Revision cochlear implant (RCI) surgery has become an important tool for the management of cochlear implantation complications. This review encompasses both common and uncommon indications of RCI, diagnostic and management considerations, outcomes, surgical principles, and emerging applications. In summary, early identification of complications and suboptimal device performance is imperative for children who are in critical periods of communicative development. Independent of indications, most RCI achieve a successful restoration of sound. Although rare, it is prudent to discuss the potential complications of revision surgery and their implications with patients and their families.
Since the first intrascalar cochlear implant (CI) placement by House and Edgerton in 1961, cochlear implantation has evolved to become an effective and widely performed procedure for the restoration of sound in severe and profound hearing impaired individuals. The number of patients benefiting from this technology continues to grow exponentially. Any threat to the functional gains evokes concern in all users, especially children and their families who fear the loss of newly found access to sound and communication. As the experience with CI grows, replacement or revision cochlear implant (RCI) surgery has become an important procedure. In 1985, Hochmair-Desoyer and Burian reported the first RCI. Since then, multiple publications have addressed the safety of this procedure, including the preservation or enhancement of speech perception performance. Initial reports encountered decrements in electrode activation, diminished speech perception, and intracochlear trauma, suggesting that RCI could have a negative functional outcome. However, with the advances in technology, particularly the use of thinner electrodes, and the increased surgical experience of the otologic community, RCI is a safe procedure that, nonetheless, requires careful consideration of the indications and expected benefits.
Indications
In general, the reported rate of revision surgery accounts for approximately 3% to 15% of CI operations in different studies. This variability can be explained by differences between centers and different study characteristics. Moreover, some of those studies combined pediatrics and adult populations, which increases the incidence of revision surgery because children have a higher rate of device failure because of an increased association with head trauma.
Although there is less data available regarding revision CI surgery in children alone, when compared with adults, the prevalence seems to double. In later studies whereby centers looked separately at their population, Arnoldner and colleagues found an incidence of RCI of 7% in adults versus 14% for children, whereas Rivas and colleagues encountered 4.8% versus 13.0%, in adults and children respectively, in a 10-year period.
To facilitate the approach for RCI and to guide required interventions, there are 2 main indications: non–device-related indications and device-related indications. The first one, although less common (26%), includes those patients who require RCI because of infections, allergic reactions, cholesteatomas, misplacements of the electrode, electrode extrusions, and, rarely, single-channel upgrades.
Non–Device-Related Indications
Infections
The incidence of infectious complications in patients with CI range from 1.7% to 8.2%. Scalp flap infections can initially present as tenderness over the receiver/stimulator, progressing to edema, erythema, and eventually an underlying abscess. The initial treatment should be conservative, usually requiring close observation and intravenous (IV) antibiotics. In cases of persistence or recurrence of symptoms, the authors proceed with wound debridement, wash out, and cultured-guided antibiotics in an attempt to maintain the implant integrity. This practice, however, represents a real challenge because of the potential formation of biofilms.
Bacterial biofilms are composed of communities of bacteria enclosed in a self-produced polymeric matrix of mainly exopolysaccharides with a propensity to attach and persist on the surface of biomaterials. The biofilm may develop defense mechanisms against both a host immune system and antimicrobial agents preventing them from treating the infection. In this circumstance, the removal of implanted devices is often inevitable to eradicate the disease. In cases whereby IV antibiotics and local wound care fail to resolve infection, the device should be removed leaving the electrode within the cochlea. The patients are treated with parenteral antibiotics for at least 6 weeks, followed by RCI.
The mechanisms responsible for intractable infection of CI are unclear. Staphylococcus aureus has been the most common pathogen identified in implant infections, which suggests a nonotologic source because this is not a common middle ear pathogen. Moreover, the safety of implanting CI with a low rate of postoperative device infection in middle ears that are chronically contaminated or intermittently, acutely infected has been reported. However, the problem is more complex than simple exposure of the device to bacteria. In fact, Ruellan and colleagues detected bacterial biofilm on 9 CI removed because of device failure, without evidence of infection, demonstrating the need for investigating the pathophysiology of CI-associated biofilms.
This finding may lead to the development of biomaterials that could limit the formation of biofilm or to the creation of implants with drug delivery systems that could treat such formation. Aids in this process include hand hygiene, aseptic technique, and routine use of careful antibiotic irrigation during implantation.
Cholesteatoma
In the 1980s, CI was contraindicated in patients with otitis media because it was hypothesized to be associated with a higher incidence of inner ear infections and intracranial complications. However, later studies of complications of CI in children did not show an increased incidence of otitis media. Most of the time these children are treated conservatively with oral antibiotics. Only a few reports of patients requiring explantation have been described. Some of them, although rare, secondary to cholesteatoma. Lin, for example, identified 4 out of 186 children with a CI having cholesteatoma; all required surgery, and 3 of them eventually needing explantation to eradicate the disease. Although the overall incidence of cholesteatoma is similar to that of children with normal hearing, the decision making and treatment options in managing cholesteatoma in children with CIs are challenging for clinicians.
