Chapter 30 Principles in Thyroid Surgery
Introduction
![]() Please go to expertconsult.com to view related videos Basic Thyroid Surgical Maneuvers and Advanced Thyroid Cancer Surgery.
Please go to expertconsult.com to view related videos Basic Thyroid Surgical Maneuvers and Advanced Thyroid Cancer Surgery.
There are a number of chapters in this text devoted to specific thyroid surgical approaches ranging from minimally invasive to extracervical, robotic, and even transoral approaches (see Chapters 31, Minimally Invasive Video-Assisted Thyroidectomy, and 36, Robotic and Extracervical Approaches to the Thyroid and Parathyroid Glands: A Modern Classification Scheme). In this chapter we endeavor to review the basic technical principles in standard thyroid surgery, which are to some degree applicable to all forms of thyroid surgery. Standard open thyroid surgery may be considered the basic starting point for all thyroid surgeons; familiarity with these surgical anatomic principles is, of course, an essential first step necessary for other less routine surgical approaches.
Extent of Thyroidectomy
Certain clinical parameters other than cytology can importantly affect the decision to operate and the extent of thyroidectomy. These include age of the patient (higher malignancy risk <20 and >60), sex of the patient (males have increased risk of malignancy), family history of thyroid malignancy syndromes such as familial papillary carcinoma, Cowden’s syndrome, MEN 2A or 2B (see Chapters 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, and 29, Familial Nonmedullary Thyroid Cancer), personal history of exposure to ionizing radiation especially as a child (see Chapter 28, Chernobyl and Radiation-Induced Thyroid Cancer), and history of rapidly growing thyroid mass (see Chapter 11, The Evaluation and Management of Thyroid Nodules).1 On physical exam, size, firmness, and fixation of the nodule are important to note. Especially important and predictive of malignancy are the findings of vocal cord paralysis and lymphadenopathy (see Chapters 14, Preoperative Radiographic Mapping of Nodal Disease for Papillary Thyroid Carcinoma, and 15, Pre- and Postoperative Laryngeal Exam in Thyroid and Parathyroid Surgery). Thyroid-stimulating hormone (TSH) elevation and certain ultrasonographic findings (including findings of solidness, irregular margins, gross calcification, microcalcification, central blood flow, taller than wide morphology, and increased elastography measures of firmness and density) also increase the risk of malignancy (see Chapter 13, Ultrasound of the Thyroid and Parathyroid Glands).2,3 Although currently not routinely applied, clinically molecular assessment of the fine-needle aspiration (FNA) cytology (including assessment for BRAF, RET-PTC, RAS, PAX8/PPAR gamma and immunohistochemical staining for galectin 3) will likely significantly impact malignancy risk assessment (see Chapters 11, The Evaluation and Management of Thyroid Nodules, and 17, Molecular Pathogenesis of Thyroid Neoplasia). Of course, centrally important in the decision to operate and in discussions of extent of thyroidectomy is cytopathologic analysis. It is important to note that these cytologic data, although of great importance, are always interpreted in the light of the other key clinical parameters already described and not in isolation (see Chapter 12, Fine-Needle Aspiration of the Thyroid Gland).
Extent of Surgery Based on FNA Result
In 2007, the National Cancer Institute convened a state of the science meeting to attempt to consolidate and provide guidance regarding thyroid cytopathology terminology.4 This meeting resulted in six distinct categories of thyroid FNA cytology readings, including nondiagnostic, benign, atypia of undetermined significance/follicular lesion of undetermined significance, follicular/Hurthle neoplasm, suspicious for malignancy, and diagnostic for papillary thyroid carcinoma (PTC). We will discuss the surgeon’s response to each of these cytopathologic diagnoses. As noted earlier, the clinician must respond to the overall clinical situation, including history and physical exam parameters as well as FNA data. It is not uncommon and is appropriate in the setting of significantly unfavorable clinical parameters that surgery is recommended for the patients although the FNA has been read as benign.
Benign
When the needle biopsy returns with adequate cellularity and clearly benign, generally surgery is deferred in lieu of serial ultrasonographic surveillance (see Chapter 11, The Evaluation and Management of Thyroid Nodules). Occasionally because of size of the lesion or the need for absolute assurance by the patient, surgery can be considered and in this circumstance is generally offered unilaterally.
Nondiagnostic
The nodule that has been needled as nondiagnostic should undergo repeat aspiration with ultrasound guidance; 50% of such repeated aspirates yield diagnostic information.5 Diagnostic lobectomy can be considered for the nodule with multiple nondiagnostic results; core needle biopsy may also be considered. Approximately 10% of such nodules are malignant.
