Purpose
Immunologic rejection remains a major cause of graft failure in high-risk corneal transplantation. This study was conducted to elucidate the efficacy and safety of systemic cyclosporine (CsA) in high-risk corneal transplantation.
Design
Prospective, randomized, open-labeled clinical trial with a parallel-group study.
Methods
Patients underwent high-risk corneal transplantation at the Department of Ophthalmology, Tokyo Dental College, Chiba, Japan. High-risk was defined as corneal neovascularization in more than 1 quadrant or a history of corneal grafting. Patients were assigned to either a systemic CsA group or a control group. Administration of CsA was continued for at least 6 months with blood CsA concentration 2 hours after administration of approximately 800 ng/mL, unless undesirable side effects developed. The main outcome measures were graft clarity, endothelial rejection, and local and systemic complications.
Results
Forty patients were enrolled and 39 (18 men, 21 women; mean age, 67.4 ± 11.9 years) were analyzed. In the CsA group, CsA was discontinued within 6 months in 7 patients because of side effects. With a mean follow-up of 42.7 months, endothelial rejection developed in 6 and 2 eyes in the CsA and control groups, respectively. No differences were observed in the rates of graft clarity loss between the 2 groups ( P = .16, Kaplan-Meier analysis).
Conclusions
No positive effect of systemic CsA administration for suppressing rejection in high-risk corneal transplantation was observed. With a relatively high incidence of systemic side effects, the results suggest that this protocol should not be recommended for corneal transplant recipients, especially those of advanced age.
Corneal transplantation is the most widely performed transplant and now has more than a century of history. Among the various surgical procedures available for corneal transplantation, penetrating keratoplasty (PK) is the most commonly performed one for opaque or distorted corneas, and outcomes with this procedure have been favorable. However, immunologic rejection remains a major cause of graft failure with PK, particularly in patients at high risk for rejection, who account for approximately 60% of PK cases. A history of PK (regrafting), deep neovascularization, or both are believed to be factors contributing to rejection.
There are 2 approaches to avoid immunologic rejection: use of histologically compatible donor corneas and immunosuppressive therapy. Numerous studies have investigated outcomes in terms of graft survival with human leukocyte antigen (HLA)-matched tissues, but the results have been variable: a beneficial effect has been observed with either HLA class I-matched or HLA class II-matched grafts in some studies, but not in others. The results of the Collaborative Corneal Transplantation Study indicate that the use of blood group-matched tissues had a positive effect on graft survival, although the benefit seems to have been relatively small.
Immunosuppressive therapy has been investigated over a long period. Although topical corticosteroids are the gold standard for post-PK regimens, the results seem to be unsatisfactory in high-risk cases. Systemic corticosteroids also can be used in some cases. However, the long-term use of systemic corticosteroids is not recommended because of possible local and systemic side effects.
Cyclosporine A (CsA) is a potent immunosuppressant that has shown a positive effect in transplantation of a number of organs, including the kidney and heart. Reports on its effectiveness in corneal transplantation, however, have been contradictory. Moreover, most of these studies have been retrospective, with various disease backgrounds and postoperative follow-up times. The recipients of corneal transplants tend to be older than other transplant recipients, which makes the influence of general health a potential concern.
In the current study, we tested the hypothesis that systemic CsA can reduce the incidence of immunologic rejection and hence can improve graft survival after high-risk corneal transplantation. A prospective, randomized trial was conducted to elucidate the effectiveness and safety of this treatment.
Methods
Study Design and Participants
This was an open-labeled, parallel-group study conducted in a single referral hospital. Eligible participants were all adults aged 20 years of age or older who underwent high-risk corneal transplantation at the Department of Ophthalmology, Tokyo Dental College. Patients with deep neovascularization in more than 1 quadrant, a history of corneal transplantation (regrafting), or both were considered to be at high risk for rejection of a transplanted corneal graft. Patients with poorly controlled diabetes mellitus and hypertension; abnormal kidney or liver function, or both; or active infectious diseases were excluded from the study. Patients with eyes showing signs of active infection or an uncontrolled increase in intraocular pressure also were excluded.
Patients were assigned randomly to receive postoperative medication with systemic CsA (CsA group) or no postoperative systemic CsA (control group). Randomization was performed based on a computer-generated randomization list that was kept by an independent examiner. The primary end point for efficacy of CsA administration was the proportion of eyes without endothelial rejection after corneal transplantation. Eyes with clear grafts also were analyzed. The incidence of local or systemic side effects also was a study outcome.
The sample size of 20 eyes in each group was chosen to provide at least 80% power to detect a 50% difference in rejection rates at a 1-sided a error level of 0.05. This assumption was made based on previous reports that had used a protocol similar to ours, in which a difference of approximately 100% was noted between patients receiving CsA for 4 to 6 months and the control group.