Different approaches are used in an attempt to maintain the implant survival, prevent complications, and eradicate the cholesteatoma. Intact canal wall tympanomastoidectomy and modified radical mastoidectomy with nonobliterative closure of the external auditory canal are the most-used techniques to clear the ear of disease. The type of procedure performed depends on the extent of the cholesteatoma and the involvement of the disease with relation to the electrode array.
In cases whereby the electrode array is uninvolved and the cholesteatoma is small, a canal wall up cartilage tympanomastoidectomy is recommended in an attempt to maintain the ear canal and the existing CI ( Fig. 1 ). This technique provides postoperative visualization of the tympanic membrane to monitor recurrence. This visualization is the major advantage over canal overclosure techniques whereby entrapment of squamous epithelium is a risk and magnetic resonance imaging cannot be perform because of the magnet and the signal void around the CI.
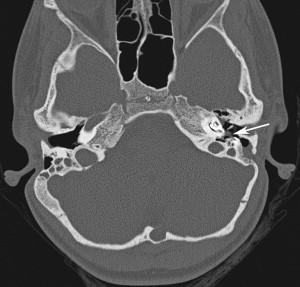
Regardless of the technique used, most surgeons prefer a staged procedure to confirm eradication of the disease or to excise any recurrence. Moreover, when the cholesteatoma surrounds the electrode array, explantation is necessary to prevent tracking of the squamous epithelium into the cochleostomy. This decision is difficult to make because the removal of the CI imparts the loss of auditory input to a child who is already cognitively impaired, especially when reimplantation is often difficult because the soft-tissue tract that the electrode occupies can close. The use of a nonmetallic spacer has been proposed to keep this tract open during absence of the implant. The replacement of the CI should be delayed 6 to 12 months if the disease is eradicated.
Despite repeated procedures, it may not be possible to completely and permanently eradicate all cholesteatoma. In these cases, because of the risk of meningitis, permanent explantation should be considered with implantation of the contralateral ear.
In conclusion, although the treatment of cholesteatoma in children with CI is challenging, it is possible to preserve the CI in some cases. If the electrode array is involved, the CI must be removed and appropriate measures must be taken to prepare the ear for reimplantation and to minimize potential risks, such as the recurrence of entrapped epithelium, extrusion of the device, and meningitis. An appropriate procedure should be based on the individual clinical presentation at the initial evaluation and time of surgery.
Allergic reactions
Although wound infection is the most common cause of device extrusion, other rare causes must be suspected when recurrent extrusion is encountered. Silicone allergy should be considered, particularly in delayed-onset device extrusion with negative wound culture results or no response to antibiotics. Four cases of CI extrusion as a result of silicone allergy have been reported. Puri and colleagues documented hypersensitivity to the silicone liquid silicone rubbers (LSR)-30 component of a Nucleus 24 Contour device (Cochlear Ltd, Sidney, Australia), whereas Kunda and colleagues documented extrusion secondary to allergic reactions to the room temperature vulcanization (RTV) silicone adhesive from the CI casing. The silicone components used in CI differ in the 3 major CI manufacturers: Advanced Bionics, Cochlear, and Med-El. LSR-70 is used in the receiver-stimulator of Advanced Bionics devices, whereas cochlear devices are cased in liquid silicone rubber LSR-30 and RTV silicone adhesive. Med-El devices use RTV silicone adhesive, liquid silicone rubber LSR-40 in the active electrode lead, and silicone tube high consistency rubber 50 durometer in the reference electrode lead.
All 3 CI manufacturers carry an allergy kit. These kits provide the samples of materials contained in the specific type of CI. The different tests that can be used in allergy testing of CI components include the patch test, prick test, and intradermal test. Patch testing is used in contact dermatitis and is appropriate for nonsoluble materials. In this test, a small amount of allergen is placed over the skin and covered with a watertight bandage for 48 to 96 hours. Evidence of edema or erythema over at least half of the tested area is considered a positive result. This test has a sensitivity and specificity of 70%. The prick test is used with soluble or crushed, diluted nonsoluble allergens. This test involves pricking or scratching the skin with a drop allergen. When a result is negative or equivocal, intradermal testing is a more sensitive option. This test includes a dilute sterile extract with diluting agent alone as a negative control and histamine as a positive control. A result is considered positive if a wheal forms and it is at least 5 mm larger than the control reaction after 15 minutes.
