Purpose
To evaluate the prophylactic efficacy of selective laser trabeculoplasty for preventing an increase in intraocular pressure (IOP) after intravitreal triamcinolone acetonide injection.
Design
Prospective, comparative, interventional case series.
Methods
We studied 31 eyes with a baseline IOP of 21 mm Hg or more of 31 patients for which intravitreal triamcinolone acetonide injection was planned for diabetic macular edema. The patients were divided into 2 groups, a study group and control group. The study group comprised 15 eyes of 15 patients that underwent selective laser trabeculoplasty a mean of 8.3 ± 4.1 days before intravitreal triamcinolone acetonide injection. The control group comprised 16 eyes of 16 patients who underwent only intravitreal triamcinolone acetonide injection. Main outcomes measures were mean IOP and number of patients requiring antiglaucomatous therapy.
Results
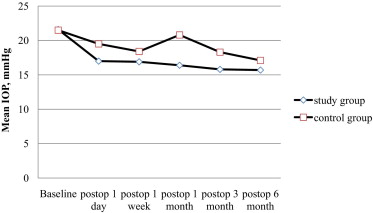
Mean baseline IOP was 21.6 ± 0.9 mm Hg in the study group and 21.5 ± 0.8 mm Hg in the control group ( P = .98). Mean IOP at 1 day after injection was 17.0 ± 2.0 mm Hg in the study group and 19.5 ± 4.3 mm Hg in the control group ( P = .23). Mean IOP at 1 week after injection was 16.9 ± 1.7 mm Hg and 18.4 ± 4.0 mm Hg, respectively ( P = .49); mean IOP at 1 month after injection was 16.4 ± 1.5 mm Hg and 20.8 ± 5.6 mm Hg, respectively ( P = .003); mean IOP at 3 months after injection was 15.8 ± 2.5 mm Hg and 18.3 ± 5.5 mm Hg, respectively ( P = .01); and mean IOP at 6 months after injection was 15.7 ± 1.4 mm Hg and 17.1 ± 1.5 mm Hg, respectively ( P = .03). The number of patients requiring antiglaucomatous therapy during follow-up was 0 of 15 eyes in the study group and 8 of 16 eyes in the control group ( P = .001).
Conclusions
The IOP elevation after intravitreal triamcinolone acetonide injection may be prevented by performing selective laser trabeculoplasty before intravitreal triamcinolone acetonide injection, especially in cases with a baseline IOP of 21 mm Hg or more.
Intravitreal triamcinolone acetonide injection has become an important therapeutic method for several retinal diseases, including diabetic macular edema, age-related macular, degeneration, proliferative diabetic retinopathy, retinal vein occlusions, and noninfectious posterior uveitis. Several side effects of intravitreal corticosteroids have been reported, such as endophthalmitis, increased intraocular pressure (IOP), cataract formation, vitreous hemorrhage, retinal detachment, and central serous chorioretinopathy. Elevated IOP, however, is the most common side effect of intravitreal triamcinolone injection. The reported prevalence of IOP elevation after different doses of intravitreal triamcinolone varies among studies, ranging from 40% to 77%.
Among the risk factors for steroid-induced glaucoma, a history of pre-existing glaucoma, high baseline IOP, younger age, history of IOP elevation after previous intravitreal triamcinolone injections, and first-degree relatives with primary open-angle glaucoma have been reported. The IOP elevation after intravitreal triamcinolone injection usually can be treated medically with either monotherapy or combination therapy. In some cases, however, IOP elevation is not controlled adequately with medical therapy, or the patients may be intolerant to the ocular and systemic adverse effects of these drugs. Some patients require surgical intervention, including surgical removal of the corticosteroid, trabeculectomy, Ahmed valve placement, or pars plana vitrectomy.