Follicular or Hurthle Cell Neoplasm
When the needle biopsy result reveals follicular or Hurthle cell neoplasm, generally lobectomy without frozen section is offered for complete capsular histologic evaluation. The patient is counseled about the potential (generally considered approximately 20%) need for completion thyroidectomy. Total thyroidectomy may be considered when the lesion is more likely to be malignant, which in turn depends on the sex of the patient (males with higher risk of cancer than females) and the size of the lesion (generally > 4 cm).6 Also initial total thyroidectomy may be considered in patients who, based on their age, may have a less favorable prognosis should a cancer be diagnosed or if there is substantial unbiopsied contralateral lobe nodularity.
Suspicious for Papillary Carcinoma
Needle aspirates read as suspicious for papillary carcinoma have a risk of papillary cancer of approximately 60% and are generally best treated with lobectomy with frozen section and intraoperative touch cytologic prep (see Chapter 12, Fine-Needle Aspiration of the Thyroid Gland). If a diagnosis of papillary carcinoma can be made intraoperatively, total thyroidectomy can be offered. Biopsy of suspicious lymph nodes may be helpful for diagnosing papillary carcinoma.
Diagnostic for Medullary Carcinoma, Anaplastic Carcinoma, or Lymphoma
An initial biopsy read as diagnostic for medullary carcinoma of the thyroid requires a workup to exclude pheochromocytoma, hyperparathyroidism, and radiographic evaluation for nodal disease with consideration for total thyroidectomy, central neck dissection, and often unilateral or bilateral lateral neck dissection (see Chapters 23, Sporadic Medullary Thyroid Carcinoma, 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, and 25, Sporadic Medullary Thyroid Microcarcinoma). Blood testing for calcitonin, CEA, and RET mutational analysis can be informative regarding the extent of disease and whether the patient has inherited or sporadic disease. With a diagnosis of anaplastic carcinoma, surgery is offered, especially in the relatively uncommon circumstance when all gross disease can be resected. Given the significance of the diagnosis of anaplastic, open biospy confirmation rather than FNA alone can be considered. A more common surgical procedure appropriate for anaplastic carcinoma is isthmusectomy to confirm tissue diagnosis often combined with tracheotomy if the airway is deteriorating. Aspirates read as diagnostic for lymphoma often need to be subtyped, and frequently this requires either core biopsy or open biopsy/isthmusectomy (see Chapter 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma).
Diagnostic for Papillary Carcinoma of the Thyroid
The extent of thyroidectomy for patients with well-differentiated thyroid carcinoma, specifically PTC, has been actively debated within head neck and surgical oncologic circles for decades. PTC, in most cases, is associated with prolonged survival.7 The controversy as to extent of thyroidectomy has been fueled by the prolonged long-term survival enjoyed by the majority of patients with papillary thyroid carcinoma, making randomized prospective studies impractical. Based on Bilimoria et al. and others, total or near total thyroidectomy is recommended by the American Thyroid Association (ATA) and the National Comprehensive Cancer Network (NCCN) guidelines for all PTC > 1 cm, as this facilitates follow-up with radioactive iodine treatment and scanning and serial thyroglobulin analysis.8–10 The best surgical plan for a patient with a preoperative FNA read as diagnostic of papillary carcinoma of the thyroid has been controversial in part because of the unique features of PTC (see Chapters 18, Papillary Thyroid Cancer, and 19, Papillary Thyroid Microcarcinoma).
Unique Features of Papillary Carcinoma
The prevalence and favorable prognosis of small PTC lesions
The first of these unique features of papillary cancers is the entity of occult or microscopic carcinoma. Such lesions are typically defined as less than 1 cm and typically as intrathyroidal.11,12 Such lesions are highly prevalent in humans and are often found incidentally during thyroidectomy performed for other lesions, occurring on an average in 8.5% of surgical specimens.13,14 Comparing known autopsy rates of PTC, which range from 5% to 36% depending on the country, to SEER clinical prevalence rates, it is estimated that, in fact, only 2% of existing PTC lesions in humans ever present with clinical disease.15,16 Although small occult carcinomas can metastasize to regional nodal beds in the neck in up to 30% of cases, they are rarely associated with clinically significant metastatic disease or death.17,18 The favorable prognosis of small PTC lesions has been well studied. In 1960, Woolner described six patients with occult papillary carcinoma, identified through cervical lymph node biopsy, who were followed without any thyroid surgery at all without progression of their disease over many years.11 In Japan, trials of observation alone for patients with biopsy-positive small PTC lesions showed over 94% fail to demonstrate disease progression during prolong follow-up.19
