Wound Healing
The wound healing process can effectively be broken into three distinct but overlapping phases: the inflammatory phase, the proliferative phase, and the remodeling phase.
The inflammatory phase (immediate onset lasting 2 to 5 days): This initially begins with hemostasis over the first 5 to 10 minutes via vasoconstriction and activation of the clotting cascade. This includes platelet aggregation and deposition of thrombin and fibrin. Vasodilation follows and peaks at approximately 20 minutes before vascular tone returns to its baseline. Also beginning immediately, aggregating platelets and local phagocytic cells (predominantly neutrophils for the first 24 to 48 hours) release a variety of proinflammatory and chemotactic cytokines. This in turn leads to an influx of macrophages. These are the essential mediators of early wound healing and predominant cell type from 48 hours through the remainder of the inflammatory phase. The macrophages for their part phagocytize bacteria and damaged tissue and release their own milieu of cytokines, growth factors, and chemotactic agents. This subsequently promotes the arrival of fibroblasts, endothelial and epithelial cell migration, and wound contraction by myofibroblasts.
The proliferative phase (day 2 through 3 to 4 weeks): Fibroblasts enter the wound around day 2, which signifies an overlap period of the inflammatory and proliferative phases. As the inflammatory phase ends and macrophages decline in number, fibroblasts take over as the predominant cell type. Through the resulting fibroplasia, granulation tissue forms, and extracellular matrix (ECM) is deposited consisting of disorganized collagen (mostly type III), elastin, and fibronectin. Angiogenesis and wound contraction (both begin around day 4) follow and are mediated by endothelial cell migration and wound margin myofibroblasts, respectively. Wound contraction is maximal around days 12 to 15. Reepithelialization also occurs during this phase through migration of epithelial keratinocytes from the wound margins and the bases of pilosebaceous subunits preserved in the wound at the level of the dermis. The process occurs best over a moist surface of viable tissue. This underscores the importance of a moist wound environment that not only aids migration but also prevents excess scabbing that acts as a barrier to migration. Reepithelialization ceases when the opposing sheets of migrating epithelial cells meet and reestablish contact inhibition.
The remodeling phase (3 weeks to 2 years): Collagen remodeling occurs with fibers becoming aligned in a more parallel fashion and type I collagen being deposited in greater proportion. Excess cells are removed via apoptosis and phagocytosis. Also during this phase, the wound’s tensile strength will increase roughly according to the following progression: 15% of original strength by 3 weeks, 60% by 6 weeks, and 80% by 3 to 6 months, which is the maximum achieved (
2).
Deregulation or interruptions of this wound healing process, including interventions by the surgeon, can lead to alterations in the scar that ultimately forms. Other systemic and/or external processes including malnutrition, diabetes mellitus, smoking, excessive sun exposure, and immunosuppression can severely impede this process and lead to poor wound healing and worsened outcomes.
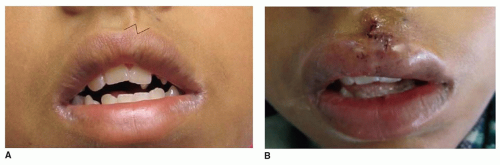
Scar Types
Scars can be classified broadly into hypertrophic scars (Hsc)/keloids and non-Hsc (
1). Hsc and keloids are quite similar in nature and represent an aberration in the wound healing process. Patients prone to these types of scars have been shown to have down-regulation of various apoptotic genes and overexpression of signaling factors such as IL-6 and TGFβ1 and β2. Resulting histologic findings include ECM overproduction and deposition of highly disorganized collagen by hypermitotic fibroblasts (
2,
3). Keloids, by definition, are elevated fibrous scars that extend beyond the borders of the original wound, do not regress, and usually recur after excision. They can take between 3 months and years to develop. Hsc develop quicker (within 8 weeks), are confined to the wound borders, and usually will regress over a period of 12 to 18 months (
2,
4). Risks for keloid formation include darkly pigmented skin (15- to 20-fold increase), familial predisposition, and high wound tension. Hsc are less likely to be related to skin pigmentation. Care must be taken by the surgeon when working with patients at risk for keloid formation as there is a very high rate of recurrence, especially when surgical excision is used alone (
5).
Non-Hsc can be divided into either atrophic (depressed) scars or scars that are flat or protrude minimally above the level of the adjacent normal skin (
1). While less prone to recurrence and complications than keloids and Hsc, these scars can nonetheless be disfiguring and challenging to treat. The pathophysiology of this scar type is likely related to a hypoxic wound environment, abnormal levels of cellular apoptosis, and limited angiogenesis. The ultimate result is reduced deposition of ECM and collagen and a depression of the wound bed with regard to surrounding normal skin. Similar to keloid and Hsc, excessive tension on wound edges increases risk of atrophic scar formation.