Since 2002, selective laser trabeculoplasty (SLT) has become available for the treatment of glaucoma. SLT is an effective treatment method for lowering IOP in patients with open-angle glaucoma such as primary open-angle glaucoma, pigmentary glaucoma, pseudoexfoliation glaucoma, and normal-tension glaucoma. The aim of this study was to assess the effect of preinjection SLT treatment for preventing IOP elevation after intravitreal triamcinolone injection in patients with a baseline IOP of 21 mm Hg or more.
Methods
Study Population and Design
This prospective, randomized study was performed at the Beyoglu Eye Research and Training Hospital in the Glaucoma and Retina Departments. The study included patients with a baseline IOP of at least 21 mm Hg for whom treatment with an intravitreal injection of 4 mg triamcinolone acetonide was planned for diabetic macular edema. All patients were in a nonproliferative diabetic retinopathy phase. The patients were divided into 2 groups. The study group comprised consecutive patients who underwent SLT before the intravitreal injection. The control group comprised consecutive patients who underwent only intravitreal triamcinolone injection. Patients with a history of glaucoma, those with first-degree relatives with primary open-angle glaucoma, those with gonioscopically occludable angles, and those with previous ocular surgery were excluded. Also, patients with a follow – up period of less than 6 months were excluded from the analysis. Both verbal and written informed consent were obtained from all patients for both the intravitreal triamcinolone injection and SLT procedures.
Selective Laser Trabeculoplasty Procedure
All SLT procedures were performed by the 2 physicians (E.B., N.K.). All of the patients were administered 1 drop of topical proparacaine (Alcaine; Alcon, Fort Worth, Texas, USA) and a Latina SLT lens (Ocular Instruments, Bellevue, Washington, USA) was used to visualize the angle. The laser beam was focused on the trabecular meshwork. The initial energy setting was 0.7 mJ and was increased or decreased by 0.1 mJ until intermittent small cavitation bubbles were visible. The inferior or nasal 180 degrees were treated with contiguous laser spots. A mean of 54.4 ± 5.6 SLT spots were applied. One drop of brimonidine 0.2% was administered immediately after the laser procedure. Floromethalon (Flarex; Alcon Laboratories, Inc) eye drops were administered 4 times daily for 1 week.
Intravitreal Triamcinolone Injection Procedure
All injections were performed by the same vitreoretinal surgeon (A.T.Y.). The intravitreal triamcinolone was administered with topical anesthesia using proparacaine HCl 0.5% (Alcaine). After sterilization of the ocular surface and periocular area with 10% povidone iodine, 4 mg triamcinolone acetonide (0.1 mL of 40 mg/1.0 mL suspension; Kenacort [Bristol-Myers Squibb, Istanbul, Turkey]) was injected with a 30-gauge needle at the inferonasal or inferotemporal pars plana (3.5 mm from the limbus in phakic eyes, 3.0 mm in pseudophakic eyes). Paracentesis was not performed in any patient. After the procedure, a topical antimicrobial agent was administered to all patients.
The patients were followed up for 6 months after the intravitreal triamcinolone injection. Additional data were collected, including age, treated eye, and sex. IOP measurements before and at 1 day, 1 week, 1 month, 3 months, and 6 months after intravitreal injection were recorded. The IOP was measured by the same investigator with same calibrated Goldman applanation tonometer. He was masked when measuring the IOP at each visit. Three measurements were obtained from each eye. The mean of the 3 measurements was used in the statistical analyses. When IOP after injection exceeded 24 mm Hg, an antiglaucomatous drug was administered. The patients who had received antiglaucomatous medication were followed up for glaucomatous damage.
Statistical Analysis
All statistical tests were performed using SPSS software version 16 (SPSS, Inc, Chicago, Illinois, USA). The independent-samples t test, Mann–Whitney U test, and Fisher exact test were performed for statistical analysis. Differences with a P value of less than .05 were considered significant.
