


Figure 27.1 Surgical technique of RPE–choroid transplantation. A. Delimitation with laser at the site which the graft is taking. B. The graft is gripped on the choroidal surface with an aspirating cannula. C. The patch is held with forceps, and the graft is gently pulled to the subfoveal area. D. A perfluorocarbon bubble is used to hold the graft to ensure correct unfolding and a subfoveal position.
The graft is gripped on the choroidal surface with an aspirating cannula and then slid through the original retinotomy into the subfoveal space (Fig. 27.1B). The patch is held with forceps, and the graft is gently pulled to the subfoveal area (Fig. 27.1C). A perfluorocarbon bubble is used to hold the graft to ensure correct unfolding and a subfoveal position (Fig. 27.1D). Retinopexy laser is not applied to the macular retinotomy, and the eye is filled with silicone oil or C3F8.
In another technique, the surgeon creates a retinal detachment of temporal retina by injecting a balanced salt solution into the subretinal space through a 42-gauge subretinal cannula; this retinal detachment includes the macula and involves repeated fluid air exchanges (Fig. 27.2A and B). A 180-degree temporal retinotomy is performed with curved scissors, as close as possible to the ora serrate (Fig. 27.2C and D). The retina is folded on itself exposing the subretinal space, facilitating to remove the RPE damage and the CNV. The bleeding from the choroidal feeder vessels of the CNV is stopped with gentle pressure or diathermy. A full-thickness patch of choroid, choriocapillaris, BM, and RPE is isolated from the midperiphery. First the graft is delineated with three intense crowns of diode laser. After demarcation of a rectangular excision area helped by the use of diathermy, the graft is subsequently cut out as a full-thickness RPE choroid graft after peeling off the overlying neurosensory retina. When the patch is fully cut to 360 degrees, choroidal bleeding was controlled by diathermy or intraocular pressure elevation (Fig. 27.2E). The graft is gently pulled to the subfoveal area. During this maneuver, the patch is held with forceps at its anterior edge, in order to prevent damaging it with extensive manipulation. In order to assure adequate adhesion between the patch and the posterior pole, the temporal retina is flipped over its original position, and the perfluorocarbon liquid (PFCL) is injected into the preretinal space to reattach the retina. Peripheral laser endophotocoagulation is performed at the edge of the retinotomy being careful to avoid the pigment epithelium. The eye is filled with silicone oil. This technique facilitates the manipulation and implantation of the graft, less cell loss of RPE, and better centering. However, intraoperative and postoperative complications are more likely. Proliferative vitreoretinopathy is the most frequent complication, affecting visual outcome irremediably.





Figure 27.2 Alternative autologous RPE–choroid transplantation technique. A. After vitrectomy, we perform a controlled retinal detachment with BSS in the temporal sector. B. Exchange liquid air to increase the macular detachment. C. A 180-degree temporal retinotomy is performed as close as possible to the ora serrata. D. The temporal retina is overlying the nasal retina and the graft is taking. E. When the patch is fully cut to 360°, choroidal bleeding was controlled by diathermy or intraocular pressure elevation.
Anatomical and Functional Outcome
Following autologous RPE–choroid graft transplant, the graft has a brown appearance. Unfortunately, the graft can fold over (Fig. 27.3) or become a fibrotic patch. Sometimes an irregular accumulation of hyperpigmentation at the margins, near the graft or on the graft, can be seen.
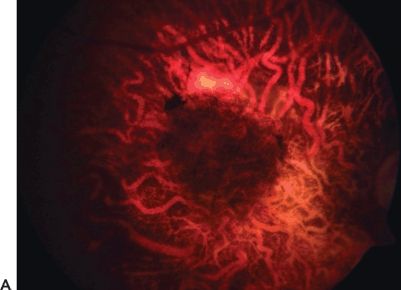


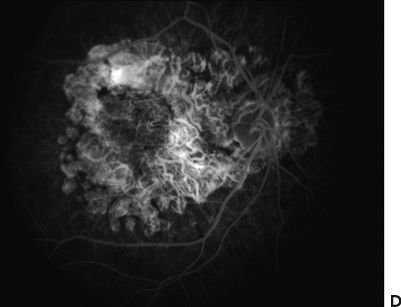
FIGURE 27.3 A. Follow-up of a patient presenting after autologous translocation of the RPE and choroid in geographic atrophy. B–D. The graft showed good vascularization.
Angiographic patterns in patients with revascularization of the graft (Fig. 27.4) may be observed if the large choroidal vessels of the graft are visible. The revascularization pattern of the grafts suggests connection of the choroidal channels of the transplant with those of the underlying recipient choroid. There are different revascularization patterns. Most patients will have revascularization of the graft as early as 3 weeks after translocation.

Figure 27.4 Optic coherent tomography shows the RPE–choroid complex folded and the presence of an epiretinal membrane after 3 months to perform a surgical procedure.
If the graft is not rejected, the OCT imaging shows an intact outer nuclear layer, an external limiting membrane, and a junction between photoreceptor inner and outer segments, indicating the presence of intact photoreceptors (Figs. 27.5 and 27.6). In the rejected graft, a photoreceptor layer cannot be identified. A late postoperative complication is the development of classic or occult CNV.



Figure 27.5 Autologous RPE–choroid transplantation OCT of the left eye. A. The picture shows the folding complex placed in the macula. B, C. OCT shows integration of the graft to the original choroid.


FIGURE 27.6 OCT imaging (A) and (B) 12 months after autologous RPE–choroid transplant. This patient has improvement in visual acuity from 5/200 to 20/200.
In regard of the functional outcome, the visual acuity must be carefully assessed before and after surgery. Relevant studies in which visual acuities were evaluated are summarized in Table 27.1. There is a disparity in the improvement of visual acuity between research studies. One factor for the variability in outcomes may be variability in the degree and type of disease, including CNV, geographic atrophy, and/or nonresponders. Moreover, the type of graft is another factor to consider. If it is allogenic, this is accompanied by the risk of immunosuppression. Another possible explanation for the visual loss after RPE–choroid patch transplant is an inadequate positioning of the graft. Even though a functioning and intact RPE cell layer is transplanted, patients with initial extrafoveal fixation may have no benefit from the patch. Thus, preoperative microperimetry and spectral domain OCT is essential to select patients with central fixation and intact photoreceptor.
Table 27.1 SUMMARY OF RETINAL TRANSPLANTATION STUDIES

RD, retinal detachment; PVR, proliferative vitreoretinopathy; EM, epiretinal membrane.
On the other hand, the neurosensory retina of a patient with geographic atrophy is more adherent to the choroid compared to exudative AMD. Separating the macula intraoperatively from the choroid may damage the outer retina irreversibly; even a macular hole can be formed. Another cause of failure to improve visual acuity is nonperfusion of the graft.
Finally, it is worthwhile to mention that surgical skill is an important variable. During the surgery, complications may occur, such as trauma of the subfoveal choroid with bleeding, tearing of the retinotomy with bleeding, a peripheral retinal tear, a retinal detachment, failure of the subfoveal release of the graft, and an inadequate positioning or incomplete flattening of the graft, which was partly folded or wrinkled underneath the fovea. An important postoperative complication is proliferative vitreoretinopathy with a high risk of retinal detachment. The excision of peripheral full-thickness retina and choroid generates inflammation, which probably stimulates proliferative vitreoretinopathy. Recurrent CNV membrane, rhegmatogenous retinal detachment, postoperative hemorrhages, acute glaucoma, and retinal puckering/cellophane maculopathy (Fig. 27.3) are other causes of poor prognosis.
Future perspectives
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


