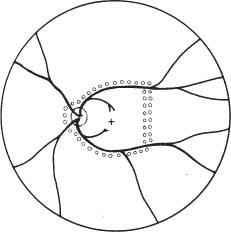
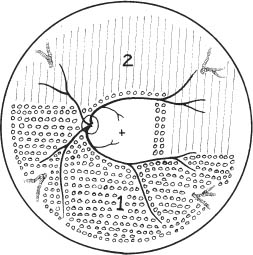
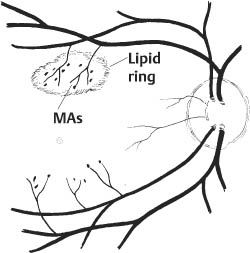
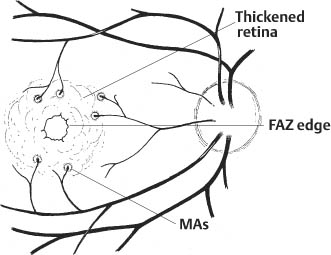
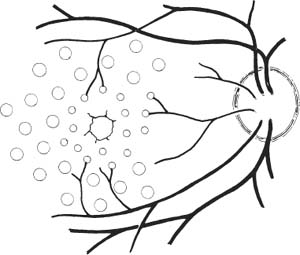
67 1. Diabetic retinopathy. a. The Early Treatment Diabetic Retinopathy Study (ETDRS) has shown that panretinal photocoagulation (PRP) decreases the incidence of severe visual loss in patients with “high risk” proliferative diabetic retinopathy (i.e., risk of severe visual loss was ~50% over 5 years among untreated patients versus ~25% among treated patients). “Severe visual loss” is defined as visual acuity < 5/200 on two consecutive visits separated by 3 months. The following clinical features define high risk proliferative diabetic retinopathy (HRPDR): i. Neovascularization at or within 1 disc diameter of the optic disc (NVD) that is larger than NVD in a standard photograph (~1/4 –1/3 disc area in size). ii. Any NVD in the presence of vitreous or preretinal hemorrhage. iii. Moderate to severe neovascularization elsewhere (NVE) (i.e., neovascularization ≥1/2 disc diameter in size), in the presence of vitreous or preretinal hemorrhage. b. PRP should probably be offered to most type II diabetic patients with PDR even in the absence of “high risk” characteristics. c. PRP is somewhat beneficial for patients who have severe nonproliferative diabetic retinopathy (NPDR). One should consider offering PRP to such patients if they cannot be followed properly or if the fellow eye has been lost due to complications of PDR. It may be offered to patients with bilateral severe NPDR. PRP should probably be deferred in patients with clinically significant macular edema (CSME) and severe NPDR until either high risk features develop or the macular edema resolves (e.g., in response to focal macular laser photocoagulation). Severe NPDR can be defined using the “4-2-1” rule: i. 4 quadrants of “severe” intraretinal hemorrhage, or ii. 2 quadrants of “severe” venous beading, or iii. 1 quadrant of “severe” intraretinal microvascular abnormalities (IRMAs). iv. “Severe” is defined as a degree of abnormality greater than or equal to that present in standard photographs used during the ETDRS. d. Rubeosis iridis even in the absence of neovascularization of the posterior pole. This occurs uncommonly but can be seen particularly after pars plana vitrectomy or in the setting of complete posterior vitreous detachment. 2. Retinal branch vein occlusion. a. The Branch Vein Occlusion Study showed peripheral scatter photocoagulation is effective in: i. Decreasing the incidence of neovascularization following retinal branch vein occlusion and ii. Decreasing the incidence of vitreous hemorrhage (VH) in patients who have developed retinal neovascularization (30% incidence among treated patients versus 60% among controls). b. The study investigators recommended that patients without NV should be followed until new blood vessels develop, at which point they should be treated. Ap proximately 40% of patients with areas of retinal capillary nonperfusion ≥ 5 disc diameters in size develop NV. Among patients with NV, ~60% develop VH. Among patients with VH, ~12% develop a loss of 5 Snellen lines of vision. Thus, scatter photocoagulation is not recommended until NV develops. c. Note: Laser treatment of retinal branch vein occlusion is placed only in the area drained by the occluded vein. This does not involve four-quadrant scatter photocoagulation such as is applied in the treatment of PDR. Generally, treatment is applied no closer than 2 disc diameters away from the edge of the foveal avascular zone. 3. Central retinal vein occlusion (CRVO) with rubeosis iridis or with retinal NV and VH. 4. Sickle cell retinopathy: Scatter photocoagulation applied in the quadrant around areas of peripheral retinal NV is the preferred method to treatment patients with recurrent vitreous hemorrhage and/or retinal neovascularization secondary to proliferative sickle cell retinopathy (most common in patients with SC disease). One should avoid direct treatment over the long posterior ciliary arteries to reduce the chance for anterior segment ischemia. 5. Radiation retinopathy: Radiation can damage retinal vascular cells and produce a retinal microangiopathy that resembles diabetic retinopathy clinically. Usually, patients must be exposed to radiation doses of 35 Gy or more for retinopathy to develop. Concurrent administration of chemotherapy or coexisting disease such as diabetes mellitus lowers the threshold for the development of radiation retinopathy. To treat proliferative radiation retinopathy, scatter photocoagulation is applied as described for PDR. 1. Complete retinal examination to evaluate presence of NV and retinal ischemia. 