PTC cervical lymph node micrometastasis
The second unusual feature of papillary carcinoma is that cervical lymph node metastasis at presentation while highly prevalent in microscopic form in regional nodal beds seems to, for many patients, have little prognostic implication.7,20 Initial reports suggested an improved prognosis in N-positive patients, but when investigated this was ultimately found to result from the younger age of patients presenting with lymphadenopathy.20,21 Some evidence suggests that the presence of cervical lymph node metastasis, although not significantly affecting survival, may increase recurrence rate and may have greater prognostic significance in elderly patients.22,23 Recent work, including a Surveillance, Epidemiology, and End Results (SEER) database analysis of nearly 10,000 patients, suggests that nodal disease when in macroscopic form is in fact an important determinant of tumor-related survival, especially when present in patients over 45 years of age.24–26
Papillary carcinoma frequently spreads to both central and lateral regional nodal beds. Although 30% of patients have clinically positive nodes (i.e., macroscopically positive) at presentation (based on physical or ultrasound examination), histologic studies show microscopic spread to regional neck nodes in 50% to 80% of patients and to the contralateral thyroid lobe in up to 80% of patients.27–29 This high prevalence of microscopic disease in the neck is in stark contrast to the low rate of the development of clinical disease in the untreated neck.30,31 Presumably, in the majority of cases, microscopic disease in the neck is inherently inactive. The discordance between the presence of microscopic disease in the neck and its lack of clinical significance has led to the abandonment of past recommendations of elective lateral neck dissection in the N0 setting.27,29 Controversy still exists as to the applicability of this argument in the ipsilateral central neck.32
PTC’s robust prognostic risk grouping segregation
PTC patients can be successfully segregated into prognostic groups. Starting with Woolner in the 1960s, age and degree of invasiveness were used to segregate patients into diverse prognostic groups (see Chapter 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer).33 The identification of key prognostic variables allows us to segregate patients with papillary carcinoma into various risk groups. The risk groups are based on various prognostic factors, which are essentially the same in various series (Table 30-1) and are related to both patient (age and gender) and the tumor (size, extrathyroid extension, histologic grade) factors. Based on these factors, two or three risk groups for well-differentiated thyroid cancer patients (i.e., follicular and papillary cancers) are separated with discrete patterns of recurrence and survival.21,34,35
Risk-group stratification appropriately allows aggressive treatment in the high-risk group and avoidance of excessive treatment and its complications in low-risk groups (Table 30-2). Key elements of existing prognostic schemes for papillary carcinoma include the following:
2. Degree of invasiveness or extrathyroid extension (increased invasiveness increases risk of local, regional, and distant recurrence and decreases survival)
3. Metastasis (presence of distant metastasis decreases survival)
4. Sex (males generally have a poorer prognosis than females)
5. Lesion size (lesions larger than 4 cm have a poorer prognosis, and lesions smaller than 2 cm have an improved prognosis)
6. Clinically palpable lymph node metastasis, especially with extranodal extension
Brierly reviewed 382 patients with 10 different commonly used prognostic scoring systems and found AGES, TNM, EORTC, MACIS, and AMES systems to be comparable in predicting prognosis.36 The two best-known prognostic schemes are those devised by Hay and Cady.21,34 Hay’s scheme for papillary carcinoma is summarized by the mnemonic AGES for age, gender, extent, and size. The mnemonic AMES (age, metastasis, extent, and size) summarizes Cady’s prognostic system for papillary carcinoma and follicular carcinoma. The risk groups defined by these well-known risk assessment systems are quite broad. Cady defined his low-risk group as males younger than 40 years of age or females under age 50 without distant metastasis. Also in Cady’s low-risk group are older persons with intrathyroidal papillary carcinoma or follicular carcinoma with minimal capsular involvement, no evidence of distant metastasis, and lesions less than 5 cm. Cady’s high-risk group includes all patients with distant metastasis, all younger patients with follicular carcinoma with major capsular involvement, and all older patients with extrathyroidal papillary carcinoma, follicular carcinoma with major capsular involvement, or papillary carcinoma of greater than 5 cm. Eighty-nine percent of Cady’s patients fit into his low-risk category. Recurrence in this low-risk category was 7.7% and mortality was 1.8%. Eleven percent of Cady’s patients fit into his high-risk group, which had a 59% incidence of recurrence and 46% mortality (see Table 30-2).
