women.13 This may be due to loss of muscle tone associated with physiological aging.14 Additional theories include decreased hormone levels15 and changes in the ratio of fat to lean body mass.13 Menopausal status appears to be a major determinant in the rate of OSA in females. In the large population-based Wisconsin Sleep Cohort Study, postmenopausal women were found to be 3.5 times more likely than premenopausal women to have AHI > 15.15 OSA may be underdiagnosed in women due to a lower index of suspicion in practitioners, and atypical symptoms reported by female patients. Insomnia, heart palpitations, and ankle edema were more common complaints in women with OSA.16
Although the prevalence of OSA appears to increase steadily with age (and weight), the overall prevalence of the disorder appears to level off after the age of 65.17 The reason for this may be explained in one of three ways: (1) the incidence of new cases decreases after age 65, (2) the mortality rate of OSA increases after age 65, or (3) OSA remits with older age. Because there is little evidence to support death caused directly by OSA (except in extreme cases) or spontaneous remission, a reduction in incidence after age 65 is the currently favored explanation13; however, further investigation is needed to clarify this issue.
 Sleep Patterns in Older Adults
Sleep Patterns in Older Adults
Changes in sleep duration and architecture occur as a normal part of the aging process.18 The duration and need for sleep appear to lessen with age. The average 70-year-old sleeps only 6 hours per night but may make up for loss of nocturnal sleep with daytime naps of 1 to 2 hours.19 In addition, many older adults have increased difficulty falling asleep once in bed (increased sleep latency), and have greater difficulty staying asleep (decreased sleep efficiency).20 The number of comorbid medical conditions that have an impact on sleep increases with age and includes depression, arthritis, gastroesophageal reflux, prostate hypertrophy, and renal and pulmonary disorders.21 In addition, older adults are more likely to take medications such as diuretics that cause nocturia. As a result, up to 40% of older individuals complain of sleep disturbance and undesired daytime fatigue.22 Additionally, the increased sleep latency, decreased sleep efficiency, and lack of awareness regarding the need for less sleep all combine to create greater complaints of insomnia in this population.
The challenge for the physician presented with complaints of sleep disturbance in older individuals is to determine the degree to which symptoms are related to normal age-related changes in sleep pattern versus an underlying medical disorder or primary sleep disorder. A thorough review of the patient’s past medical history, medications, and alcohol and stimulant (caffeine, nicotine) use is required. Improving the management of comorbid conditions (i.e., BPH, arthritis, gastroesophageal reflux disease [GERD], depression) will often result in sleep improvement. A sleep diary to determine times of sleep can also be helpful because many older adults may suffer from advanced sleep phase circadian disorder (early evening sleep; early morning awakening). Advanced sleep phase disorder can often be effectively managed by encouraging more sunlight exposure and walks in the late afternoon as well as melatonin therapy during early morning hours.
Sleep is divided into rapid eye movement (REM) and non–rapid eye movement (NREM) stages. About 80% of normal sleep is spent in NREM and is staged according to electroencephalographic measures. Stage N1, formerly stage 1, is a transition from wakefulness to sleep with a mixed voltage pattern with waves of 3 to 7 cycles per second. Most patients feel as though they are awake during this stage. Stage N2, formerly stage 2, is identified by spindles and K complexes and marks the onset of true sleep. Stage N3, formerly stage 3 and 4, is also referred to as slow wave sleep and is distinguished by delta waves. Stage R is characterized by low-frequency mixed amplitude waves and rapid eye movements. REM typically alternates with NREM periods in 90 minute cycles and increases in length as the night goes on. Early-onset REM (< 8 min) is a marker of narcolepsy, which is formally diagnosed with multiple sleep latency testing (MSLT).
Time spent in REM sleep decreases with age.23 A large meta-analysis of 65 overnight studies representing 3,577 subjects across the entire age spectrum reported that with age, the percentage time in REM sleep decreased, whereas the percentages of light sleep (N1 and N2) increased.24 Muscle tone is also decreased during REM sleep, so many patients with OSA have worsening of their upper airway collapse and AHI during REM. This fact may also help explain why elderly patients show no progression or worsening in their OSA with time. Bixler et al studied over 4,000 patients age 22 to 100 and found that the prevalence of OSA increases with age but the severity decreases.25 For this reason, OSA severity based on AHI is often adjusted in elderly populations such that sleep-disordered breathing is considered present when AHI is > 10 to 15 in patients older than 60.