2. Fluorescein angiogram to evaluate capillary dropout (in cases of retinal vein occlusion or rubeosis iridis secondary to PDR without posterior pole retinal neovascularization) and to distinguish venous collaterals (BRVO) and IRMA (diabetic retinopathy) from NV. 1. Anesthesia: Topical (e.g., proparacaine) is preferred. Retrobulbar anesthesia may be necessary in a patient who has experienced excessive pain on previous treatment or who is unable to maintain steady fixation. If retrobulbar anesthesia is administered, the surgeon should be prepared to manage infrequent systemic complications such as respiratory depression and seizures. 2. Place contact lens with methylcellulose solution. 3. Establish clear view of fundus and identify landmarks. 4. Focus aiming beam, tilting the contact lens as necessary to produce a round, not elliptical, spot. 1. Spot size: a. 500 μm (set laser at 200–300 μm if using Rodenstock, quadraspheric, or equator plus lens). b. 200 μm if treating within vascular arcades. 2. Duration: 0.05–0.2 seconds. Short durations (e.g., 0.05 second) tend to be associated with less discomfort. 3. Power: Start at ~150 mW (100 mW for krypton) and adjust as necessary to produce desired effect. 4. Typical treatment schema: a. Number of laser applications: i. PRP for PDR: Apply ~1500 spots over two sessions. It is probably best not to focus on the number of treatment spots but to focus on the total surface area treated. The usual goal is to treat the retina outside the temporal arcades as far peripherally as the equator for 360 degrees. ii. PRP for CRVO: Apply treatment as above. Patients with rubeosis iridis may require treatment extending to the ora serrata. iii. Scatter photocoagulation for BRVO: Treatment is confined to the quadrant involved by the vein occlusion. iv. Scatter photocoagulation for sickle cell retinopathy: Treatment is applied surrounding areas of peripheral retinal NV out to the ora serrata and for 1 clock hour on either side of the NV. For patients in whom follow-up is uncertain, one may elect to treat the periphery for 360 degrees. One should avoid direct treatment to long posterior ciliary arteries to reduce the chance of anterior segment ischemia. v. Total number of spots and density of burns varies with clinical response to treatment. I. Clinical response to treatment typically requires ~3 weeks. II. Clinical response is evident as a loss of the capillary brush border of the NV, the development of fibrosis of the NV, and/or as regression of the NV. vi. Separate treatment sessions bŶ1–2 weeks. b. Space spots ½ –1 burn width from each other. c. Some surgeons apply test burns in peripheral area of relatively normal retina to obtain baseline power for treatment. d. Adjust laser power to produce a gray-white (not intense white) burn. e. Session 1: Figure 67.1 i. Circumscribe area not to be treated with 1–2 rows of spots (Fig. 67.1). I. Do not treat papillomacular bundle. II. Do not treat closer than one third disc diameter to optic nerve head nasally. III. Usually avoid treatment within vascular arcades temporally (may place a few spots just inside arcades if they are greater than 2 disc diameters from the edge of the foveal avascular zone). IV. Temporally: place 1–2 rows of burns, no closer than 2 disc diameters from the edge of the foveal avascular zone (assuming a relatively normal sized foveal avascular zone of 300–500 μm is present). Do not treat nasal to this demarcation. ii. Select a section of retina for treatment (i.e., inferior fundus at first session [especially if vitreous hemorrhage is present]; superior half of fundus at second treatment session). I. Apply enough evenly scattered spots to cover the target area. II. Vary power as needed to obtain uniform burn intensity. III. If Rodenstock or transequator lens is used, may switch to Goldmann or quadraspheric lens to reach far periphery if necessary. IV. To facilitate treatment of peripheral retina, have patient look away from the mirror of a Gold-mann lens or in the direction of the quadrant to be treated with a Rodenstock and quadraspheric lens. f. Subsequent sessions: i. Treat previously untreated regions. ii. May treat more peripherally in previously treated quadrants. Figure 67.2 iii. An example of one possible multiple-session treatment sequence is illustrated (Fig. 67.2). g. Treatment tips: i. May directly treat small areas of flat NVE with confluent application of laser burns if they do not regress in response to scatter (i.e., indirect) treatment. ii. Do not treat NVD directly. iii. Do not treat areas of fibrovascular traction or elevated retina (to avoid inducing retinal breaks). iv. Do not treat directly over retinal vessels (to avoid inducing vascular occlusion). v. If patient is uncomfortable (e.g., when treating horizontal meridian near ciliary nerves), decrease laser duration and power. vi. Repeatedly verify landmarks and aiming beam position to avoid inadvertently treating the macula. 