Other factors that affect prognosis for well-differentiated thyroid carcinoma have been studied. If gross disease is left at completion of initial surgery, prognosis worsens.37 If radioactive iodine and T4 suppression are given postoperatively, prognosis generally improves, although a minority of thyroid cancer experts do not accept this.38,39 Some have used histologic grading in prognostic schema.40
The ATA has recently recommended the AJCC/UICC TNM staging system be employed for all patients with differentiated thyroid cancer.9 It is important to appreciate that although this widely used AJCC/UICC TNM staging system for differentiated thyroid cancer provides a convenient and familiar method of assessing the extent of the tumor, it does not take into account several independent prognostic variables and was developed to predict primarily risk of death, not recurrence. Current risk stratification schemes also do not take into account certain aggressive histologic subtypes of cancer and other pathologic findings including frequent mitosis, tumor necrosis, microscopic degrees of extrathyroidal extension capsular invasion, or molecular characterization of the primary tumor. TNM data are based primarily on clinical and pathologic data available after thyroidectomy and therefore do not change over time, whereas risk of recurrence or death may change depending on the patient’s response to treatment (see Chapter 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer). To improve the estimation of risk of recurrence, the ATA has offered a risk of recurrence stratification system, which segregates patients into low, intermediate, and high risk of recurrence.9 The ATA low risk of recurrence group includes patients without distant metastasis, with all macroscopic tumor resected, without tumor invasion of local structures, without aggressive histologies or vascular invasion, and without uptake outside of the thyroid bed following initial whole-body scanning (WBS). The ATA intermediate risk of recurrence group includes patients with microscopic local invasion, cervical nodal disease, or uptake outside of the thyroid bed at the first WBS scanning after ablation and patients with aggressive histology or vascular invasion. The ATA high risk of recurrence group includes patients with macroscopic tumor invasion, patients with incomplete tumor resection, and patients with distant metastasis or with thyroglobulin out of proportion to findings on initial WBS.9 The risk of recurrence for the low, intermediate and high ATA risk groups are 14%, 44% and 86% respectively (see Chapter 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer).
Extent of Thyroidectomy for PTC > 1 cm
As already mentioned, there is recent important evidence from Bilimoria and coworkers, who studied a group of more than 52,000 national cancer database patients with papillary carcinoma. They found—using Cox hazards modeling adjusting for age, race, income, lymph node status, distant metastasis, radioactive iodine use, and year of diagnosis—that for lesions greater or equal to 1 cm there is significant improvement in both survival and recurrence at 10 years with total thyroidectomy as opposed to less than total thyroidectomy.8 Recommendation 26 of the revised ATA guidelines of 2009 suggests total thyroidectomy is the preferred procedure for most patients with the preoperative diagnosis of PTC.9 Current NCCN guidelines also suggest total thyroidectomy for all patients with PTC except those with extremely favorable risk profiles (young age, no history of XRT, no distant metastasis, no cervical lymphadenopathy, no aggressive histology, and size <4 cm).10 Strong consideration for total thyroidectomy is a repeated theme in most endocrine published guidelines including those of the American Association of Clinical Endocrinology as well as European guidelines.41,42
Although the mainstream recommendation for total thyroidectomy characterizes most current treatment recommendations of endocrine organizations, one must acknowledge that extent of surgery, especially for low-risk PTC, has been actively researched for decades and that there exists an extensive past literature investigating both survival and recurrence both supporting7,22,23,38,43–51 and arguing against18,31,35,40,47,48,52–58 total thyroidectomy for PTC patients.
Total Thyroidectomy for PTC: Additional Considerations
Arguments other than those already discussed about improvement in survival and recurrence supporting total thyroidectomy are numerous and include that total thyroidectomy provides an opportunity for the routine use of whole-body scanning and the sensitive use of thyroglobulin as a marker postoperatively. Radioiodine ablation of thyroid gland remnants after total thyroidectomy can allow for whole-body scanning and the sensitive use of thyroglobulin as a postoperative marker.59 Measurement of serum thyroglobulin as a tumor marker is most reliable when normal thyroid tissue is absent.60 It should be noted that some feel thyroglobulin can also be followed when a lobe or a fraction of a lobe is present. Schlumberger found that thyroglobulin was useful even in patients who had less than total thyroidectomy without ablation.61 Harvey et al. measured serum thyroglobulin in 84 patients with well-differentiated thyroid cancer after lobectomy and 58 patients after total thyroidectomy. Tumor recurrence was heralded by increased thyroglobulin in both groups. The authors concluded that despite residual thyroid tissue, serum thyroglobulin could exclude the presence of significant metastatic disease in most patients after lobectomy for thyroid cancer.62
Following total thyroidectomy, even in expert hands, a significant number of patients have residual thyroid tissue that requires postoperative ablation. Auguste showed that after total thyroidectomy, 13 out of 80 patients required radioiodine ablation.63 Marchetta also noted average neck uptake of 15% after total thyroidectomy.64 Surprisingly, Szilagy et al. noted that in their series, 20% of patients having had total thyroidectomy did not require T4 replacement.65 There is no question, however, that when a complete total thyroidectomy is done, hypothyroidism is achieved sooner than with less than total thyroidectomy and postoperative radioactive iodine ablation. The three regions that often contribute to postoperative uptake after total thyroidectomy are discussed later in this chapter.
