6
Selection of Cochlear Implant Candidates
Teresa A. Zwolan
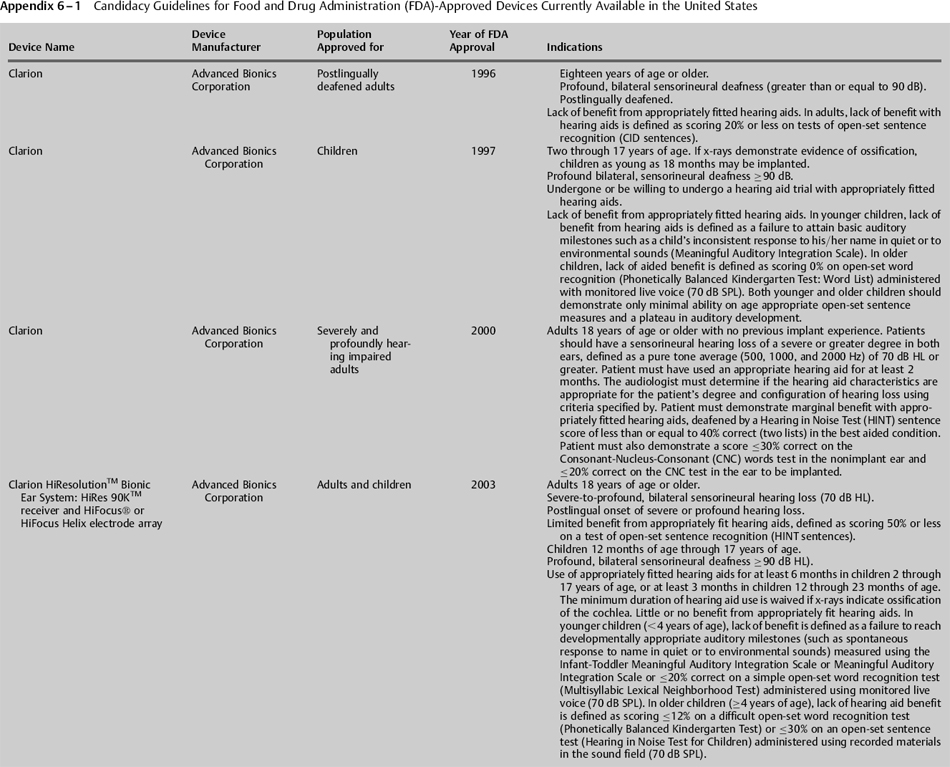
In the United States, the Food and Drug Administration (FDA), a consumer protection and health agency, has historically had great influence on cochlear implant candidacy. This agency oversees the selling, distribution, and marketing of drugs, medical devices, and other products. Importantly, the FDA determines if the specific wording used in device labeling, including information regarding indications for its use, is appropriate. Prior to receiving approval, device manufacturers must provide the FDA with details of their product, including a description of its function, results of clinical trials on specific groups of patients, information on adverse effects, and details on the materials that comprise the product. The FDA reviews the information and determines if it considers the product to be safe and effective for widespread use. The information provided by the FDA is a guideline for professionals. Thus, the determination of implant candidacy is ultimately based on the best knowledge and judgment of the managing physician. This may result in implantation of patients who do not meet candidacy requirements specified on the product’s label. Such “off-label” uses of cochlear implants are infrequent, as many insurance companies may refuse payment if a device is not indicated for the recipient based on FDA guidelines. According to the FDA’s Web site, “If physicians use a product for an indication not in the approved labeling, they have the responsibility to be well informed about the product, to base its use on firm scientific rationale and on sound medical evidence, and to maintain records of the product’s use and effects.” Information regarding past and present candidacy guidelines for cochlear implant devices available in the United States is provided in Appendix 6–1.