 Pathophysiology
Pathophysiology
OSA is caused by physical obstruction of the pharyngeal airway during sleep. Most patients with OSA have worsening of symptoms during REM sleep, when muscle paralysis is physiological. Obesity, soft tissue (soft palate, tonsil, tongue base) hypertrophy, and craniofacial structure (retrognathia) contribute to one’s propensity to collapse by increasing the extraluminal tissue pressure surrounding the upper airway. Obstruction occurs when the pharyngeal dilator muscles are unable to overcome the negative pressure in the upper airway. The four primary sites of obstruction are the nasal airway, palate, tongue base, and hypopharynx.
Nasal obstruction may worsen OSA or contribute to poor continuous positive airway pressure (CPAP) tolerance, but it is rarely the sole cause of OSA. Patients often report more restful sleep and improved symptoms of snoring and fatigue after surgery to improve nasal airflow, but objective measures of OSA severity (i.e., AHI and lowest oxygen saturation) rarely change.26 This may be due to lessened open-mouth breathing during sleep, which increases upper airway collapsibility and may decrease the efficacy of pharyngeal dilator muscles.27
Obesity is a major risk factor for OSA. Increased fat deposition in the soft tissues of the neck narrows the upper airway and may offset the ability of the pharyngeal dilators to maintain airway patency.28
Neuromuscular tone can decrease with age and certainly contributes to the patency of the upper airway. The genioglossus muscle is considered the most important muscle in maintaining pharyngeal airway patency in OSA. Klawe and Tafil-Klawe showed that genioglossus muscle activity increases in response to hypoxia, but there is a decreased genioglossus muscle response to hypoxia in older cohorts.29 Surgery to address tone and tension in this region has been developed,30,31 and clinical trials are ongoing for hypoglossal nerve stimulation therapy.32
 Clinical Sequelae of Untreated OSA
Clinical Sequelae of Untreated OSA
Untreated OSA has been associated with reduced quality of life and serious health conditions. In a retrospective review, He et al found that untreated OSA patients with an apnea index (AI) > 20 had a statistically significant increase in mortality compared with patients whose AI was < 20.33 The authors also found that untreated patients with moderate to severe OSA had a 63% probability of surviving 8 years, compared with 96% in people with mild or absent OSA. Fatal and nonfatal motor vehicle accidents increase 2.5-fold in patients with untreated OSA.34
Reduced Quality of Life
Daytime sleepiness caused by OSA often results in decreased energy, loss of concentration, poor job performance, and reduced social interaction. Executive function, attention, and working memory have all been shown to improve with CPAP treatment.35 In addition, the loud snoring that often accompanies OSA may result in poor sleep for the sufferer’s bed partner. Quality of life has been shown to be improved in both the treated individuals and their bed partners.36
An AHI > 5 is associated with concentration difficulty but not memory tasks on self-assessment exams.37 OSA patients demonstrated significant improvement in daytime sleepiness38 and in numerous quality of life parameters, including physical functioning, social functioning, vitality, and general health perception after treatment.39 The relationship between sleepiness (defined by the Epworth Sleepiness Scale [ESS]) and severity of OSA (defined by AHI) is not well defined, especially in older populations. Many elderly people have an AHI > 5 but report minimal or no daytime sleepiness.40 Unless the AHI is > 15 or there is significant oxygen desaturations (< 90%) and hypertension or coronary artery disease, most would not argue for invasive treatment in this subset of nonsleepy elderly patients.
Cardiovascular Morbidity and Mortality
Evidence supports an increase in cardiovascular mortality 5 years after diagnosis in untreated patients with severe OSA compared with treated patients.33,41 Another study found that men under age 60 with snoring and EDS were twice as likely to die over a 10-year study period compared with subjects without snoring or snoring without sleepiness.42 Two large prospective studies suggest that untreated, loud, habitual snorers have a 30 to 40% greater risk of myocardial infarction or stroke relative to nonsnorers.43,44 The risk of sudden cardiac death is significantly increased in patients with OSA and nocturnal hypoxemia, with increased risk in patients with more severe disease.45
The foregoing information is contradicted by a recent study by Johansson et al.46 In community-dwelling elderly age 71 to 87, OSA does not appear to be associated with cardiovascular disease (CVD) or mortality, but central sleep apnea might be a pathological marker of CVD and impaired systolic function associated with higher mortality. One limitation of this study was use of in-home sleep studies and a limited sample size (331 patients).