Total thyroidectomy also treats any potential contralateral lobe papillary carcinoma foci that could be the source of additional recurrence or, although rare, dedifferentiation. One must keep in mind that management of papillary thyroid cancer varies in different parts of the world with quite different approaches. In Japan, one common approach is a partial thyroidectomy leaving a large remnant in the contralateral lobe but performing ipsilateral or bilateral nodal surgery. Interestingly this algorithm, which targets microscopic disease from a different perspective emphasizing the ipsilateral nodal beds and de-emphasizing the contralateral thyroid lobe, has been met with favorable results.66
As a final point, in the surgical literature, appreciation of “risk groups” has appeared since the 1990s. Confusion has been evident, however, between (1) occult carcinomas and microcarcinomas and (2) the AMES and AGES low-risk groups. These two are not synonymous. It is, however, obvious that many more patients are included in the low-risk groups than just patients with 1 cm or smaller papillary lesions. Most patients within the low-risk group enjoy excellent long-term prognosis, however.53
Extent of Thyroidectomy for PTC <1 cm (PTMC)
Bilimoria’s recent work described previously revealed no difference in survival or recurrence rates between lobectomy and total thyroidectomy for lesions less than 1 cm (i.e., micropapillary carcinomas, MPTC).8 For MPTC, there is a variety of work supporting the notion that lobectomy is equivalent to total thyroidectomy.8,67–69 The decision as to extent of thyroidectomy when a papillary microcarcinoma is preoperatively identified should be a team decision made by the surgeon and the endocrinologist, and the patient. The decision should, of course, incorporate nodal status in the presence of contralateral lobe ultrasound abnormalities. The preoperative identification of papillary microcarcinomas will likely increase as our current medical environment embraces robust diagnostic scrutiny of small thyroid nodules. This is especially true given that the latest ATA nodule guidelines recommend FNA evaluation for nodules as small as 5 mm in certain high-risk groups.9
Extent of Thyroidectomy for PTC <1 cm (PTMC) Detected on Lobar Specimens: Completion Thyroidectomy
The finding of MPTC in lobar specimens is a common problem occurring from 1.3% to 21.6%, with a mean of 8.5% patients in a number of recent studies.70–80 Recent ATA guidelines recommend completion thyroidectomy should be offered to those patients for whom a near-total or total thyroidectomy would have been recommended had the diagnosis been available before the initial surgery. This, the ATA guidelines note, includes all patients with thyroid cancer except those with small (< 1 cm) tumors, unifocal tumors, intrathyroidal tumors, node-negative tumors, or low-risk tumors.9 NCCN recommendations for completion thyroidectomy are somewhat more liberal, allowing for observation as opposed to completion surgery in some favorable lesions up to 4 cm (with no extrathyroidal extension, negative margins, no multifocal diseases, no cervical lymphadenopathy, nonaggressive pathologic variants, and no contralateral lesions).10 Interestingly when reviewing the literature on MPTC rates of multifocality, bilaterality, extrathyroidal spread, lymph node metastasis, distant metastasis, rates of recurrence and BRAF positivity overlap significantly with standard (i.e., nonmicropapillary carcinoma) of the thyroid.12,19,81–90
However, in a number of studies, there is a suggestion that below 5 mm there seems to be a favorable change in the expression of MPTC, at least in terms of rates of extrathyroidal spread and lymph node metastasis and perhaps distant metastasis, survival, and recurrence.14,81,82,91–95 This size (i.e., ≤ 5 mm) MPTC has been termed minute carcinoma.96 Contralateral disease can be predicted in the setting of ipsilateral MPTC, by multifocal primary, presence of contralateral lobe nodules on preoperative ultrasound, ipsilateral aggressive histology, and history of radiation therapy, and familial or papillary carcinoma.90,92,97–101 It has been reported that individual foci of MPTC have a distinct molecular profiles.102 Recent work suggests completion thyroidectomy is a safe procedure when performed by an experienced thyroid surgeon.103,104
Given the preceding discussion, it is best to take into account a number of factors when considering completion thyroidectomy for MPTC. Important factors include size (i.e., if the primary MPTC is > 5 mm), evidence of multifocality, evidence of contralateral lobe nodularity on preoperative ultrasound, aggressive histology of the MPTC, extrathyroidal spread, and clinical or ultrasound apparent lymphadenopathy. Practical management issues also need to be considered. Thyroid hormone availability to the patient and an expressed desire by endocrinologists for whole-body scanning and postoperative thyroglobulin assessment are also important determinants of this decision. Probably one of the most important determinants in offering completion thyroidectomy is the postoperative laryngeal exam105 and the intraoperative and pathologic assessment of parathyroid preservation from the first side. These factors importantly relate to risk of completion surgery. Most important, this should be a team decision made jointly by the patient, the endocrinologist, and the surgeon after thorough discussion.