On confirmation of silicon allergy and the specific component to avoid, explantation and reimplantation is warranted. There are different options for RCI, including a custom-made CI without the affecting allergen or the use of a ceramic receiver stimulator. Because only a few cases have been reported in the literature, there is no consensus on the time of reimplantation. However, all reported cases have been reimplanted in a delay fashion. The authors advocate the removal of the receiver stimulator, leaving the electrode within the cochlea to prevent ossification, followed by reimplantation at least 6 weeks later.
Extracochlear electrodes
Extracochlear electrodes are the most common cause of reimplantation after device-related indications in children. In this group, there can be misplacement of the electrode array ( Fig. 2 ) or, more commonly, electrode array extrusion.
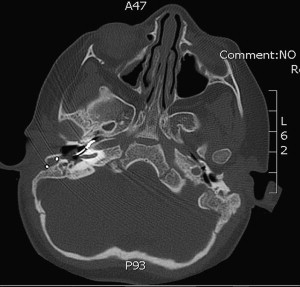
Patients experiencing migration of the electrode array usually present with reduced speech perception and lack of progress in receptive and productive skills. Pain with audition, facial nerve stimulation, and vertigo has also been reported. Audiologic evaluation usually shows an increase in clinical unit levels, a loss of neural response telemetry, and an increase in impedance levels. Programming changes alleviated some of these symptoms through the removal of basal channels from the map. However, all of these findings can also be encountered in device failures. Computed tomography scan makes the differentiation and reveals the displacement of electrodes in migration cases.
Extrusion occurs more frequently in patients who had only partial insertions initially and in patients with known cochlear ossification. This condition poses a particular challenge for RCI because of fibrosis and obliteration of the distal electrode tract, which often prevents the possibility to attain a full reinsertion.
Electrode extrusions tend to occur in younger-aged patients, potentially because of the decreased stability of the electrode lead in smaller mastoid cavities. This condition can cause a springlike effect leading to progressive electrode withdrawal. The slow decline in speech perception found in these patients before RCI suggests that extrusion may be a dynamic process that can progress.
The underlying cause of electrode extrusion is unknown, but several theories exist. In children, it is proposed that extrusion occurs as the skull grows, putting a strain on the electrode, although Roland and colleagues have shown no electrode migration in the pediatric population over time. Ossification of the cochlea and the physical forces placed on the outer wall by the electrode have been associated with extrusion. The use of perimodiolar electrodes may decrease this occurrence.
To reduce the probability of electrode extrusion, some techniques have been proposed. Probably the most widely used is tightly packing the cochleostomy with tissue (fascia, muscle, or perichondrium) to hold the electrode in place. Cohen and Kuzma have used a titanium clip to hold the electrode to the incus buttress. Balkany and Telischi developed the split-bridge technique in an attempt to reduce the incidence of electrode migration.
Device-Related Indications
Device-related indications have been the most common cause of reimplantation in most centers (74%) and include those cases where there is facial nerve stimulation and confirmed or suspected device failures.
Facial nerve stimulation
The facial nerve electric stimulation is a complication after CI with rates between 1% and 15%. Possible explanation of this adverse effect is a leakage of currents caused by a change in the electric properties of the bone or close proximity of the facial nerve to the outer wall of the cochlea, together with the need for high electric current to stimulate the auditory nerve (ie, malformations or ossified cochleae).
In theory, the perimodiolar electrodes with contacts facing toward the modiolus have less current flow toward the outer wall of the cochlea and might reduce the problem of facial nerve stimulation. Reimplantation, therefore, seems a viable option when the new device is equipped with such a perimodiolar electrode.
Confirmed or suspected device failures
It is important that clinicians are able to diagnose suboptimal or anomalous implant performance and counsel CI candidates, and their families, on prospects for improved performance with RCI. An important concern relates to the fact that device defects may be missed in children with limited language capabilities unless an observant parent, therapist, or teacher is able to detect a decline in speech perception or less-than-expected progress. This concern is particularly important because Marlow and colleagues documented that younger children are more likely to achieve and exceed previous peak performance than their older counterparts. This observation emphasizes the detrimental effect of delayed RCI in children who are in crucial periods of speech recognition and language development.
When concerns with implant performance arise, 3 principal factors are considered. The audiologist will first exchange external equipment and adjust program settings. The medical status of patients and the placement of the electrode array are then assessed. Finally, internal-device integrity testing may be needed. In cases whereby patients report no sound, defective external equipment has been ruled out, and the telemetric locking capability of the system is lost, the device is confirmed to be defective (hard failure).
Confirmed device failure is defined as the absence of auditory input or electronic lock between external and internal components. These patients receive no auditory input from the device, which usually occurs as a sudden loss of sound perception. Approximately 30% of patients might experience preceding non–life-threatening head trauma or short-term (less than 4 months) signs and symptoms of atypical tinnitus, intermittent function, shocking sensation, fluctuating impedances, loss of electrodes, or open circuits before the loss of sound or telemetric lock. The evaluation confirms that no connection can be made with the device by external means and the decision to revise is straightforward.