Numerous clinical trials have been conducted by the FDA since cochlear implants were first introduced, and numerous supplements have been submitted to the FDA as these devices have undergone technological improvements. Changes in candidacy have primarily included implanting persons with increasing amounts of residual hearing, implanting persons with increasing amounts of preoperative open-set speech perception skills, implanting children at younger ages, and implanting greater numbers of persons with abnormal cochleae. The primary reason that selection criteria have changed is that patients with implants are obtaining increasing amounts of open-set speech recognition with the available devices. Although this increased performance is largely due to the technological advancements that have occurred in the field, it may also be due, at least in part, to the fact that patients with greater amounts of residual hearing and greater amounts of residual speech recognition skills are receiving cochlear implants.
♦ Pre- and Postoperative Assessment and Evaluation
The preoperative evaluation is an extremely important part of the implant process. The primary purpose of the preoperative evaluation is to determine if the patient is medically and audiologically suitable for a cochlear implant. Additionally, information obtained during the preimplant evaluation process can be compared with postoperative performance to evaluate patient progress and to evaluate device efficacy. Test procedures commonly included in the preoperative test battery for determining implant candidacy are described in the following sections.
Medical Evaluation
During the preoperative medical evaluation, the physician obtains a complete medical history and performs a physical examination. The surgeon attempts to identify the cause of the hearing loss if it is not known, determines if treatment options other than a cochlear implant are more suitable for the patient, and determines if the cochlear implant candidate’s general health is suitable for implantation. Additional information regarding surgical decisions that may affect candidacy for a cochlear implant may be found in other chapters in this book. Postoperatively, patients should be seen biannually by their implant surgeon for routine evaluations. They should also be seen by the surgeon if any otologic problems arise or if problems with the electrode array are noted by the audiologist.
Cochlear Imaging
Computed tomography (CT) or magnetic resonance imaging (MRI) of the temporal bone are routinely performed as part of the preoperative evaluation process and provide an image of the structure of the cochlea and help identify any cochlear anomalies. The results of such testing may be used to determine the most appropriate ear for implantation. Although severe anomalies and/or cochlear ossification may affect insertion depth, placement of the electrode array, or type of device selected, they do not necessarily preclude the patient from implantation (Balkany et al, 1996; Eisenman et al, 2001; Tucci et al, 1995). Postoperatively, imaging studies may be used to evaluate device placement and to determine if changes have occurred regarding the location of the electrode array within the cochlea. Additional information regarding imaging and its role in patient selection and evaluation may be found in Chapter 7.
Audiological Evaluation
The primary purpose of the preoperative audiological evaluation is to determine the type and severity of hearing loss. Typically, this evaluation will include determination of unaided air and bone conduction thresholds, unaided speech discrimination, speech reception threshold (SRT) (if possible), speech detection threshold (SDT), otoacoustic emissions (OAEs), and immittance testing to include tympanometry and acoustic reflexes.
Candidacy Guidelines Regarding Audiometric Test Results
In the early clinical trials, only persons who demonstrated a bilateral profound sensorineural hearing loss [pure tone average (PTA) greater than or equal to 90 dB HL] were considered to be candidates for a cochlear implant. Past and present candidacy guidelines for cochlear implant devices available in the United States are provided in Appendix 6–1. Present guidelines are more lenient than guidelines used in the past and allow for persons with slightly better hearing (particularly in the low frequencies) to be considered for a cochlear implant. Additionally, these indications vary slightly among the currently available devices. For example, guidelines for the Nucleus Freedom with Contour Advance indicate that postlinguistically deafened adults who are candidates for the implant typically have “moderate to profound hearing loss in the low frequencies and profound (≥90 dB HL) hearing loss in the mid-to-high speech frequencies.” Guidelines for the Clarion HiResolution Bionic Ear System (Advanced Bionics; Valencia, CA) indicate that adults should have “severe to profound, bilateral sensorineural hearing deafness (≥70 dB HL),” while indications for the Med-El Pulsar CI100 (Innsbruck, Austria) states “a pure tone average of 70 dB or greater at 500 Hz, 1000 Hz, and 2000 Hz.”
Guidelines for children are often stricter than those for adults and may vary depending on the age of the child. For example, recommended guidelines for the Nucleus Freedom implant with Contour Advance indicate that children who are less than 24 months of age should demonstrate a bilateral profound hearing loss, whereas children 24 months of age or older only need to demonstrate a severe to profound hearing loss bilaterally. Indications for children for the HiResolution Bionic Ear System and for the Med-El Pulsar CI100 indicate that all children 12 months of age through 17 years should demonstrate a profound, bilateral sensorineural hearing loss.