Surgical Complications
Surgical treatment must blend an aggressive oncologic approach with a commitment to minimize the potential risk of complications. Certainly, in skilled hands total thyroidectomy can be offered with low morbidity.106 It is of note that so-called nonexpert surgeons perform 86% of routine thyroid surgery107 and 50% of patients undergoing thyroid surgery in the United States have it performed by surgeons performing fewer than five thyroidectomies per year.108 Complications are more common in such patients.107 Clearly despite isolated centers of surgical expertise, the bulk of the literature supports increased complication rates, mainly recurrent laryngeal paralysis and permanent hypoparathyroidism with bilateral thyroid surgery when compared with unilateral surgery40,109 (see Chapters 45, Pathophysiology of Recurrent Laryngeal Nerve Injury, 46, Management of Recurrent Laryngeal Nerve Paralysis, 47, Non-Neural Complications of Thyroid and Parathyroid Surgery, and 48, Endocrine Quality Registers: Surgical Outcome Measurement).
RLN paralysis
Permanent recurrent laryngeal nerve (RLN) paralysis rates in expert hands generally are in the 1% to 2% range.110 However, many reports reveal significantly higher rates, in the 6% to 8% range with rates as high as 23% being reported (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve).48,57,58 In two studies including those by Hockauf, who reviewed more than 1000 patients, and Segal, who reviewed 61 pediatric patients, a 10% incidence of vocal cord paralysis is reported.111,112 Sinclair, on the other hand, reported a 1.1% incidence of RLN paralysis with routine thyroidectomy and a 17.5% incidence in surgery for retrosternal goiters.113 Martensson showed that the incidence of RLN paralysis increased with bilateral thyroid surgery, revision thyroid surgery, and surgery for malignancy or in patients brought back for bleeding.114 The reported prevalence of RLN injuries after thyroid surgery, in fact, varies widely. A recent analysis of 27 articles reviewing more than 25,000 patients undergoing thyroidectomy found the average immediate postoperative vocal cord paralysis (VCP) rate was 9.8%. The rate of permanent VCP varied 10-fold, according to the method of examining the larynx and ranged from 0% to 18.6%.115 With the advent of nationwide surgical outcome databases we have broad cross-sectional perioperative information, which may give insight into these issues. The Scandinavian quality register (SQR) for thyroid and parathyroid surgery, reporting in 2008 on 40 endocrine surgical units from Sweden and Denmark specializing in endocrine surgery, noted an immediate VCP rate of 4.3%.116 In the national U.K. British Association of Thyroid and Endocrine Surgeons (BAETS) audit, a VCP rate of 2.5% was found.117 One must keep in mind that both Scandinavian and British quality registers derive from surgeon-reported data without routine postoperative laryngeal exam. For the SQR, the rate of VCP doubled when patients were subjected to routine laryngeal exam as opposed to postoperative laryngoscopy that was performed only in patients with persistent and severe voice changes. Administrators of these two national databases deem the rates of temporary and permanent RLN palsy to be severely underestimated.116,117 Bilateral thyroidectomy is a unique offering within head and neck surgery in that both left and right cranial nerves that innervate the airway introitus are subject to risk at one surgical procedure. Experienced workers who champion total thyroidectomy have reported transient bilateral vocal cord paralysis requiring tracheotomy in between 2% and 3% of patients undergoing thyroidectomy.118 The frequency of bilateral vocal cord paralysis in patients undergoing bilateral thyroid surgery has led to the development of “indications for tracheotomy.”119
RLN paralysis rates in currently available reports are likely to be underestimates in that not all patients in these studies undergo postoperative laryngeal examination. Lo found a 6.6% incidence of vocal cord paralysis when all patients had postoperative laryngoscopy. Of these, only 1.1% had nerve damage recognized during their thyroidectomy.120 Rates of RLN paralysis should be appropriately described as incidence of paralysis per nerve at risk. De Roy Van Zuidewign found a 3.1% incidence of paralysis per nerve at risk.121 Bilateral surgery therefore implies greater risk. Thomusch found RLN injury was associated with extent of surgery, surgery for recurrent disease, and with cases where the surgeon failed to visualize the nerve.122 Most experienced surgeons believe that the RLN should be clearly identified and dissected to demonstrate its entry into the larynx. The identification is generally visual but can be supplemented by neurophysiologic stimulation. Such stimulation is safe and allows the surgeon to identify a neurapraxic nerve injury and possibly defer contralateral thyroid surgery (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve).123,124
Hypoparathyroidism