1. Usually prescribe oral analgesic (e.g., Tylenol, 650 mg p.o.) for headache. Some surgeons treat with topical atro-pine 1% b.i.d. and prednisolone acetate 1% q.i.d. after extensive treatment (e.g., entire fundus in cases of rubeotic glaucoma) to prevent development of secondary angle closure due to laser-induced choroidal detachment. For treatments involving fewer spots, it may be reasonable to prescribe oral analgesic only. 2. Follow-up in ñ1 week. 1. Inadvertent foveal burn 2. Vitreous hemorrhage 3. Choroidal effusion with possibility of secondary angle closure glaucoma 4. Macular edema with possible permanent decrease in visual acuity 5. Epiretinal membrane formation 6. Retinal tear formation 8. Constriction of visual field (usually with extensive scatter photocoagulation) 9. Decreased dark adaptation 10. Decreased accommodative amplitude Treat eyes with retinal thickening involving or threatening the center of the macula (clinically significant macular edema [CSME] as defined by the ETDRS). CSME is defined as: 1. Thickening of the retina at or within 500 μm of the center of the macula, or 2. Hard exudates at or within 500 μm of the center of the macula associated with adjacent areas of retinal thickening, or 3. Areas of retinal thickening one disc diameter or larger any part of which is within one disc diameter of the center of the macula. 4. Macular edema is defined by the presence of retinal thickening on clinical exam. Dye leakage seen on fluorescein angiography may or may not indicate retinal thickening, depending on how well the RPE and retina clear the fluid delivered to the retina via incompetent retinal vessels. Also, areas of nonperfused retina may be thickened. Optical coherence tomography (OCT) detects macular edema more sensitively than clinical exam. One study showed that clinicians cannot reliably detect thickened retina with contact lens biomicroscopy unless the thickness is > 300 μm. OCT-3 can detect thickness of 200–300 μm reliably. Use of OCT measurements to guide treatment recommendations for CSME is not standardized at this time. 5. Given a patient with CSME the ETDRS defines treatable lesions as: a. Discrete points of hyperfluorescence or leakage on the fluorescein angiogram (e.g., microaneurysms [MAs], IRMAs, capillaries). b. Diffuse sites of leakage within the retina (e.g., leaking capillary bed). c. Thickened avascular areas of retina. 6. Among untreated patients, moderate visual loss (doubling of visual angle or a 3-line loss of vision on the ETDRS vision chart) occurred in ~24% during 3 years of follow-up versus ~12% who underwent macular photocoagulation. a. The treatment benefit was most marked for patients with thickening involving the macular center. b. Only ~3% of patients experienced moderately improved visual acuity after treatment, so the major goal of treatment is to stabilize vision, not to improve it. The Branch Vein Occlusion Study concluded that eyes with visual acuity of 20/40 or less secondary to macular edema benefit from photocoagulation to the involved area. 1. Patients are eligible for treatment if: a. Visual acuity is 20/40 or worse and not spontaneously improving, and b. BRVO is at least 3 months old, and c. Decreased vision is not due to blood in the fovea and/or macular capillary nonperfusion, and d. Fluorescein angiography documents fluorescein leakage in the macula. 2. Among untreated patients, after 3 years of follow-up, 34% had visual acuity ≥ 20/40 versus 60% of treated patients. 1. Perform stereoscopic contact lens fundus exam to evaluate location and extent of retinal thickening from macular edema. 2. Perform fluorescein angiogram and color fundus photos to precisely identify areas of focal and diffuse leakage as well as areas of macular capillary nonperfusion. 3. Perform baseline OCT exam to document severity of macular thickening and to serve as comparison for future measurements after treatment (optional). 