However, in patients who continue to perceive sound but experience diminished benefit, fail to progress, or develop aberrant symptoms, a device defect may be suspected.
Suspected device failure (soft failure) is considered a clinical malfunction of the CI whereby the device may still provide some auditory input but patients develop a decrement in performance ( lack of progress ) or unacceptable aversive symptoms. Those symptoms include intermittent CI function; programming difficulties; aberrant sounds; increased loudness; or nonauditory symptoms, like pain, vertigo, or headaches. Information about declining performance, particularly when accompanied by programming difficulties and open or short circuits, should prompt consideration of RCI.
The clinical impression of suspected device malfunction must be investigated by clinical, audiologic, and radiographic criteria in an effort to exclude medical problems and hardware- or software-related causes of performance deterioration. However, clinical acuity is the cornerstone of the decision to proceed with revision surgery in these cases because most of these children present with normal integrity testing. Furthermore, integrity tests done by the manufacturers before RCI have shown that approximately 50% of the suspected device-failure cases function within manufacturer specifications, and integrity tests do not correlate with ex vivo device analysis. Therefore, the diagnosis is supported by the return of function with subsequent reimplantation.
On completion of RCI surgery, all explanted failed devices must be sent to the manufacturer for ex vivo device analysis and identification of the source of the failure. The most common causes of device failure include case fracture or loss of hermetic seal.
Outcomes of revision cochlear implantation
Since the Hochmair-Desoyer and Burian report, others studies addressed the safety of RCI, including the preservation or enhancement of speech perception performance. RCI has been shown to be a safe procedure with postoperative hearing results comparable to the initial CI performance.
When comparing preoperative speech performance immediately before RCI and speech performance 6 or more months after reimplantation, most studies have demonstrated that revision surgery maintains or improves speech perception in 89% to 100% of the cases ( Table 1 ). This finding is particularly true for non–device-related indication and confirmed device failures. Those few cases whereby patients do not return to peak performance are usually suspected failures. Marlow and colleagues found that depending on the definition of success, (1) a return to previous peak performance or (2) an increase in speech perception in cases of low performance and failure to progress, suspected device failure was deemed to be successful in 67% of the cases with low and stagnant performance versus 83% of the cases with previous decrements in performance. Importantly, aversive auditory symptoms, like intermittent/decline or poor performance, atypical tinnitus, increased loudness, or nonauditory symptoms, like pain or headache, were the most common clinic manifestations that usually resolve with RCI and were found to be positive prognostic factors of improvement in speech perception.
| Report | Incidence RCI a /Device Failure b (%) | Average Duration First Device (y) | Indication hf/sdf/other (n) | Postoperative Speech Perception Better/Same/Worse c (%) | Device Defect Found (%) |
|---|---|---|---|---|---|
| Chung et al, 2010 d | 7.5 | 4.5 | 26/14/73 | 71/29/0 e | 26 |
| Marlow et al, 2010 | 13/- | 3.4 | 26/18/18 | 87/10/3 | 84 |
| Brown et al, 2009 d | 5.5/4.2 | — | 24/10/10 | — | 86 |
| Cullen et al, 2008 | 11.2/- | 2.6 | 49/16/42 | — | 88 |
| Battmer et al, 2007 d | -/3.8 | — | — | — | — |
| Cote et al, 2007 | 8.0/6.2 | 6.4 | 19/2/6 | 26/63/11 | 68 |
| Migirov et al, 2007 | 12.5/6.9 | — | 21/1/16 | — | — |
| Fayad et al, 2006 | 5.6/3.2 | — | 17/1/9 | — | — |
| Tambyraja et al, 2005 d | — | — | 416/71/305 | — | — |
| Lassig et al, 2005 | 6.4/4.2 | 3.9 | 16/4/10 | 72/16/11 | — |
| Beadle et al, 2005 | -/26.7 f | 3.4 | — | — | — |
| Weise et al, 2005 | -/5.4 | 2.0 | — | — | — |
| Arnoldner et al, 2005 | -/13.9 | — | — | — | — |
| Maurer et al, 2005 | 12.6/11.2 | — | 15/0/2 | — | 100 |
| Parisier et al, 2001 | -/6.7 | 2.7 | — | 37/63/0 | — |
| Alexiades et al, 2001 | 4.7/4.3 | 2.7 | 17/1/2 | 53/47/0 | — |
| Balkany et al, 1999 d | 5.7/2.2 | — | — | — | — |
| Miyamoto et al, 1997 | -/6.3 | — | — | — | — |
| Parisier et al, 1996 | -/14.9 | — | — | — | 100 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