Hypertension
It has been hypothesized that systemic arterial hypertension is the cause of the increased cardiovascular morbidity and mortality observed in patients with sleep-disordered breathing.47 Laboratory evidence has demonstrated that sustained arterial hypertension can be induced in animal models subjected to intermittent airway occlusion during sleep.48 The current epidemiological evidence shows a strong and consistent association between OSA and hypertension. Four large population-based cross-sectional studies found that the odds of hypertension were 1.4 to 2.5 times greater in patients with an AHI > 5 compared with controls.49–51 Finally, prospective analysis of the Wisconsin Sleep Cohort study found that even minimal elevation in AHI scores was associated with a 42% increased risk of developing hypertension over a 4 year period.52
Level I evidence shows that treatment of OSA with CPAP has been reported to lower blood pressure (mean, systolic and diastolic) by ~ 10 mm Hg.53 In this study, 60 consecutive patients with moderate to severe OSA were randomized to either effective or subtherapeutic nasal CPAP for 9 weeks. Apneas and hypopneas were reduced by 95% in the treatment group and 50% in the subtherapeutic group. The blood pressure reduction demonstrated in the therapeutic CPAP-treated group would be predicted to reduce coronary heart disease event risk by 37% and stroke risk by 56%.
Motor Vehicle Accidents
Epidemiological and laboratory evidence both suggest that patients with OSA are at greater risk for motor vehicle accidents (MVAs). Patients with an AHI > 15 were 7.3 times more likely to have had multiple MVAs in the 5 years before their study, compared with those with lower or no apnea.54 Hospitalized MVA victims were found to be 6.3 times more likely to have an AHI > 5 compared with community controls.9,54,55 In addition, patients with severe OSA performed significantly worse on a driving simulator than controls without OSA.56
Insulin Resistance
Untreated OSA has been shown to be an independent risk factor for insulin resistance.57 Thus OSA may contribute to the development of diabetes and metabolic syndrome (the term used to describe concomitant obesity, insulin resistance, hypertension, and dislipidemia). Assoumou et al reviewed a cohort of 806 elderly healthy patients (age 68) and found that 9.8% had metabolic syndrome, and 55.9% had OSA (diagnosed on home sleep study, positive for AHI > 15).58 Of the subset with OSA, 12.5% had metabolic syndrome. The oxygen desaturation levels and AHI were found to be significantly higher in the subset of patients with metabolic syndrome. Hypoxia and the oxyhemoglobin desaturation index appeared to be the most important factors explaining the association between metabolic syndrome (specifically hyperglycemia and hypertension) and OSA.
 Diagnosis of Sleep Disorders in Elderly Patients
Diagnosis of Sleep Disorders in Elderly Patients
A high index of suspicion should accompany evaluation of geriatric patients with obesity (BMI > 30), loud nightly snoring, restless sleep, and daytime fatigue or somnolence. In a large review by Young et al, 70% of adult patients with OSA were found to be obese.59 Patients who have symptoms without obesity and patients with a positive family history of OSA should also warrant investigation with a detailed sleep history and physical examination. The ESS is an easy to administer, widely used scale to risk stratify patients with daytime sleepiness at risk for OSA (Fig. 17.1). OSA is much more likely in patients with an ESS > 10.60
A thorough sleep history (Table 17.3) helps identify patients at risk for primary sleep disorders seen most commonly in older adults: OSA, restless leg syndrome, insomnia, and sleep maintenance insomnia. Sleep maintenance insomnia is characterized by early morning awakening and is highly associated with alcohol abuse or depression in older individuals.61 Patients suspected of a primary sleep disorder should undergo overnight PSG to establish the presence and severity of a sleep disorder.
Table 17.3 Elements of sleep history
Time in bed |
Time to sleep |
Number of awakenings |
Time awake |
Food consumption and timing of meals |
Amount and time of alcohol consumption |
Presence of snoring, witnessed apnea, gasps, or choking spells |
Presence of involuntary leg motion or jerking |
Level of daytime sleepiness or fatigue (Epworth Sleepiness Scale score) |
Time, duration, and number of daytime naps |
Medications, including over the counter and herbal |
Waking with a headache |
Three screening questions are helpful in determining the likelihood of sleep disorder in elderly patients1:
1. Is your sleep restorative, or restful?
2. Does lack of sleep or fatigue intrude in your daily activities?
3. Does your bed partner or caregiver report any unusual sleep behavior such as breathing pauses, snoring, gasping, or abnormal movements?