1. Project recent representative fluorescein angiogram for reference during treatment. Figure 67.3 a. In some cases (e.g., pregnancy, allergy to fluorescein dye), a fluorescein angiogram cannot be done. In this situation, if there is a circinate lipid ring, the leaking lesions are almost always located in the center of the lipid ring, and treatment can be directed to this area (Fig. 67.3). If there is no lipid ring, grid treatment can be considered, but the patient must be cautioned that the treatment may be placed too close to the edge of or even within the foveal avascular zone (FAZ) inadvertently with resultant symptomatic scotomata or loss of central visual acuity. This situation is perhaps more likely to arise if there is pathological enlargement of the FAZ. 2. Anesthesia: Topical (e.g., proparacaine) is preferred. Retrobulbar anesthesia may be necessary in a patient who has experienced excessive pain on previous treatment (rare in this setting) or who is unable to maintain steady fixation. a. If retrobulbar anesthesia is administered, the surgeon should be prepared to manage infrequent systemic complications such as respiratory depression and seizures. b. Also, after a perfect retrobulbar injection, some torsional eye movement is possible because the superior oblique muscle will not be paralyzed by the injection. 3. Place fundus contact lens with methylcellulose solution. 4. Establish clear view of fundus and identify landmarks. 5. Focus aiming beam, tilting the contact lens as necessary to produce a round, not elliptical, spot. 6. Treat focal areas of leakage. a. Use argon green or dye yellow laser. b. Treat all discrete points of hyperfluorescence or leakage that lie within 2500 μm of the center of the macula as determined with the angiogram. One may also treat thickened retina that is nonperfused. Treatment of nonleaking lesions (e.g., MAs versus intra-retinal hemorrhage) less than 125 μm in diameter is optional. c. Do not treat lesions closer than 500 μm to center of the macula during the initial treatment. d. Do not treat the edge of the FAZ. e. If retinal thickening involves the center of the macula and visual acuity is 20/40 or better, treat leaks no closer than 500 μm to the macular center at the initial treatment. After ~3 months follow-up, if CSME persists and if vision is 20/40 or better, consider treatment of leaking lesions located 300–500 μm from the center of the macula provided that the treatment spares the edge of the FAZ and patient understands and accepts the risk of developing paracentral scotomata or central visual loss. f. One may treat intraretinal hemorrhages ≤ 125 μm in diameter that lie 1500 μm or more from the macular center and outside the papillomacular bundle. g. Do not treat flame-shaped hemorrhages. 7. Laser parameters: a. Spot size: 50–100 μm. i. Recommend 50 μm spot when treating 300–500 μm from center of FAZ. ii. Recommend 100 μm spot when treating > 500 μm from center of FAZ. b. Duration: 0.05–0.1 seconds. Consider using 0.05s when treating close to the edge of FAZ to avoid inadvertent treatment of foveola due to saccade. c. Power: Start at ~100 mW and increase as necessary to produce mild whitening of the RPE. 8. Patterns of focal treatment: Direct versus grid laser photocoagulation. Figure 67.4 a. Direct treatment: Laser spot is targeted only to leaking lesions in thickened retina (Fig. 67.4). i. Size: 50–100 μm spot. ii. Duration: 0.05–0.1 second iii. Power: enough to create mild whitening of RPE under treated lesion. When treating MAs ≥ 40 μm in size, may consider increasing the power enough to whiten the MA, although it is not clear that this intensity of treatment is required to elicit a clinical response. iv. When treating with the goal of whitening MAs, consider first treating the subjacent RPE with a 100 μm spot to produce mild RPE whitening. This may reduce the power needed to create whitening of the MA. Figure 67.5 b. Grid treatment: Laser spots are applied in a “grid” pattern in areas of retinal thickening without any specific attempt to treat leaking lesions (Fig. 67.5). i. Size: 50–200 μm (usually, 100 μm). ii. Spacing: 1–2 burn widths apart depending on the degree of retinal thickening (close spacing for marked thickening). iii. Duration: 0.05–0.1 sec (usually 0.1 sec). iv. Power: enough to create mild whitening of RPE. v. Distribution: above, below, and temporal to fovea. Extend treatment to PRP burns, if present. vi. Treat conservatively in the papillomacular bundle and use 50 μm spots. vii. Stay at least 500 μm from macular center and from margin of optic nerve head. 9. Treatment tips:
Retina Laser Procedures
 Panretinal Photocoagulation
Panretinal Photocoagulation
Indications
Preoperative Procedure
Instrumentation
 Argon green (514–527 nm), krypton red (647 nm), tunable dye (577–630 nm), or diode (790–830 nm) laser. The longer wavelengths (e.g., krypton red and diode modalities) are especially useful for eyes with nuclear sclerotic cataracts or vitreous/preretinal hemorrhage.
