Purpose
To determine whether a second-generation trabecular meshwork (TM) bypass stent (iStent inject ) influences outflow facility in cultured human anterior segments.
Design
Prospective laboratory investigation using normal human donor eyes.
Methods
Human anterior segments (n = 7) were placed in perfusion organ culture. One or 2 iStent inject stents were inserted into the TM within the nasal and/or superior quadrants using a specially designed injector. Anterior segments were returned to culture and perfused for an additional 24 hours. Morphology of the TM and Schlemm canal (SC) was assessed by scanning electron microscopy (SEM) and 3-dimensional micro-computed tomography (3D micro-CT).
Results
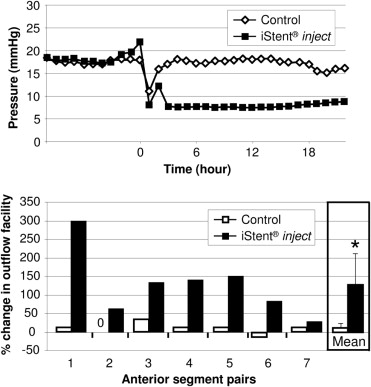
Insertion of 1 iStent inject into the nasal or superior quadrant of the TM increased outflow facility from 0.16 ± 0.05 μL/min/mm Hg to 0.38 ± 0.23 μL/min/mm Hg ( P < .03, n = 7), with concurrent pressure reduction from 16.7 ± 5.4 mm Hg to 8.6 ± 4.4 mm Hg. Addition of a second iStent inject further increased outflow facility to 0.78 ± 0.66 μL/min/mm Hg (n = 2). SEM showed the iStent inject flange compressed against the uveal region of the TM, the thorax securely inserted within the TM, and the head located in the lumen of SC. Dilation of SC was noted around the iStent inject head and SC cell disruption was observed at the iStent inject insertion site. 3D micro-CT confirmed iStent inject placement.
Conclusion
iStent inject , a second-generation bypass stent, increased outflow facility in human anterior segment culture. The iStent inject is a promising new device to lower intraocular pressure via TM bypass.
Primary open-angle glaucoma (POAG) is a neurodegenerative disease characterized by progressive degeneration of optic nerve head axons and death of retinal ganglion cells. If left untreated, POAG will result in irreversible blindness. Of the known risk factors for POAG, elevated intraocular pressure (IOP) is the most prevalent. IOP in the normal eye is maintained through a balance between aqueous humor production by the ciliary body and drainage through the outflow pathways, particularly the conventional outflow pathway. In the conventional outflow pathway, aqueous humor filters through the trabecular meshwork (TM) and drains into the Schlemm canal (SC). From the SC, aqueous humor moves into collector channels and ultimately into aqueous and episcleral veins. In POAG, elevated IOP develops because of increased resistance to aqueous humor drainage at the interface between the juxtacanalicular region of the TM and the inner wall of SC. All current treatment modalities for POAG are targeted towards IOP reduction in order to minimize optic nerve damage and slow the progression of vision loss.
Pharmaceutical agents, laser, and surgical filtration procedures are used to lower IOP in POAG. While effective in many patients, these procedures have their inherent disadvantages. Pharmaceutical agents tend to be costly, and for many individuals, multiple medications are required to achieve an IOP reduction, reducing patient adherence and compliance. In addition, many pharmaceutical agents used to treat POAG have unwanted side effects. For example, brimonidine can cause central nervous system depression; timolol can exacerbate asthma, bradycardia, or congestive heart failure; prostaglandin analogues can change iris color and cause retinal edema; and most agents damage the ocular surface. When medications are unsuccessful in controlling IOP and POAG progresses, adjunct procedures such as laser therapy or filtration surgery can be used to lower IOP. Laser therapy is noninvasive and reduces IOP, but its long-term efficacy is generally less than filtration surgery. Short-term IOP elevation, inflammation, and discomfort are common side effects. Filtration surgery (eg, trabeculectomy) is generally more effective in reducing IOP than pharmaceutical agents or laser therapy; however, complications with hypotony, infection, and scarring reduce its effectiveness. Nearly half of the patients receiving filtration surgery have postoperative complications within 1 year.
Over a decade ago, trabecular meshwork bypass surgery was shown to be successful in lowering IOP. In the initial reports, a silicone tube was placed in the anterior chamber, inserted through the trabecular meshwork, and positioned into SC. Refinement of this procedure using iStent, a first-generation trabecular meshwork bypass stent developed by Glaukos Corporation (Laguna Hills, California, USA), has proved successful in establishing a patent opening in the TM for unobstructed fluid flow from the anterior chamber to the SC. Studies in an ex vivo human anterior segment culture model showed addition of a single iStent increased outflow facility by 84% while lowering pressure from 21.4 ± 3.8 mm Hg to 12.4 ± 4.2 mm Hg ( P < .001). Sequential addition of a second iStent reduced pressure by an additional 3 mm Hg. Clinically, multiple independent studies performed worldwide have shown that the insertion of iStent significantly lowers IOP in POAG or POAG patients undergoing cataract surgery. These studies also found that insertion of the iStent reduced the number of IOP-lowering medications required to maintain the IOP reduction. The iStent has been evaluated in a United States investigational device exemption trial, and a pre–market approval application is currently under review by the United States Food and Drug Administration.