Hypoparathyroidism is a significant problem for patients after bilateral thyroidectomy. Calcium and, occasionally, vitamin D are required several times a day with careful monitoring. Low calcium levels can result in troublesome and sometimes life-threatening symptoms, whereas high calcium levels can result in renal stones. Rates of temporary hypoparathyroidism (usually defined as less than 8.0 mg/dL within 6 months after thyroidectomy) occur in 17% to 40% of patients undergoing total thyroidectomy.125 Permanent hypoparathyroidism rates after total thyroidectomy in expert hands range from 1.2% to 6.5%.52,125 In an American College of Surgeons survey, which reviewed 24,108 thyroid surgeries, Foster noted a permanent hypoparathyroidism rate of 8%.126 Mazzaferri has noted a 13% incidence of permanent hypoparathyroidism after total thyroidectomy in the community setting.49 Even in selected tertiary care settings, rates of 29% to 48% have been described.34,127 Thomusch found parathyroid injury was associated with extent of resection, surgery for recurrent disease, advanced age, female sex, and surgery for Graves’ disease.122 The risk of hypoparathyroidism increases with invasive cancers and when lymph node dissection is performed with thyroidectomy. The risk of hypoparathyroidism is also vitally linked to the experience of the surgeon. One experienced surgeon noted hypoparathyroidism in only 3.2% of patients after total thyroidectomy overall but described a 25% incidence in his first 25 total thyroidectomy cases.29
Extent of Surgery PTC Summary: Encompass Gross Diseases in Thyroid and Nodes at First Surgery
A clear-cut divergence in prognosis exists in patients with WDTC. A large group of patients enjoy an excellent prognosis, and a small group of patients are associated with a very poor prognosis. Age and presence of distant metastatic disease at presentation are probably the most important prognostic determinants. Most specialists believe that, as Schlumberger has written, the “extent of treatment and follow-up care should be tailored to the level of risk.”128 This philosophy should be kept in mind when one is considering the extent of thyroidectomy for WDTC. The bulk of the data available suggests that the overriding principle in the surgical treatment of WDTC is that the surgeon should, at first surgery, encompass the gross disease in the thyroid and neck nodes and understand that, while present, microscopic disease in the contralateral lobe and the neck nodes has little clinical significance. Tsang wrote, “The goal of surgery should be macroscopic complete resection of tumor. Microscopic residual disease in our series did not differ significantly from its absence in terms of cause specific survival and local control possibly attesting to the effects of radioiodine…in these patients.”48 Such microscopic disease is indolent and not clinically manifest in the majority of cases. Gross disease should be encompassed at the first operation. Enlarged lymph nodes should be excised. Patients with obvious invasion of the trachea should be managed with segmental airway resection.
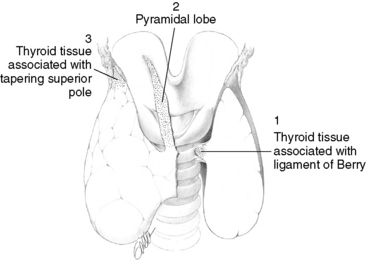
Total thyroidectomy is an excellent procedure in skilled hands. It should be offered when it does not bring with it significant morbidity. However, the feasibility of total thyroidectomy, even in experienced hands, has been questioned because residual thyroid tissue remains (typically centered at the ligament of Berry, pyramidal lobe, or superior pole) in a small but substantial fraction of patients (Figure 30-1). Aggressive bilateral thyroidectomy therefore may not obviate the need for postoperative ablation. Leaving a small contralateral lobe remnant is a reasonable option if away from the cancer and anodular, when necessary to preserve parathyroid function and to limit neural dissection. Education is required at the resident level regarding proper technique in the performance of a total thyroidectomy. This involves careful preservation of the parathyroid glands with intact blood supply and untraumatized RLNs.
It is important to emphasize that the extent of thyroidectomy should be tailored not only to the patient’s risk group and operative findings but also to the progress of the specific surgery, particularly if the contralateral lobe is not involved by cancer. If dissection of the first side has revealed two parathyroids of good color with good vascular pedicles and with an RLN that has been identified and preserved and that stimulates well electrically at the end of the dissection, contralateral thyroid surgery can be safely contemplated. If the first side has not gone well, then elective contralateral lobe resection should be deferred, at least on that day. Parathyroid color changes associated with devascularization may not always be reliable. Characteristic blackened color change may be associated with venous disruption and certainly, when present, implies parathyroid injury and potential dysfunction. However, arterial interruption may be unassociated with this color change yet result in significant parathyroid dysfunction. Proponents of recurrent laryngeal nerve monitoring feel it is helpful in identifying and dissecting the RLN and can be helpful in assessing neural function at the end of surgery (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve).124 Many experienced surgeons, however, feel comfortable with visual identification of the nerve.