Electrophysiologic Testing
Several implant centers include electrophysiologic tests in their preoperative test battery. Tests such as the auditory brainstem response (ABR) verify audiometric test results, help identify patients with auditory dyssynchrony, and help rule out the possibility of functional deafness. Such testing is particularly important when testing young children.
In some clinics, the electrophysiologic measure of electric auditory brainstem response (EABR) testing (Kileny & Zwolan, 2004; Kileny et al, 1994) is routinely performed with children who are less than 2 years of age, when a child presents with cochlear malformations, or when the preferred ear for implantation demonstrates no residual hearing. Such testing can be helpful to verify electrical stimulability of the auditory system, and is particularly useful when questions arise regarding the presence or stimulability of the VIIIth nerve in the ear to be implanted.
Hearing Aid Evaluation
The primary purpose of the preoperative hearing aid evaluation (HAE) is to evaluate the patient’s performance with appropriate amplification. This procedure has two primary components: evaluation of the patient’s detection skills and evaluation of the patient’s aided speech perception skills. The first step in the HAE should include an evaluation of the patient’s personal amplification to determine if the aid meets manufacturer’s specifications. Second, the suitability of the device should be evaluated. Some patients’ hearing loss may have progressed since the hearing aid was originally obtained, making it inappropriate for their current hearing loss. Additionally, improvements may have occurred in hearing aid technology that may increase the benefit the patient is able to receive from a different type of amplification. This can be particularly true if the patient has never experienced digital hearing aid technology. If the patient’s hearing aid does not meet manufacturer’s specifications for performance, or if the aid is not suitable for the patient’s degree and severity of loss, an appropriate hearing aid must be fitted to the patient prior to performing the preoperative evaluation.
Typically, the HAE includes determination of aided warble tone sound-field detection thresholds at 250, 500, 1000, 2000, 3000, and 4000 Hz; determination of the aided most comfortable loudness (MCL) level, uncomfortable loudness level (ULCL), aided SDT, and aided SRT; and speech perception testing. These procedures should be performed with each ear aided separately as well as in binaural aided condition.
Once aided responses have been obtained, it is important to compare these responses to those expected with a cochlear implant. On average, patients with cochlear implants demonstrate sound-field warble tone thresholds that fall between 25 and 40 dB HL for the test frequencies of 250 to 4000 Hz. The aided detection thresholds obtained by a patient with hearing aids can be compared with the levels obtained by patients with implants to determine if the patient’s detection skills will likely improve with a cochlear implant. When making such a comparison, it is important to inform patients that aided detection thresholds represent simple detection and provide no information regarding the discriminability of the sounds presented at the various test frequencies.
Postoperatively, it is important to evaluate the implant users’ ability to detect warble tones in a sound field. Such testing provides information regarding their detection skills and verifies that a particular program is appropriate. Additionally, sound field thresholds obtained with the cochlear implant can identify problem areas and can be used to guide programming of the speech processor.
Candidacy Guidelines Regarding Appropriateness of Amplification
As the criteria for implantation have become more lenient, it has become increasingly important for clinicians to ensure that all measures have been taken to determine if the implant candidate will benefit from appropriate amplification. Example procedures that can be used to determine if the hearing aid characteristics are appropriate for the implant candidate’s degree and configuration of hearing loss include the computer-assisted Phase IV Hearing Aid Selection and Evaluation Program (Mason & Popelka, 1982).
When evaluating candidacy for a cochlear implant, it is important to consider the amount of time the candidate has used appropriate amplification. This is particularly true with children, prelingually deafened adults, and adults and children who demonstrate some open-set speech recognition skills, as experience with amplification can greatly influence auditory detection and speech recognition skills.