Corneal Transplantation and Postoperative Treatment
All donor corneas were obtained from eye banks in the United States, and all met the donor quality criteria of the Eye Bank Association of America. No HLA matching was performed. During surgery, 0.25- to 0.50-mm oversized donor corneas were punched out using the Barron donor punch (Katena Products, Inc, Denville, New Jersey, USA) and secured to the recipient’s eye, after excision of the abnormal host cornea using the Hessbarg–Barron trephine (Katena). Single, continuous or interrupted 10-0 nylon sutures were used. Subconjunctival betamethasone (2 mg) was given at the completion of transplantation. Extended-wear soft contact lenses were put in place.
After surgery, topical corticosteroids such as 0.1% dexamethasone eye drops (Sanbetason; Santen Pharmaceutical Co, Osaka, Japan) were started 5 times daily in all cases. No topical CsA was given in any case. Prophylactic antibiotic eye drops (0.1% levofloxacin [Cravit]; Santen Pharmaceutical) also were administered in both groups and were tapered over a period of a few months. The steroid eye drops, however, were continued unless infection or an uncontrollable increase in intraocular pressure occurred. Systemic corticosteroids also were administered in the CsA group, starting with 8 mg/day betamethasone, which was tapered over the next 21 days.
Protocol for Cyclosporin A Administration
CsA was administered intravenously at 3 mg/kg daily, starting on the day of corneal transplantation (day 0) until day 6. On day 7, this was changed to oral administration at 5 mg/kg daily. Starting on day 7, the blood CsA concentration 2 hours after administration (C2 level) was determined weekly for the first month and then monthly thereafter. We used the C2 level for CsA monitoring because C2 levels were shown to be highly predictive of acute rejection in de novo renal patients, compared with the conventional trough level. Doses of CsA were adjusted according to the results. The C2 level was maintained at 800 to 1000 ng/mL for the first 3 months and then gradually reduced to a maintenance level of 600 to 800 ng/mL. The dose of CsA was reduced by half when creatinine or blood urea nitrogen levels increased to 50% more than preoperative values. CsA administration was discontinued when local or systemic side effects developed or at the patient’s request. Unless undesirable consequences developed, CsA was continued for 6 months after surgery and for a further 6 months, provided it was well tolerated and the patient agreed to continuation of treatment.
Clinical Assessment
The patients were examined daily for the first 2 weeks after surgery and then were monitored every 2 to 4 weeks. The primary outcome of the study was postoperative endothelial rejection, and the secondary outcome was the safety of CsA administration. We assessed the symptoms, graft condition (using slit-lamp biomicroscopy), corrected visual acuity, intraocular pressure (by either applanation tonometry or Tono-Pen XL Applanation Tonometer; Reichert Technologies, Depew, New York, USA), and other local or systemic complications. The Snellen chart was used to determine corrected visual acuity, and the decimal values were converted to logarithm of the minimal angle of resolution units for statistical analysis. Finger counting, hand movements, and light perception were converted to 0.004, 0.002, and 0.001, respectively, for analysis. All patients were followed up for at least 2 years after PK.
Criteria and Treatment of Endothelial Rejection
Endothelial rejection was defined as 1 of the following: (1) development of a rejection line; (2) a new anterior chamber reaction with keratoprecipitates in the operated eye; or (3) an acute increase in corneal stromal edema in a previously clear graft. Rejection episodes were treated with frequent use of 0.1% dexamethasone and systemic administration of corticosteroids (dexamethasone, tapering from 8 mg/day; or 500 mg/day methylprednisolone for 3 days, followed by oral dexamethasone). The blood CsA level was checked in patients receiving CsA treatment, and the dose was increased when the C2 level was less than 800 ng/mL.
Statistical Analysis
Age, gender, and other preoperative factors were compared between the CsA and control groups using a chi-square analysis with the Fisher exact test. The Student t test was used to compare continuous variables between the CsA and control groups. Kaplan–Meier plots of the estimated probabilities over time were constructed and compared between the CsA and control groups using the Fisher exact test.
Results
Patient Profile
One case in the control group was excluded from the analysis because of early dropout during follow-up. As a result, 39 eyes in 39 patients (20 in the CsA group and 19 in the control group) were analyzed. The patients comprised 18 men and 21 women, with a mean age of 67.3 ± 11.7 years (range, 37 to 88 years). Table 1 shows the demographic data of the patients. Most of the patients were considered to be at high risk because of a history of failed grafts, and more than half of the eyes showed deep corneal neovascularization in more than 1 quadrant. No significant differences were observed between the 2 groups in terms of patient profile, except that the mean age was slightly lower in the CsA group ( P = .047).