Results
The study included 31 eyes of 31 patients. All patients completed the 6-month follow-up period. The study group comprised 15 eyes of 15 patients (6 men, 9 women; mean age, 60.1 years). The control group comprised 16 eyes of 16 patients (9 men, 7 women; mean age, 58.5 years). There was no statistically significant difference between the 2 groups with regard to sex or age ( P = .47 and P = .57, respectively). The mean central corneal thickness in the study and control group was 544.4 ± 13.5 and 546.6 ± 11.5, respectively ( P = .61). The baseline cup-to-disc ratio was 0.42 ± 0.10 and 0.39 ± 0.08 in the study and control groups, respectively ( P = .458).
The indication for intravitreal triamcinolone injection was macular edema secondary to diabetes mellitus in all of the patients. In the study group, the mean period between the SLT and intravitreal injection was 8.3 ± 4.1 days (range, 7 to 20 days). IOP measurement after SLT was performed a mean of 8 days after SLT in the study group. The mean number of previous intravitreal triamcinolone injections was 0.85 ± 0.9 (range, 0 to 2) in the study group and 0.71 ± 1.1 (range, 0 to 3) in the control group and did not differ significantly between groups ( P = .51).
The Table presents the mean IOP values of the 2 groups at baseline and during the follow-up period. Only 2 patients in the study group had IOP values of more than 21 mm Hg after intravitreal injection. These 2 IOP values were 22 mm Hg and 24 mm Hg, observed at 1 day and 3 months after injection, respectively. The IOPs decreased without antiglaucomatous therapy. Mean IOP values after intravitreal injection were lower in the study group compared with the control group at all of the follow-up visits ( Figure ). Except for at day 1 and week 1 after intravitreal injection, the differences between groups were significant at each follow-up visit.
| Group | IOP (mm Hg) | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | After SLT a | After IVTA | |||||
| 1 Day | 1 Week | 1 Month | 3 Months | 6 Months | |||
| Study group | |||||||
| Mean ± SD | 21.7 ± 0.9 | 16.6 ± 2.1 | 17.1 ± 2.0 | 17.0 ± 1.7 | 16.5 ± 1.6 | 15.8 ± 2.4 | 15.7 ± 1.5 |
| Range | 21 to 24 | 15 to 21 | 14 to 22 | 14 to 20 | 15 to 21 | 14 to 24 | 14 to 18 |
| Control group | |||||||
| Mean ± SD | 21.6 ± 0.9 | — | 19.5 ± 4.3 | 18.5 ± 4.0 | 20.9 ± 5.6 | 18.4 ± 5.5 | 17.2 ± 1.6 |
| Range | 21 to 24 | — | 16 to 30 | 12 to 27 | 15 to 38 | 14 to 36 | 15 to 20 |
| P value b | .90 | — | .095 | .234 | .007 | .012 | .032 |
a Performed a mean 8.3 days after SLT.
None of the patients required antiglaucomatous eye drops during the 6-month follow-up after intravitreal triamcinolone injection in the study group, but IOP-lowering medications were required in 8 (50%) of the 16 eyes in the control group ( P = .001). In the control group, an antiglaucomatous eye drop was required in 4 patients at 1 day after injection, in 4 patients at 1 week after injection, in 7 patients at 1 month after injection, in 6 patients at 3 months after injection, and in 6 patients at 6 months after injection. The number of antiglaucomatous drops varied from 1 to 3 for patients who required antiglaucomatous medication in the control group. No further interventions, such as glaucoma surgery, were required.
In the control group, 8 of 16 patients (50%) who had received antiglaucomatous medications underwent optic nerve head examination, visual field test with Goldman perimetry, and peripapillary nerve fiber layer measurement with Stratus optic coherence tomography (Carl Zeiss Meditec, Dublin, California, USA). There were no glaucomatous changes such as glaucomatous cupping, glaucomatous visual field defects, and thinning of the peripapillary retinal nerve fiber.
Adverse events after SLT included a mild transient anterior chamber reaction in 2 patients (13%) and transient ocular discomfort in 3 patients (20%). During the follow-up period, no severe complications, such as endophthalmitis, retinal detachment, or vitreous hemorrhage, occurred.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