Candidacy Guidelines Regarding Trial with Amplification
Guidelines regarding the need for hearing aid use prior to cochlear implantation are usually provided in the indications for use in children but are not provided for adults, as it is assumed that adults will have had adequate experience with hearing aids. If they do not have such experience, such as in the case of sudden hearing loss, a hearing aid trial is essential. In the past, guidelines for children required longer trials with amplification than are recommended today. For example, a minimum 6-month trial with appropriate amplification and rehabilitation was recommended to ascertain the potential for aided benefit with the Nucleus 22 device. Currently, the recommended duration of a hearing aid trial varies slightly among devices that have most recently received FDA approval. Current guidelines for the Nucleus Freedom with Contour Advance and Med-El Pulsar CI100 device recommend a 3-to 6-month hearing aid trial for children without previous hearing aid experience. Candidacy guidelines for the Clarion HiResolution Bionic Ear System for children more specifically recommend a 6-month trial for children 2 through 17 years of age, and at least 3 months in children 12 through 23 months of age. For all three devices, the minimum duration of hearing aid use is waived if there is radiological evidence of cochlear ossification.
Speech and Language Evaluation
The purpose of the preoperative speech and language evaluation is to determine if the child demonstrates developmental language and/or articulation disorders, to describe the child’s communicative status with respect to normative models of language development, and to help define appropriate expectations for speech and language skills following intervention with a cochlear implant. The preoperative evaluation provides an opportunity to develop preliminary goals, objectives, and treatment approaches. Similar to speech perception materials, the specific tests used in the speech and language evaluation are dependent on the child’s age and language level. Postoperatively, children with cochlear implants should continue to participate in regularly scheduled speech and language evaluations. Like speech perception measures, postoperative speech and language evaluations help determine which speech cues are or are not being perceived by the child, provide information that may aid in programming the child’s device, and help determine auditory therapy goals.
Psychological Evaluation
The psychological evaluation is primarily performed with pediatric patients but may be necessary with adults who present with concerns regarding cognitive status or mental function. With children, a preoperative evaluation should include assessment of cognitive abilities to determine if factors other than hearing impairment may be hindering the child’s auditory development. Additionally, the presence of a cognitive impairment could greatly influence the child’s ability to develop spoken language skills with a cochlear implant. Thus, identification of such problems is necessary for counseling parents regarding the expected outcomes for their child. Importantly, parents of very young children should be informed that some psychological deficits (i.e., autism) often cannot be identified until the child is 2 years of age or older. As many children receive an implant prior to their second birthday, parents should be warned of the possibility that their child’s performance with the device may be affected by future identification of a cognitive impairment.
During recent years, greater numbers of children are being evaluated for a cochlear implant who present with a disability in addition to their hearing loss (Donaldson et al, 2004). This has increased the need for preoperative psychological evaluations. The results of such evaluations help determine if the child will be able to utilize the auditory signal, influencing the decision of whether or not to provide the child with a cochlear implant. Importantly, the input of the psychologist is essential when counseling parents about expectations for performance and when determining if referrals to other professionals are necessary prior to and after the child receives an implant.
Speech Perception Testing
The primary purpose of speech perception testing is to evaluate performance with an appropriate hearing aid and to determine if the patient’s speech perception skills will likely improve with a cochlear implant. Accurate documentation of preoperative speech perception skills is important as such data will determine if the intervention was appropriate and successful for the patient and will also influence future expansion of cochlear implant candidacy. Postoperatively, speech perception testing is important as it helps verify programming of the patient’s device, helps identify cues that are or are not being perceived by the implant user, and provides a measure of internal device efficacy, information regarding the implant user’s rehabilitative needs, and information that will be useful in determining future selection criteria.
Speech perception testing should be performed in a sound field using recorded test materials whenever possible (this is often not feasible, however, when testing small children). In the past, the most frequently recommended presentation level for speech perception test materials was 70 dB sound pressure level (SPL). This level has been used in numerous clinical trials. Skinner et al (1997) recommended that cochlear implant candidates and recipients be evaluated with speech tests presented at 60 dB SPL. They state that such a level represents a normal conversational level while a presentation level of 70 dB SPL represents a raised-to-loud vocal level. Recently, Firszt et al (2004) demonstrated that Clarion, Med-El, and Nucleus recipients performed similarly on sentence and word tests when stimuli were presented at 60 and 70 dB SPL. Additionally, subjects continued to demonstrate substantial open-set speech perception when stimuli were presented at the softer level of 50 dB SPL. They concluded that candidacy criteria should be based on speech recognition tests presented at 60 and/or 50 dB SPL, as such levels reflect the listening challenges that individuals encounter in natural communication situations. Because of this, many future studies will present stimuli at levels softer than the previously used level of 70 dB SPL.
During speech perception testing, patients should utilize appropriate amplification preoperatively and should utilize their speech processor set to a normal use setting postoperatively. In both instances, they should be tested in a sound-treated room that contains minimal visual and auditory distractions. The presentation level should be calculated using a calibration microphone placed at a reference point located at the center of the listener’s head. Test materials should be presented a single time only, and feedback should not be provided. Caution should be taken to ensure that test materials are used a single time only, as this will reduce the likelihood that learning will influence the patient’s test score.
With adult patients, the speech perception evaluation should include assessment of the ability to understand phonemes and words when presented in isolated monosyllables, an assessment of the ability to perceive words when presented in the context of a sentence, and a measure of lipreading ability. Monosyllabic word tests most commonly used by implant centers today include the Northwestern University (NU-6) Monosyllabic Words Test (Tillman & Carhart, 1966) and the Consonant-Nucleus-Consonant (CNC) Monosyllabic Words Test (Peterson & Lehiste, 1962). Sentence materials mot often used in clinics today include the Hearing in Noise Test (HINT) (Nilsson et al, 1994) and City University of New York (CUNY) sentences (Boothroyd et al, 1985). As many patients present preoperatively with an ability to understand some words and sentences, they can additionally be tested in noise to determine the effect that such noise has on performance. The recommended presentation level for background noise is a +10 signal-to-noise ratio.
To measure lipreading ability, sentence materials may be presented via live voice or via video display, and stimuli should be presented in three different ways. First, sentences should be presented in a “hearing alone” mode to evaluate open-set speech recognition skills. Second, stimuli should be presented in a “hearing plus vision” mode. Lastly, sentences should be presented in a “lipreading alone” mode. Subtraction of the lipreading alone score from the lipreading plus hearing score provides a measure of the amount of lipreading enhancement the patient receives from the hearing aid or cochlear implant. The lipreading alone score is particularly important in the preoperative evaluation as it provides the clinician with an idea of how well the patient will comprehend the vast amount of information that will be provided regarding cochlear implantation. If testing indicates the patient is a poor lipreader, the clinician should utilize means other than verbal communication (e.g., written communication) to ensure that the patient understands all of the information provided. Additional information regarding speech perception tests commonly used with adults in pre- and postoperative cochlear implant evaluations may be found in Table 6–1.
William Luxford of the House Ear Institute led a committee composed of participants from various professional organizations and from cochlear implant manufacturers to develop the Minimum Speech Test Battery for Adult Cochlear Implant Users (Luxford, 2000). The primary purpose of the battery was to provide centers with test materials for evaluation of the pre- and postoperative speech recognition performance of implant users, irrespective of the type of implant or speech processing strategy, so that individual and group comparisons may be made within and across implants and encoding strategies. Many cochlear implant centers utilize this test battery in their clinical practice as a tool for determination of implant candidacy.
| Test | Reference |
| CID Everyday Sentences | Silverman & Hirsh (1955) |
| Consonant-Nucleus-Consonant (CNC) Test | Peterson & Lehiste (1962) |
| Hearing in Noise Test (HINT) | Nilsson et al (1994) |
| Minimal Auditory Capabilities (MAC) Battery | Owens et al (1981) |
| Northwestern University Auditory Test No. 6 | Tillman & Carhart (1966) |
The speech perception evaluation is also a particularly important part of the pre-and postoperative process for children. Rather than utilization of a single test, batteries of tests that measure a variety of speech perception skills should be used with children, as such tests are more sensitive to individual differences in performance. Preoperatively, such tests determine if the child is receiving auditory information with the current amplification and helps determine if speech perception skills will likely improve with a cochlear implant. Postoperatively, such tests provide information regarding the speech cues that are or are not being perceived by the child, may aid in programming his/her device, and may help determine auditory therapy goals.
The specific tests used in pre- and postoperative evaluations of children vary greatly as they are dependent on the child’s age and linguistic and cognitive ability. In general, pediatric speech perception tests can be categorized into three primary types: (1) closed-set tests that measure prosodic cue, speech feature, or word perception; (2) open-set word and sentence tests that provide an estimate of the child’s ability to communicate in the real world; and (3) objective report scales, such as the Meaningful Auditory Integration Scale (MAIS) (Robbins et al, 1991), which uses parental reports to evaluate the child’s listening skills in the daily environment. Speech perception tests commonly used with children in pre- and postoperative cochlear implant evaluations are listed in Table 6–2.
Candidacy Guidelines Regarding Speech Perception Ability
In the early clinical trials, only patients who demonstrated no open-set speech recognition skills were considered to be candidates for a cochlear implant. When the first clinical trials to evaluate the Nucleus device were initiated, adult and pediatric patients were required to demonstrate no open-set speech recognition skills (Mecklenburg et al, 1991). The clinical trial to evaluate the Nucleus 22 device in severely hearing impaired adults was the first FDA-guided study that indicated it was appropriate to implant individuals with some open-set speech recognition skills. This clinical trial was initiated in 1988 and was completed in 1995. Candidates for this study were divided into two phases. Phase I included patients who demonstrated scores less than or equal to 25% correct on Central Institute for the Deaf (CID) Everyday Sentences (in the nonimplant ear), less than or equal to 10% on CID Everyday Sentences in the implant ear, and less than or equal to 30% correct when tested in a binaural aided condition. Phase II subjects included those who scored less than or equal to 25% correct on CID Everyday Sentences in either the implant or nonimplant ear and less than or equal to 30% correct when tested binaurally.
The FDA studies conducted since then have always included patients with some open-set speech recognition skills. Although all three devices recommend use of recorded sentences to evaluate aided benefit in adults, the scores used to define limited benefit vary slightly. For example, indications for the Nucleus Freedom with Contour Advance define limited benefit from amplification as a score ≤50% correct in the ear to be implanted and ≤60% in the best-aided listening condition. Indications for the HiResolution Bionic Ear System define limited benefit as a score ≤50%, whereas the Med-El Pulsar CI100 device recommends a test score ≤40% correct on such measures. It is important to keep in mind that these scores are recommended guidelines. Thus, some patients who have received cochlear implants have demonstrated scores that are better than the above-mentioned recommendations. Additional factors that go into a decision regarding implant candidacy include the patient’s willingness or ability to use amplification, the effect that reduced speech recognition has on the patient’s ability to function in occupational and social settings, as well as the patient’s ability to tolerate the amplified signal.
| Test | References |
| Bamford-Kowal-Bench (BKB) Sentences | Bench et al (1979) |
| Early Speech Perception Test, Low Verbal Version | Moog & Geers (1990) |
| Early Speech Perception Test, Standard Version | Moog & Geers (1990) |
| Glendonald Auditory Screening Procedure (GASP) | Erber (1982) |
| Lexical Neighborhood Test (LNT) | Kirk et al (1995) |
| Ling Sound Test | Ling (1989) |
| Meaningful Auditory Integration Scale | Robbins et al (1991) |
| Meaningful Auditory Integration Scale for Use with Infants and Toddlers (IT-MAIS) | Zimmerman-Philips et al 1998 |
| Minimal Pairs | Robbins et al (1988) |
| Monosyllable-Trochee-Spondee (MTS) Test | Erber & Alencewicz (1976) |
| Multisyllabic Lexical Neighborhood Test (MLNT) | Kirk et al (1995) |
| Northwestern University Children’s Perception of Speech (NU-CHIPS) Test | Elliott & Katz (1980) |
| Phonetically Balanced Kindergarten Word List (PBK-50) | Haskins (1949) |
| Word Intelligibility by Picture Identification (WIPI) | Ross & Lerman (1971) |
Selection criteria for children, which have also been expanded to include candidates with minimal open-set speech perception skills, vary among currently approved devices. FDA-approved indications for use of all three devices state that in younger children, limited benefit is defined as lack of progress in the development of simple auditory skills in conjunction with appropriate amplification and participation in intensive aural rehabilitation over a 3- to 6-month period. All three manufacturers recommended that hearing aid benefit be quantified in young children using measures such as the MAIS (Robbins et al, 1991) or the Early Speech Perception (ESP) test (Moog & Geers, 1990). With older children, all three manufacturers recommend use of the open-set Multisyllabic Lexical Neighborhood Test (MLNT) (Kirk et al, 1995) or Lexical Neighborhood Test (LNT) (Kirk et al, 1995), depending on the child’s cognitive and linguistic skills. Like the adults, the particular score used to define lack of benefit varies. For the Nucleus Freedom with Contour Advance, lack of aided benefit is defined as a score less than or equal to 30% correct, whereas the Clarion HiResolution Bionic Ear System and the Med-El Pulsar CI100 define this as a score ≤20% on such measures.
♦ Additional Factors that Might Affect Candidacy
Age at Implantation
There has been a growing trend to decrease the age at which children receive a cochlear implant. Many investigators have argued that doing so will provide children with greater access to auditory information, which is crucial for development of speech and language skills (Hoffman, 1997; Osberger, 1997). Implantation of children less than 2 years of age requires special skills on the part of the implant team. First, determination of implant candidacy poses a special challenge, as evaluation of the child’s speech and language and speech perception skills is difficult. Second, postoperative follow-up is complicated by the fact that most children in this age group are unable to provide a conditioned response, which is routinely used to program and set the device. Thus, there is an increasing need to develop and evaluate objective measures that will aid in the selection, evaluation, and postoperative treatment of such young children.
Although FDA recommended criteria indicate that cochlear implants are appropriate for children 12 months of age or older for all of the available devices, several investigators in the United States have implanted children who were less than 12 months of age at the time of surgery. Preliminary results obtained with such young children indicate that cochlear implant surgery is safe in children aged 7 to 12 months with appropriate anesthetic and postoperative support (James & Papsin, 2004), and that many children who underwent implantation as infants developed language skills commensurate with those of their hearing peers (Hammes et al, 2002).
Candidacy Guidelines Related to Age
The first clinical trial to evaluate a multichannel cochlear implant in children was the Nucleus 22 device. The candidacy guidelines for this trial indicated that appropriate candidates could be as young as 2 years of age. The first clinical trial to include children less than 2 years of age as candidates was for the Clarion device. In its clinical trial, this device could be used with children as young as 18 months of age if x-rays demonstrated evidence of ossification. All of the devices currently approved by the FDA indicate that cochlear implants are appropriate for children 12 months of age and older.
Auditory Neuropathy/Dyssynchrony
Auditory neuropathy (AN) is a term used to describe an auditory disorder characterized by recordable otoacoustic emissions or cochlear microphonic, absent, or atypical auditory brainstem responses, and speech recognition skills that are poorer than would be expected based on the audiogram (Rapin & Gravel, 2003). Because of these factors, determination of candidacy for a cochlear implant in patients with AN can be difficult. Often, such patients meet candidacy criteria based on their poor speech recognition skills but may fail to meet criteria based on their audiometric thresholds (i.e., their hearing is “too good”). Determination of cochlear implant candidacy of such individuals is additionally complicated by two important factors. First, some patients with AN have demonstrated recovery or improvement of detection and speech recognition skills over time (Neault, 2003). Second, many children who present with AN also present with additional medical diagnoses that may affect expected outcomes for performance (O’Sullivan, 2004; Rance et al, 1999).
Several investigators have reported that cochlear implant recipients with AN demonstrate postoperative outcomes that are similar to those obtained by more traditional cochlear implant recipients (Buss et al, 2002; Mason et al, 2003; Peterson et al, 2003; Sininger & Trautwein, 2002). These investigators also indicate, however, that the outcomes vary from patient to patient, similar to the results obtained by cochlear implant patients in general. Thus, determination of candidacy for a cochlear implant for a child with AN should be determined on a case-by-case basis. One factor that should receive strong consideration is the child’s development (or lack of development) of spoken language skills as the presence of a severe language delay demonstrates a need for intervention.
Children with Additional Disabilities
Previously, children were excluded from participating in early cochlear implant clinical trials if they presented with any type of cognitive delay. More recently, however, children with additional disabilities, such as cognitive and noncognitive delays, are routinely receiving cochlear implants. Pyman et al (2000) performed a retrospective analysis of data on 75 consecutively implanted children up to 5 years of age and found that children with motor and/or cognitive delays were significantly slower than other children in the development of speech perception skills after implantation. Other authors have reported similar findings, but have additionally indicated that provision of a cochlear implant can improve the life quality of profoundly deaf subjects with associated disabilities, increasing their listening and communication skills, their self-sufficiency (Filipo et al, 2004), and their ability to interact with others (Donaldson et al, 2004).
Recipients with Medicare and Medicaid Insurance
Medicare is the U.S. national health insurance program that provides health insurance to citizens age 65 and over, to those who have permanent kidney failure, and to certain individuals under 65 with disabilities. The Centers for Medicare and Medicaid Services (CMS) administers the Medicare program and works with the states to administer Medicaid, the State Children’s Health Insurance Program (SCHIP), and health insurance portability standards. Large numbers of cochlear implant candidates receive their health care coverage from Medicare, Medicaid, or SCHIP. Financial coverage of a cochlear implant for such individuals is greatly influenced by CMS’s national coverage determinations (NCDs), which specify the conditions under which recipients are covered for a cochlear implant.
Prior to 2005, the CMS NCD stated that adults must demonstrate test scores of 30% or less on sentence recognition scores from tape-recorded tests in the patient’s best listening condition, and that cochlear implants in children are not covered until the child is 2 years old and then only where deafness is demonstrated by the inability to improve on age-appropriate closed-set word identification tasks with amplification. This policy was based on previous FDA-approved labeling.
In July 2004, Cochlear Corporation (Sydney, Australia) made a formal request for revision of CMS coverage language to reflect current FDA-approved indications and candidacy standards accepted by the cochlear implant medical community. As a result, CMS reviewed its policies and on April 4, 2005, released a decision stating, “The evidence is adequate to conclude that cochlear implantation is reasonable and necessary for treatment of bilateral pre- or postlinguistic, sensorineural, moderate-to-profound hearing loss in individuals who demonstrate limited benefit from amplification. Limited benefit from amplification is defined by test scores of ≤40% correct in the best-aided listening condition on tape recorded tests of open-set sentence cognition.” Their decision also included the following: “The evidence is sufficient to conclude that a cochlear implant is reasonable and necessary for individuals with hearing test scores of >40% and ≤60% only when the provider is participating in and patients are enrolled in either an FDA-approved category B IDE [Investigational Device Exemption] clinical trial, a trial under the CMS Clinical Trial Policy, or a prospective, controlled comparative trial approved by CMS as consistent with the evidentiary requirements for National Coverage Analyses and meeting specific quality standards.” Additional information regarding CMS coverage language for cochlear implants can be obtained from the CMS Web site: http://www.cms.hhs.gov/.
♦ Conclusion
Selection of candidates for cochlear implantation is a process that has evolved greatly over the past several years. Current candidacy guidelines indicate that it is appropriate to provide cochlear implants to persons with increasing amounts of residual hearing, to persons with increasing amounts of preoperative open-set speech perception skills, and to children as young as 12 months of age. These changes are largely due to the fact that individuals are obtaining remarkable results with cochlear implants.
Technological advances and enhanced surgical techniques will certainly contribute to continued improvements in the field of cochlear implants. Careful documentation of factors that have been used to determine candidacy and reliable and consistent documentation of postoperative performance are needed if the selection criteria used to determine candidacy are to continue to evolve at an appropriate pace.