Medication review is especially important in the geriatric patient due to more pronounced side effects in this population. Hypnotic medications may diminish in effectiveness with time, and medications prescribed for insomnia, such as benzodiazepines, often worsen OSA. Don’t forget to ask about over-the-counter medications and herbal supplements! Melatonin is used by many to promote sleep, but it should be used with caution in patients on anticoagulant medications such as warfarin. Chamomile tea is also a “relaxing” supplement, but it should not be used in patients with ragweed allergy, because the plants are related. Valerian is reportedly helpful in anxiety and helping patients fall asleep, but it should not be used with other sleep aides or alcohol. One final herbal sleep aide is kava. It is metabolized in the liver and should not be used in patients who are on medications such as statin drugs. Kava use has been tied to severe liver failure, especially when taken with alcohol or other medications metabolized in the liver.
OSA is more prevalent in patients with hypertension, coronary artery disease, congestive heart failure, history of prior stroke, or diabetes as well as in those who are obese or postmenopausal. Screening via PSG is encouraged in this subset of patients with signs or symptoms of sleep disturbance.4
Physical exam findings such as BMI, blood pressure measurement, and neck circumference should be recorded. Collar size > 17 inches in men and 15 in women is associated with OSA.12 Unfortunately, the Sleep Heart Health Study showed that SDB is poorly predicted by obesity, neck circumference, and reports of witnessed apnea in elderly patients.12 This finding may be due to the high prevalence of disease in elderly patients. Maxilla and mandibular position, nasal deformity (septal deviation or valve collapse), turbinate size and response to decongestant spray, nasal polyps, tongue position, tonsil size, palate length/shape, and hyoid position should be evaluated. Patients with Angle class II malocclusion may benefit most from dental devices or maxillomandibular advancement (MMA) surgery.
Fujita developed a classification system for patterns of obstruction by anatomical location as follows: type I, collapse in the retropalatal region only; type II, collapse in the retropalatal and retrolingual regions; type III, collapse in the retrolingual region only.62 Classically described surgery to address retropalatal collapse (i.e., uvulopalatopharyngoplasty [UPPP]) often fails in poorly selected patients due to multilevel airway obstruction in most patients with moderate or severe OSA.63
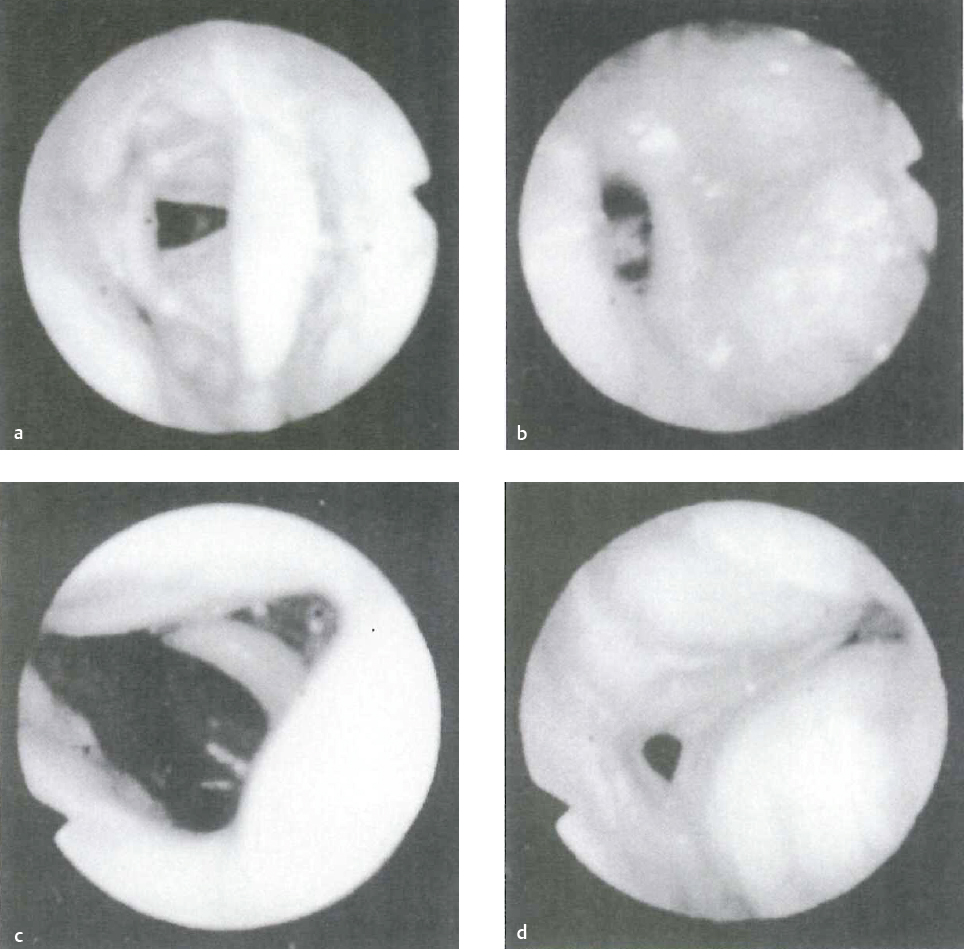
Otolaryngologists frequently use flexible fiberoptic nasopharyngoscopy (preferably with supine positioning) with or without sedation or sleep simulation to evaluate for upper airway collapse (Fig. 17.2). The Müller maneuver was described to simulate upper airway collapse in an office setting by inhaling against a closed glottis with the nose and mouth closed. This results in negative pharyngeal pressure and allows the physician to identify the site(s) of collapse. Aboussouan et al found that using the Müller maneuver to guide the decision on UPPP resulted in an AHI reduction by 50% in 78% of patients who had retropalatal collapse, compared with only 36% who had multilevel obstruction.64 The biggest limitation with the Müller maneuver is it fails to demonstrate lower levels of airway obstruction (retrolingual, hypopharyngeal), and it is performed on awake and alert patients with normal muscle tone. It may be more difficult to obtain reliable results with this maneuver in elderly patients with early cognitive impairment.