Thyroidectomy in Pregnancy
A large study of the Healthcare Utilization Project National Inpatient Sample reviewed 201 pregnant patients undergoing thyroidectomy (or parathyroidectomy) during pregnancy and found that pregnant patients had twice the risk of perioperative complications for both benign and malignant conditions than nonpregnant controls, with 5.5% having fetal complications (e.g., fetal distress and abortion) and 4.5% having maternal complications (e.g., cesarean section and hysterectomy).129 The Endocrine Society has suggested that when necessary, surgery should be performed in the second trimester, before fetal viability after organogenesis. Clearly, discussion between the patient, obstetrician-gynecologist, endocrinologist, and surgeon should be thorough, and surgery, especially for low-risk PTC, should be avoided if at all possible for most pregnant patients until postpartum.130
Nomenclature of Thyroidectomy
The name of the procedure performed should reflect in a straightforward manner the extent of resection. Partial lobectomy, performed in rare cases, implies that a portion of the lobe has been removed. Lobectomy implies that an entire lobe has been completely removed with its capsule intact without isthmus resection. Hemithyroidectomy implies that a lobe has been completely removed, with the isthmus and usually with any associated pyramidal lobe. Subtotal thyroidectomy implies that a lobe and the isthmus have been completely removed and that the contralateral lobe has been partially removed, usually in its medial and ventral portion typically leaving a posterior element. Bilateral subtotal implies a significant portion of both lobes is removed. In a near-total thyroidectomy, all thyroid tissue is removed except for a 1-gram remnant on one side, typically left to assist in preserving the adjacent parathyroid tissue or to avoid distal RLN dissection.131 Such a remnant must be anodular and away from the cancer focus. In a total thyroidectomy, the surgeon resects all gross thyroid tissue. When thyroid tissue is left during a thyroidectomy, it is important not to just clamp across the remaining parenchyma without identification of the course of the RLN. Isthmusectomy implies complete removal of the isthmus, typically as a biopsy technique for thyroid lymphoma, anaplastic carcinoma, or Riedel’s thyroiditis, where it may be combined with tracheotomy. Less frequently, isthmusectomy may be performed for benign disease that is confined to the isthmus, such as the isthmus toxic nodule or follicular or Hurthle neoplasia. In this chapter we describe hemithyroidectomy (total unilateral, extracapsular lobectomy, and isthmusectomy), the minimum surgical procedure for unilobar WDTC, understanding that for the majority of patients with malignancy the best treatment is total thyroidectomy. Partial lobectomy for thyroid cancer is unacceptable because reoperation in a previously dissected field is technically complex and fraught with risks to the RLN and parathyroid glands.
Thyroidectomy Surgical Steps
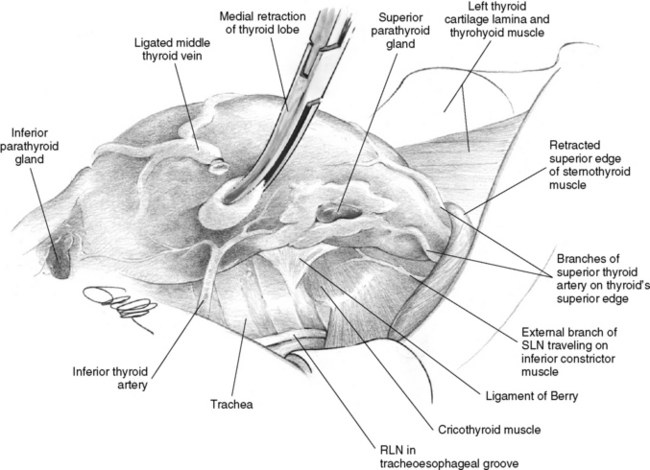
Thyroidectomy can be considered an endeavor that proceeds in a logical sequence of steps from ventral (skin of the ventral neck) to dorsal (toward the vertebral column). After dissecting the platysma muscle, the surgeon interacts with the ventral surface of the thyroid isthmus and the strap muscles, which are separated in the midline. Then next most dorsal structure encountered is the middle thyroid vein. This attaches to the lateral thyroid lobe in a ventral position (Figure 30-2). The next most dorsal anatomic structure of importance to be encountered during thyroidectomy is often the inferior parathyroid gland, which is almost always situated anterior/ventral to the RLN. Once this is dissected free from the inferior pole, the next most dorsal important structure is the recurrent laryngeal nerve. After the nerve has been identified, the final and deepest most dorsal structure of consequence during thyroidectomy, the superior parathyroid gland, is encountered. It is almost always situated at the level of the cricoid cartilage adjacent to the recurrent laryngeal nerve laryngeal entry point. We think it is helpful to consider the sequential structures encountered as one proceeds from ventral to dorsal during thyroidectomy.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree