Postoperative Uveitis
Ann M. Carrasquillo
Lucienne C. Collet
Debra A. Goldstein
Postoperative inflammation may result from various factors including exacerbation of underlying uveitis, retained intraocular foreign material after surgery, retained lens material, intraocular lens (IOL) chafing, phacoanaphylactic endophthalmitis, sympathetic ophthalmia, and infection. Differentiating noninfectious from infectious endophthalmitis is extremely important but may be quite difficult. Infectious inflammation may occur concurrently with noninfectious inflammation, making diagnosis even more challenging. If significant inflammation exists, especially in the presence of a hypopyon, possible infectious causes must be investigated. After microbial infection has been ruled out, other causes of postoperative uveitis should be considered.
Normal Inflammation
Surgical manipulation of the eye causes breakdown of the blood-aqueous barrier resulting in protein leakage and cellular reaction in the aqueous humor, manifested clinically as flare and cell. Some degree of inflammation after surgery is normal. However, what constitutes normal inflammation and what is out of the ordinary may be unclear. Knowing patient and surgical characteristics that may predispose individuals to increased inflammation following surgery is, therefore, important for postoperative diagnosis and management.
Using anterior segment fluorophotometry, Sanders et al1 demonstrated that large limbal incisions caused significant blood-aqueous barrier breakdown. Clinically, phacoemulsification has been observed to cause less postoperative inflammation than extracapsular cataract extraction.2 Oshika et al3 compared patients undergoing surgery by planned 11-mm incision extracapsular cataract extraction to patients receiving phacoemulsification with nonfoldable IOLs (7-mm incision) and foldable IOLs (4-mm incision) and found that both aqueous flare and cell count measured with the laser flare cell meter were highest postoperatively in the 11-mm incision group, with less inflammation noted in the 7-mm group and even less in the group with 4-mm incisions. In addition, flare intensity in all incision groups remained significantly higher than those of age-matched normal controls for up to 6 months following surgery.3 However, Corbett et al4 reported no difference in postoperative inflammation between extracapsular cataract extraction through a 12-mm incision or phacoemulsification with a 7-mm incision (small incision phacoemulsification was not included).
Other factors that may increase normal postoperative inflammation include a history of previous intraocular surgery, greater technical difficulty of the operation, nonwhite race, and brown iris pigmentation.4 Intraoperative factors such as iris manipulation, anterior vitrectomy, prolonged surgical time, and toxic reactions to irrigating solutions; and viscoelastic products or polishing agents on IOLs may also cause greater blood-aqueous barrier breakdown and leakage leading to more pronounced postoperative inflammation.5,6,7,8,9
Infectious Postoperative Uveitis
Early Bacterial Endophthalmitis
Despite the low frequency with which this entity is encountered, bacterial endophthalmitis remains one of the most dreaded and destructive complications of intraocular surgery. The combination of insults from the invading bacteria and the patient’s own immune system may produce significant destruction, leaving the eye with a poor visual prognosis. Prompt diagnosis and treatment are essential for achieving improved visual and functional potential. A key factor to early diagnosis is a high suspicion of endophthalmitis in postoperative cases with greater inflammation or pain than expected.
Epidemiology
Cataract surgery is the most frequently performed intraocular surgical procedure, and 90% of acute postoperative endophthalmitis follows this surgery.10
The incidence of bacterial endophthalmitis has decreased significantly over the last century. In the late 1800s, the incidence of bacterial endophthalmitis was reported to be approximately 10%.11 By the early to mid-1900s, the incidence had dropped to 0.58%,12,13,14,15,16,17,18 and by the mid-1980s, the incidence had been reduced even further to 0.074%.19 One series published by Kattan et al16 in 1990 reviewed 30,003 surgical cases performed at a single teaching hospital between 1984 and 1989. They found the incidence of culture-proven acute postoperative endophthalmitis to be 0.072% following extracapsular cataract extraction. Fisch et al20 reported the incidence of culture-proven postoperative endophthalmitis to be 0.31% following all penetrating ocular surgery and 0.32% after cataract surgery. In 1991, Javitt et al21 reported the results of postoperative endophthalmitis in a nationally representative sample by reviewing all U.S. Medicare beneficiaries undergoing cataract extraction in 1984. Among the 338,141 patients analyzed, the risk of endophthalmitis within 1 year of surgery was 0.17% for intracapsular cataract extraction (ICCE) and 0.12% for extracapsular cataract extraction (ECCE). In 1996, Norregaard et al22 reported similar results from a cross-national comparison using data from the National Danish administrative hospital register. Because of the nature of broad national record collection in these studies, however; not all cases were verified by culture. In 1996, Aaberg et al19 reported the results of a 10-year review of the incidence of culture-proven acute-onset endophthalmitis. They found the overall incidence during this period to be 0.093%. As expected, incidence varied by surgical type. Cataract surgery with or without IOL placement had an incidence of 0.082%, pars plana vitrectomy (PPV) 0.046%, penetrating keratoplasty 0.178%, secondary IOL 0.366%, and combined cataract extraction and penetrating keratoplasty 0.194%.1
However, this trend of progressively decreasing incidence of endophthalmitis seems to have recently reversed. In 2000, a large meta-analysis of endophthalmitis after cataract surgery concluded that the rate of endophthalmitis was 0.265% after 2000 compared to 0.087% in the decade of 1990.23 Subsequent studies have also demonstrated that endophthalmitis rates have increased over the past decade, primarily attributed to the practice of clear corneal incisions. It has been observed that there is a threefold to fourfold increased risk of endophthalmitis with clear corneal incisions versus scleral tunnel incision.24,25 It is possible that some of the increased incidence is also owing to the shift from superior incisions to a temporal approach. One study conducted in Sweden on 225,471 patients demonstrated a trend toward increased incidence of endophthalmitis after cataract surgery with clear corneal incisions (0.053%) compared with sclerocorneal incisions (0.036%).26 The results in terms of incision location also reflected a trend toward increased risk with temporal (0.055%) versus superior approach (0.040%).
Most early studies evaluating the incidence of postoperative endophthalmitis looked at patients undergoing surgery as inpatients or at major teaching hospitals. As more surgery is being performed at outpatient surgery centers, studies comparing the rates of endophthalmitis in the outpatient versus inpatient setting are of paramount importance. By 1984, about half of all cataract surgery performed on Medicare recipients was completed in an outpatient care setting.27 Between 1983 and 1984, Medicare implemented policy requiring that its recipients undergo outpatient cataract surgery except in special instances. In 1992, Javitt et al27 published data examining the rates of postoperative endophthalmitis encountered in an outpatient versus inpatient setting. By comparing results from their survey of Medicare recipients undergoing cataract surgery in 1984 (before changes in Medicare reimbursement) to a cohort of patients undergoing surgery between 1986 and 1987, they found a statistically significant decrease in the rate of endophthalmitis. At 1 month postoperative, the risk of endophthalmitis was 0.055% in the group undergoing outpatient surgery compared with 0.085% in the inpatient group. At 1 year, the risks were 0.081% and 0.12%, respectively.27 The two cohorts of patients evaluated in the study, however, were not entirely comparable. Antibiotic prophylaxis and operative technique including the proportion of cases done by small incision versus large incision surgery were not specified and may limit the clinical significance of the results.
Risk Factors
What causes some patients to develop endophthalmitis is unclear. Factors such as inoculum size, virulence of bacterial strain, intraoperative complications, and deficits in local or systemic host defenses likely play a role.28
There is significant evidence supporting the theory that bacterial entry through the incision during surgery is the source for organisms that produce infectious postoperative endophthalmitis.29,30 In 1989, Sherwood et al29 cultured fluid aspirated from the anterior chamber and drained from the conjunctival sac during cataract surgery in 101 patients. They demonstrated bacterial growth by culture of aspirate fluid from 29 patients and from the conjunctival sac of 90 patients.29 In addition, in all patients, conjunctival fluid stained with fluorescein flowed into the anterior chamber during irrigation and aspiration performed during extra-capsular cataract surgery. They demonstrated that this fluid is contaminated with potentially pathogenic organisms, yet none of the patients in their study developed endophthalmitis.