Argon green (514–527 nm), krypton red (647 nm), tunable dye (577–630 nm), or diode (790–830 nm) laser. The longer wavelengths (e.g., krypton red and diode modalities) are especially useful for eyes with nuclear sclerotic cataracts or vitreous/preretinal hemorrhage.
 Fundus contact lens.
Fundus contact lens.
 Goldmann 3-mirror lens:
Goldmann 3-mirror lens:
 Image is oriented with same spatial relationships as retina undergoing treatment.
Image is oriented with same spatial relationships as retina undergoing treatment.
 May use mirrors to treat peripheral fundus.
May use mirrors to treat peripheral fundus.
 Burn size = spot size set on laser.
Burn size = spot size set on laser.
 Rodenstock, quadraspheric, or transequator lenses:
Rodenstock, quadraspheric, or transequator lenses:
 Image is inverted and backward (similar to image obtained with indirect ophthalmoscope).
Image is inverted and backward (similar to image obtained with indirect ophthalmoscope).
 A much larger area in a given field may be treated than with the Goldmann lens.
A much larger area in a given field may be treated than with the Goldmann lens.
 Burn size ≅ approximately twice the spot size set on laser (e.g., 250 μm setting gives approximately a 500 μm spot size).
Burn size ≅ approximately twice the spot size set on laser (e.g., 250 μm setting gives approximately a 500 μm spot size).
Operative Procedure
Laser Parameters
Postoperative Procedure
Complications
 Laser Photocoagulation for Macular Edema
Laser Photocoagulation for Macular Edema
Indications
Macular Edema in Diabetic Retinopathy
Macular Edema in Branch Retinal Vein Occlusion
Preoperative Procedure
Instrumentation
 Argon green (514–527 nm), krypton red (647 nm), tunable dye (577–630 nm), or diode laser (790–830 nm):
Argon green (514–527 nm), krypton red (647 nm), tunable dye (577–630 nm), or diode laser (790–830 nm):
 Some surgeons prefer argon green or dye yellow for discrete areas of vascular leakage (e.g., MAs and IRMA) since these wavelengths are absorbed well by hemoglobin.
Some surgeons prefer argon green or dye yellow for discrete areas of vascular leakage (e.g., MAs and IRMA) since these wavelengths are absorbed well by hemoglobin.
 Krypton red, dye red, and diode laser may be preferred for macular grid treatments since these wavelengths are not absorbed well by retinal blood vessels or macular xanthophyll pigment, thus sparing (relatively) the overlying retina. Although these wavelengths may also be advantageous in situations where blood partially obscures the planned treatment area, one should not treat over blood in this setting, as it may stimulate epiretinal membrane formation or produce undesirably intense retinal burns.
Krypton red, dye red, and diode laser may be preferred for macular grid treatments since these wavelengths are not absorbed well by retinal blood vessels or macular xanthophyll pigment, thus sparing (relatively) the overlying retina. Although these wavelengths may also be advantageous in situations where blood partially obscures the planned treatment area, one should not treat over blood in this setting, as it may stimulate epiretinal membrane formation or produce undesirably intense retinal burns.
 Fundus contact lens:
Fundus contact lens:
 Goldmann three-mirror lens or macular contact lens:
Goldmann three-mirror lens or macular contact lens:
 Image is oriented with same spatial relationships as eye undergoing treatment.
Image is oriented with same spatial relationships as eye undergoing treatment.
 Burn size = spot size set on laser.
Burn size = spot size set on laser.
 Rodenstock lens: probably should be avoided for macular treatments due to image inversion and relative minification of view.
Rodenstock lens: probably should be avoided for macular treatments due to image inversion and relative minification of view.
Operative Procedure
Treatment of Diabetic Macular Edema
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree