Fig. 10.1
MRI revealed a 2.9 × 2.2 cm lesion in the intersection of deep and superficial lobes of the left parotid gland, as well as lymphadenopathies in levels Ib, II, and III. Red arrows showing the primary lesion and pathological neck nodes
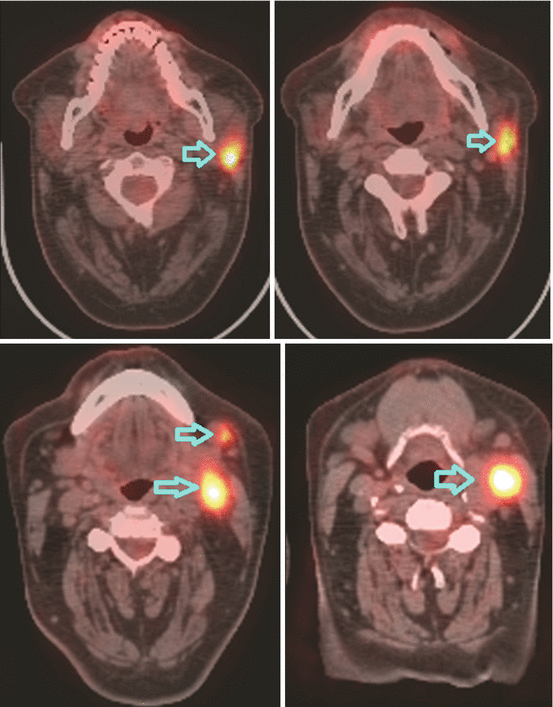
Fig. 10.2
Lymphadenopathies in levels Ib, II, and III of the left side of the neck seen on PET-CT scan. Blue arrows showing FDG uptake in pathological neck nodes
The histologic evaluation of the fine-needle aspiration of the left parotid was reported as a high-grade salivary gland carcinoma .
After facial nerve decompression, radical parotidectomy with modified radical neck dissection of left side was performed. The pathology revealed salivary duct carcinoma, coherent with sarcomatoid component. The tumor was 3 cm in size. Surgical margins were positive in anterior and posterior margins as well as the margin over facial nerve. Out of 40 resected lymph nodes, 6 were metastatic, and ECE was positive.
The patient was staged as T2N2bM0 parotid gland cancer.
2 Evidence-Based Treatment Approaches
Unless the nerve is involved, facial nerve-preserving surgery with adjuvant radiation therapy (RT) is the preferred treatment. More aggressive surgery has not been shown to increase local control (LC). Adjuvant RT does not have any negative effect on the function of the facial nerve [8]. In patients with clinically positive lymph nodes, neck dissection should be performed prior to adjuvant RT [9]. However, in case of high-risk factors for local recurrence (LR), where adjuvant RT is indicated postoperatively, neck treatment can be performed solely with RT [10, 11]. Exception to this is patients with parotid tumors with facial nerve involvement , submandibular tumors, or tumors of minor salivary glands in the floor of mouth, tongue, pharynx, and larynx, in whom elective nodal dissection of levels I–III is the standard treatment [10].
Several studies reported LC rates that ranged between 51 and 99 % and 5- and 10-year overall survival (OS) rates up to 78 and 67 %, respectively, with different treatment modalities. In patients treated with surgery alone, high rates of LR have been reported [3]. Retrospective studies showed that adjuvant RT improves LC, particularly in cases with locally advanced disease, positive lymph nodes, close or positive surgical margins, bone invasion, or PNI [9, 12–16].
Dose-response relationship was reported for photons [9], and a minimum dose of 66 Gy is recommended. Neutron therapy was shown to result in higher LC rates in unresectable and recurrent tumors. However, late complications were also higher and survival rates were equal with neutrons compared to photons [17–19].
Pleomorphic adenomas are benign tumors, and superficial parotidectomy is the standard treatment with high rates of LC [20]. Radiotherapy is indicated in case of positive surgical margins, recurrent and unresectable tumors [21–24]. Adenoid cystic carcinomas are known for their late recurrences [15]. Simple excision is not adequate for these tumors, as residual tumor is highly probable. Fortunately, nodal recurrence risk is relatively low. Combined treatment strategies yield high LC rates [13, 25–28]. Salivary duct carcinoma is highly aggressive; therefore, all cases should receive adjuvant RT. The surgical approach for most minor salivary gland tumors is more difficult; thus adjuvant RT is indicated nearly in all cases.
Sublingual gland tumors are mostly locally advanced and high grade but have a low propensity for lymph node involvement. Adjuvant RT is indicated in the majority of the patients as they are mostly high risk.
Chemotherapy (CT) is used for palliative treatment only [29]. There are limited data regarding the efficacy of concurrent adjuvant chemoradiotherapy (CRT). In patients with T3–4 tumors, positive surgical margins, positive lymph nodes, or PNI, CRT was shown to have a LC rate higher than 90 %, but the number of patients was limited and severe acute toxicity was reported [30, 31]. In another study where 24 patients were evaluated, postoperative CRT was found to be superior to RT alone in terms of OS [32].
3 Target Volume Determination and Delineation Guidelines
Gross Tumor Volume (GTV)
GTV should include the gross tumor and involved lymph nodes detected by clinical examination, CT, MRI, PET/CT, and intraoperative findings, if operated. GTV is divided into two; GTVp defines the primary tumor, and GTVn defines the involved lymph nodes. In postoperative cases GTV is not stated as it is assumed to be no tumor or grossly involved lymph nodes left.
Following structures should be evaluated carefully whether they are involved in parotid tumors:
Anteriorly:
Is the skin involved?
Laterally:
Is the skin involved?
Are the pterygoid plates intact?
Medially:
Is there tumor extension into the mandible?
Is the masseter muscle involved?
Posteriorly:
Does the tumor extend to the stylohyoid foramen which the facial nerve passes through?
Is there tumor extension through the ear canal?
Is the carotid artery intact?
Superiorly:
Is the skull involved?
Inferiorly:
Is there extraparenchymal extension?
Clinical Target Volume (CTV)
CTV1 includes the entire ipsilateral parotid gland, parapharyngeal space, infratemporal fossa, and ipsilateral subdigastric nodes. It is not necessary to treat the surgical scar to full skin dose as scar failure is less than 1 %. In case of PNI, the cranial nerve pathway from the parotid up to the base of the skull should also be delineated.
CTV2 covers the entire operative bed as it is the region of high risk for subclinical disease and potential routes of spread.
CTV3 is the low-risk regions of subclinical disease. For parotid tumors, elective neck irradiation is indicated in patients with locally advanced disease, squamous and undifferentiated histologies, facial nerve involvement at diagnosis, and recurrence. Ipsilateral levels Ib, II, and III should be treated. Bilateral elective neck irradiation is not recommended. In case of positive lymph nodes after neck dissection, ipsilateral neck between level I and IV should be included. For submandibular tumors, elective ipsilateral levels I–IV should be treated. If the tumor extends toward the midline, bilateral neck irradiation is indicated. Elective neck irradiation is not necessary in small acinic cell and adenoid cystic cancers. Delineation of the cranial nerve pathway also is not required in adenoid cystic carcinomas of submandibular gland with focal PNI as the recurrence rate of this site is very low with a risk of significant morbidity; however, this is not the case for minor salivary gland adenoid cystic carcinomas. The base of skull also is included in tumors of the palate and paranasal sinuses. Elective neck irradiation is indicated for tumors of the tongue, floor of the mouth, pharynx, and larynx. Lingual and hypoglossal nerves may also be invaded in sublingual tumors. In this case, the pathway from the gland to the skull base should be delineated as well.
Planning Target Volume (PTV)
A margin of 3–5 mm is added in all directions; however, it may be minimized to 1 mm in areas adjacent to critical structures.
Case Contouring
Case contouring is demonstrated in Fig. 10.3.
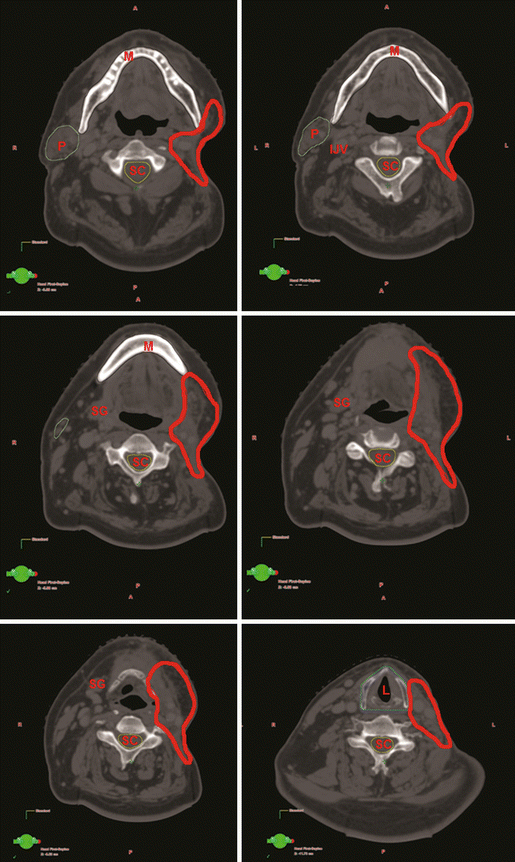

Fig. 10.3
Delineation of target and normal volumes for T2N2bM0 parotid gland cancer (CTV64 Gy = red)
4 Treatment Planning
Guidelines for Target Volume Doses: Guidelines for target volume doses are summarized in Table 10.1.
Table 10.1
Guidelines for target volume doses
TNM
CTV1 (60 Gy/30 fr)
CTV2 (54 Gy/30 fr)
CTV3 (50 Gy/30 fr)
T1–2 N0
GTVp (whole gland)
–
–
T3–4 N0
GTVp (whole gland)
0.5 cm
Ipsilateral Ib–III and intraparotidal except for adenoid cystic and acinic cell carcinomas
T1–4 N+
GTVp (whole gland), GTVn
Ipsilateral adjacent lymph nodes (intraparotidal, the one level above, and the one level below)
Remaining lymph nodes (ipsilateral Ib–IV, contralateral I–III for submandibular tumors extending toward midline)
Guidelines for Normal Tissue Constraints: Guidelines for normal tissue constraints are summarized in Table 10.2.
Table 10.2
Guidelines for normal tissue constraints
Structure
Constraints
Brain< div class='tao-gold-member'>Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access