| Characteristic | CsA Group (n = 20) | Control Group (n = 19) | P Value |
|---|---|---|---|
| Male-to-female ratio | 9:11 | 9:10 | .88 |
| Age (yrs) | 63.7 ± 13.0 | 71.1 ± 9.0 | .047 |
| Mean quadrants of NV | 2.16 | 2.06 | .86 |
| More than 2 quadrants of NV | 11 (55%) | 11 (58%) | 1.00 |
| Original disease (eyes) | .22 | ||
| BK | 12 | 10 | |
| Scar | 3 | 7 | |
| Herpes | 1 | 1 | |
| Dystrophy | 4 | 0 | |
| Other | 1 | 1 | |
| Mean no. of previous grafts | 1.30 (1 to 2) | 1.21 (1 to 3) | .52 |
| Surgical method | .69 | ||
| PKP only | 17 | 15 | |
| Combined cataract surgery | 3 | 4 | |
| CVA at presentation | 0.015 | 0.0094 | .44 |
| Mean follow-up (mos) | 48.8 ± 20.0 | 36.8 ± 19.5 | .052 |
Tolerance and Side Effects of Cyclosporin A
In the CsA group, the initial 6-month CsA administration schedule was completed in 13 (65.0%) of 20 patients. The administration protocol was not completed in 7 patients, and in 3 of these, CsA was discontinued within 1 month after surgery. The administration of CsA was discontinued in 4 of these 7 patients owing to systemic side effects, including eczema, back pain, chest pain, and nausea (1 each). In 2 other patients, CsA was discontinued at the patient’s request, and in 1 case, CsA was discontinued on the development of infectious keratitis ( Candida keratitis) at 1 week after surgery ( Table 2 ). In all cases in which CsA was discontinued, local corticosteroid eye drops were continued. In 1 other patient (a 74-year-old man), a decrease in kidney function developed at 6 months after surgery, which necessitated discontinuation of CsA. Thus, CsA was discontinued because of systemic side effects in 25.0% (5/20) of the cases.
| Case No. | Age, Gender | Reason | Timing | Rejection (Timing) |
|---|---|---|---|---|
| 2 | 70, F | Nausea | 3 mos | No |
| 10 | 52, M | Eczema | 5 mos | No |
| 11 | 71, F | Infection ( Candida keratitis) | 1 wk | No |
| 13 | 59, F | Chest pain | 1 day | Yes (5 mos) |
| 14 | 38, M | Back pain | 1 day | Yes (20 mos) |
| 18 | 76, M | Patient request | 1.5 mos | No |
| 20 | 65, M | Patient request | 3 mos | Yes (12 mos) |
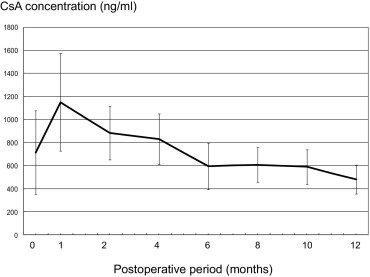
Figure 1 shows the mean blood concentration of CsA (C2 level) during the observation period. The C2 level was maintained at approximately 1000 ng/mL for the first 4 months and then at 600 to 800 ng/mL thereafter.
Graft Clarity and Rejection Episodes
In the CsA group, immunologic rejection developed in 6 (30.0%) of 20 eyes, with the length of time between transplantation and rejection ranging from 23 to 91 weeks. In 3 of these 6 cases, CsA had been discontinued within 6 months after surgery (within the first week in 2 eyes and within 3 months in 1 eye). In the remaining 3 patients, rejection occurred within 18 to 28 months. Therefore, rejection occurred in all 6 eyes after discontinuation of CsA. In 3 of the 6 eyes in which rejection developed, graft clarity was recovered by immunosuppressive therapy (reversible rejection); in the remaining 3 eyes, decompensation occurred with no recovery (irreversible rejection). At final observation, 5 of the 6 eyes with immunologic rejection showed endothelial decompensation. Four other eyes also showed endothelial decompensation during the observation period, resulting in 9 (45.0%) of 20 eyes with graft failure in the CsA group (final graft clarity rate, 55.0%; Table 3 ).
| Event | CsA Group (n/Total n) | Control Group (n/Total n) | P Value |
|---|---|---|---|
| Rejection (mos) | |||
| 6 | 0/20 | 1/18 | .47 |
| 12 | 1/19 | 1/17 | 1.00 |
| 24 | 3/16 | 1/15 | .60 |
| Overall | 6/20 | 3/18 | .24 |
| Nonimmunologic failure (mos) | |||
| 6 | 0/20 | 0/18 | NA |
| 12 | 0/19 | 1/17 | .47 |
| 24 | 1/16 | 2/15 | .60 |
| Overall | 4/20 | 5/18 | .44 |
| Failure (all causes; mos) | |||
| 6 | 0/20 | 0/18 | NA |
| 12 | 1/19 | 1/17 | 1.00 |
| 24 | 4/16 | 3/15 | 1.00 |
| Overall | 9/20 | 8/18 | 1.00 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