A second-generation stent design, iStent inject , has been developed by Glaukos Corporation on the clinical and surgical foundation of over 1500 iStent procedures worldwide. The iStent inject system was developed to enable implantation of multiple stents to access more of the SC, and thus provide further IOP lowering in POAG patients. In addition, an injector that is preloaded with two iStent inject stents has been developed to improve ease of implantation. The iStent inject is smaller and has a different design than the iStent. However, the underlying principle of both devices remains the same: Bypassing the TM will improve fluid flow from the anterior chamber into SC. The purpose of this study was to determine the capabilities of the second-generation iStent inject by comparing pressure, outflow facility, and morphology in human anterior segment cultures with and without stents.
Methods
iStent inject
The iStent inject is a single-piece heparin-coated titanium stent ( Figure 1 , Left). The posterior side contains a flange at the base that is designed to form an insertion stop on the uveal side of the TM. The middle portion (thorax) is narrower than the anterior and posterior sides and is designed to provide retention within the TM. The head of the iStent inject contains 4 evenly spaced outlets to allow fluid passage from the anterior chamber into the SC. The length of the device is 360 μm and the maximum width of the conical head is 230 μm. The stent is symmetrically designed so that it can be used in either the left or right eye, without requiring a specific radial orientation.
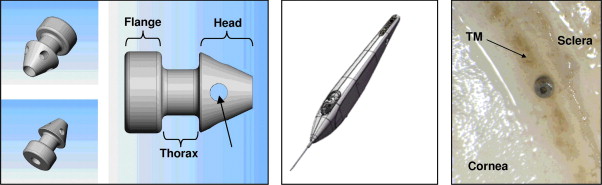
A custom injector has been engineered for implantation of multiple stents ( Figure 1 , Middle). Two stents come preloaded in each injector system. The injector system is designed to deliver both implants into SC without exiting the eye between the implantation of the first and second stents. The portion of the injector that enters the eye is a 23-gauge stainless steel insertion sleeve. The injector features a surgeon-activated release button on the housing, which is pressed to allow the stent to move over a small guiding trocar to exit the injector. The injector system can be stored at room temperature. Figure 1 (Right) shows the posterior view (flange) of an inserted iStent inject into the TM.
Human Anterior Segment Culture
Seven pairs of normal human eyes from 5 male and 2 female donors (68 ± 21 years, mean ± SD; age range 28 to 90 years) were obtained from the MN Lions Eye Bank and placed in anterior segment perfusion culture within 12.8 ± 6.0 hours of death. No eyes had glaucoma or were from patients on topical eye medications. For placement in culture, eyes were bisected at the equator and the iris, lens, ciliary body, and vitreous were removed. The remaining anterior segment was clamped in a modified Petri dish and the eye was perfused with Dulbecco’s modified Eagle’s media (DMEM; Mediatech, Inc, Manassas, Virginia, USA) containing antibiotics (penicillin: 10 000 units; streptomycin: 10 mg; amphotericin B: 25 mg; and gentamicin: 1.7 mg in 100 mL medium). Anterior segments were maintained at 37 C in a 5% CO 2 atmosphere while being perfused at the normal human flow rate of 2.5 μL/min. Pressure was continuously monitored with a pressure transducer connected to a second access cannula built into the modified Petri dish. Pressure was recorded in real time with an automated computerized system.
Once baseline pressure was established, anterior segments were removed from culture and an iStent inject was inserted through the TM into SC from the posterior side. The anterior segment was placed with the corneal epithelium down. A specially designed injector preloaded with an iStent inject was centered against the uveal layer of the TM at a 45-degree angle. Once correctly positioned, the injector spring was released and the head of the iStent inject penetrated the TM into the SC. Anterior segments containing an iStent inject were returned to culture and pressure was monitored for up to 72 hours. Seven anterior segments received 1 stent while 2 of these also received a second stent approximately 24 hours after the first stent placement. The second stent was placed approximately 90 degrees superiorly to the first. Fellow sham control anterior segments were removed from culture, underwent similar manipulations, and were placed back in culture without an iStent inject .
Outflow facility (C; μL/min/mm Hg) in this study was calculated at 0 (C h0 ), 6 (C h6 ), 12 (C h12 ), and 24 hours (C h24 ) post stent insertion by dividing the flow rate (2.5 μL/min) by the pressure reading (mm Hg). Change in outflow facility was plotted as percent of C h0 and obtained by [(C 6h /C ho ) – 1] × 100. The outflow facility of anterior segments containing the iStent inject were grouped and compared to the outflow facility calculated from the fellow sham control anterior segments. Group means and standard deviations were calculated and significance analysis performed by a paired, 2-tailed Student t test. Differences were considered significant when P < .05.
Histologic Examination
Light Microscopy
At the completion of the anterior segment culture experiments, tissue wedges 180 degrees apart that included the TM and SC were isolated and fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.2). These tissue wedges were embedded in JB4 plastic, sectioned at 3 μm, and stained with toluidine blue. Examination of the tissue sections was performed in a masked fashion using predetermined criteria as described previously. TM was considered normal if sections had numerous cells attached to the lamellae with no disruption of the juxtacanalicular tissue. SC was considered normal if the endothelium of the inner and outer walls was continuous and undisrupted.
Scanning Electron Microscopy
Tissue wedges fixed in 4% paraformaldehyde/2% glutaraldehyde in 0.1 M phosphate buffer (pH 7.2) that contained the TM, SC, and the iStent inject were processed for scanning electron microscopy (SEM). Tissue wedges were rinsed twice in 0.1 M phosphate buffer and dehydrated in an ascending ethanol series. Dehydrated tissue was critical point dried and sputter coated with gold palladium. The tissue specimens were examined on a Hitachi H4700 scanning electron microscope (Hitachi High Technologies America, Inc, Schaumburg, Illinois, USA). Examination included 4 tissue regions from 3 different anterior segments.
Three-dimensional Micro-Computed Tomography
Two anterior segments containing 1 iStent inject in the nasal quadrant and a second iStent inject in the superior quadrant were selected for analysis by 3-dimensional micro-computed tomography (3D micro-CT). Two tissue wedges containing iStent inject stents were excised from each anterior segment and fixed in 4% paraformaldehyde/2% glutaraldehyde in 0.1 M phosphate buffer (pH 7.2). Each tissue wedge was rinsed twice in 0.1 M phosphate buffer for 10 minutes followed by immersion in 1% osmium tetroxide for 90 minutes to increase the radiograph opacity. Tissue was rinsed in 0.1 M phosphate buffer, dehydrated in an ascending series of ethanols, and incubated in 2 changes of propylene oxide for 10 minutes each. The tissue specimens were incubated overnight in a 1:1 mixture of propylene oxide and Epon-Araldite (Electron Microscopy Sciences, Hatfield, Pennsylvania, USA). The tissue quadrants were embedded in flat tissue molds with the corneal endothelium facing the top of the mold in Epon-Araldite (Electron Microscopy Sciences). The resin was polymerized at 60 C overnight in an embedding oven.
Processed tissue wedges were scanned at 2 μm voxel resolution at the National Synchrotron Light Source (NSLS) at Brookhaven National Laboratories (BNL) in New York. The advantages of using the synchrotron source include higher radiation flux for shortened scan times and parallel, tunable, monochromatic illumination to maximize resolution and tissue contrast. The 3D images were examined using ANALYZE, a 3D image software analysis and display program.
Results
Outflow facility increased in all 7 anterior segments that received an iStent inject and the increase was maintained throughout the 24-hour incubation ( Figure 2 and Table ). Collectively, insertion of 1 iStent inject into human anterior segments decreased pressure within 6 hours from 16.7 ± 5.4 mm Hg to 8.6 ± 4.4 mm Hg (n = 7) and increased outflow facility from 0.16 ± 0.05 μL/min/mm Hg to 0.38 ± 0.23 μL/min/mm Hg ( P < .03, n = 7). In contrast, the fellow sham control anterior segment had minimal change in pressure (17.6 ± 4.9 mm Hg to 16.4 ± 5.7 mm Hg) and outflow facility (0.15 ± 0.05 μL/min/mm Hg to 0.17 ± 0.06 μL/min/mm Hg, P > .67). In 2 of the 7 anterior segments, a second iStent inject was added superiorly to the first stent. In these 2 anterior segments, insertion of 1 iStent inject decreased pressure from 17.0 ± 4.2 mm Hg to 10.5 ± 5.0 mm Hg. Addition of the second iStent inject further reduced pressure to 5.0 ± 4.2 mm Hg. Outflow facility increased from 0.15 ± 0.05 μL/min/mm Hg to 0.26 ± 0.13 μL/min/mm Hg with insertion of 1 iStent inject. Addition of a second iStent inject increased outflow facility to 0.78 ± 0.66 μL/min/mm Hg.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