Office-based supine airway evaluation with fiberoptic scopes may provide more insight into the level of airway collapse than seated erect patient position.65 Sleep MRI has also been used with some success in predicting level of airway obstruction in snoring patients.66
Drug-induced sleep videoendoscopy has been described to guide more effective surgical intervention.67 Sleep is induced in a monitored setting via pharmacological means, and fiberoptic nasopharyngoscopy is performed to better identify the site(s) of airway collapse. This idea seems to hold the most promise for surgical planning, but the downside is the expense and risk associated with sedation. The levels of obstruction can be treated with surgery to relieve obstruction in each area for patients that can’t tolerate or refuse CPAP. The modified Mallampati score (i.e., I–IV without tongue extrusion) (Fig. 17.3) has been shown to correlate poorly with airway level obstruction on sleep endoscopy in patients with OSA.68 It does predict difficult intubation and surgical success (along with tonsil hypertrophy) using UPPP alone to treat OSA (more success with palate position I or II and tonsil hypertrophy).
Nocturnal PSG is the gold standard for diagnosis of OSA, but more and more patients are being screened with home studies. Home studies are most appropriate in patients with suspected mild disease or without complex medical comorbidities. Home sleep studies may miss other causes of excessive daytime sleepiness such as UARS or periodic leg movement during sleep disorder (PLMD). A full diagnostic study includes electroencephalogram, electro-oculogram, submental electromyogram, electrocardiogram, nasal and oral airflow monitors, thoracoabdominal effort, anterior tibial electromyogram, body position monitors, tracheal microphone, and pulse oximetry. Optional additions (14 channel) are end-tidal carbon dioxide monitor, esophageal pressure monitor, and nasal CPAP or BiPAP. These studies are attended by and analyzed by a trained technologist and interpreted by a physician . Split night studies, in which patients demonstrate OSA early in the evening, allow for pressure titration of CPAP or BiPAP for home use.
The anesthesia perioperative assessment literature has popularized the mnemonic questionnaire “STOP-Bang” as a screen for OSA in previously undiagnosed patients. Patients and/or significant others are asked if the patient has: loud Snoring, daytime Tiredness, Observed apneas, high blood Pressure, elevated BMI > 35, Age > 50, Neck circumference > 40 cm, and male Gender. Patients are high risk for OSA if 3 or more are positive and low risk if fewer than 3.8 The sensitivity of this test was found to be 93% for AHI 15 and 100% for AHI 30. The Society of Ambulatory Anesthesia recommends screening all adult patients with this questionnaire prior to ambulatory surgery.69