Additionally, it has been reported that intraoperative contamination of the anterior chamber with bacteria occurs in 2% to 22% of phacoemulsification cases and 24% to 43% of extracapsular cataract extraction procedures.29,31,32,33,34,35,36
Menikoff et al6 conducted a case-control study of 24,105 patients undergoing intraocular surgery between 1988 and 1990 to identify risk factors for postoperative endophthalmitis. One factor identified as a significant independent risk for the development of endophthalmitis was intraoperative communication with the vitreous cavity.6 This complication was associated with a risk ratio of 13:7.6 Likewise, in their study of Medicare beneficiaries undergoing cataract extraction, Javitt et al21 found that surgery accompanied by anterior vitrectomy was associated with more than a fourfold increase in the development of postoperative endophthalmitis at 1 month compared with patients undergoing cataract surgery alone. Several other studies have also found a higher incidence of endophthalmitis after vitreous communication has been introduced.7,22,28,37,38
Vitreous communication seems to be important in the development of postoperative endophthalmitis for several reasons. Removal of potentially pathogenic bacteria from the vitreous cavity appears to be less efficient than from the anterior chamber.9,28,39 Vitreous cultures have been demonstrated to yield a significantly higher percentage of confirmed growth and higher colony counts compared with aqueous.16,40,41,42 It has been suggested that vitreous may be more supportive of bacterial growth than aqueous fluid and that the viscous properties of vitreous may retard bacterial clearance.7,28,39,42
Certain types of IOLs may also be associated with an increased risk of infectious endophthalmitis. Sherwood et al29 showed that fluid bathing the conjunctiva during surgery is contaminated with potentially virulent bacteria. IOLs may touch the ocular adnexa during lens insertion. Vafidis et al43 demonstrated that factors such as electrostatic charges may cause bacteria to adhere to IOLs, which allows bacteria to be carried into the eye with the lens or to adhere after IOL insertion when contaminated irrigation fluid flows through the anterior chamber.29 Several investigators have shown that bacteria adhere preferentially to lenses with haptics constructed of polypropylene compared with polymethylmethacrylate.6,44 Similarly, transscleral fixation of posterior chamber IOLs with exposed polypropylene sutures has also been associated with an increased risk of infectious endophthalmitis.6
Numerous other factors have also been identified as increasing the risk of postoperative endophthalmitis. Increased intraoperative instrumentation and duration of the operative procedure place patients at increased risk.6,7,9 This finding may be partly attributable to complicated cases that require anterior vitrectomy, a factor that is independently associated with an increased risk of infection. Menikoff et al6 found patients with a history of drug allergy to be at significantly increased risk of developing endophthalmitis. The authors hypothesized that these patients may have a form of atopy, which is often associated with denser bacterial colonization of the adnexa and an altered immune response. Several studies have suggested that diabetics are at greater risk of developing infectious endophthalmitis.16,20,37,45 This finding may be secondary to an impaired immune system in many of these patients. Similarly, advanced age has been associated with a greater risk of endophthalmitis. As in patients with diabetes, this finding may be explained by a relative diminution of host defenses.22
Clinical Features
A key factor in the prognosis of early postoperative endophthalmitis is prompt diagnosis and treatment. Although the ophthalmologist rarely encounters this entity, it must be kept in mind in the differential diagnosis of a postoperative patient with excessive inflammation or pain. Early postoperative endophthalmitis usually presents within the first few days after surgery. In their 10-year review of the incidence and outcomes of cataract surgery at a single institution, Aaberg et al19 reported a range of 1 to 40 days, with a median of 5 days, from the initial procedure to the development of acute postoperative endophthalmitis. Kattan et al16 reported similar findings in 1990, with a range from 1 to 40 days and an average of 8 days. Results from the Endophthalmitis Vitrectomy study, a multicentered randomized prospective clinical trial of 420 patients at 24 centers with clinical evidence of postoperative endophthalmitis following cataract extraction or secondary IOL implantation, reported a median time of 6 days until patient presentation to a study center.45 Of these patients, 24% presented within 3 days of the initiating procedure, 37% within 4 to 7 days, 17% within 8 to 13 days, and the remaining 22% presented within 2 to 6 weeks.45
Common findings include pain, conjunctival injection, lid edema, and decreased vision. Signs and symptoms are variable, however, and they often depend on the virulence of the underlying organism.28 In the Endophthalmitis Vitrectomy study, blurred vision was the most common symptom.45 In that study, 86% of patients presented with a visual acuity of less than 5/200. Pain was reported in 74% of patients.45 Other signs include hypopyon, inflammatory pupillary membrane, vitreous opacification and inflammation, anterior chamber cell and flare, and a diminished red reflex 28,45,46 (Fig. 55.1). Less common findings may include an afferent papillary defect and corneal ulcer or infiltrate.45 Again, these findings vary and likely depend on the underlying microorganism as well as the individual’s immune response.
Etiology
It is generally agreed that the greatest source of bacteria causing postoperative endophthalmitis originates from the patient’s own eyelid and adnexal flora.16,20,47,48,49 The ocular adnexa has been shown to be densely colonized with organisms commonly implicated in the etiology of postoperative endophthalmitis.47,50,51,52 Ariyasu et al49 demonstrated that organisms cultured intraoperatively at the time of wound closure had identical typing and antibiotics sensitivities to organisms isolated from the eyelids and conjunctiva. By using restriction enzyme endonuclease studies, Speaker et al48 found that 82% of organisms isolated from the vitreous of patients with postoperative endophthalmitis were genetically indistinguishable from isolates recovered from the patient’s eyelid, conjunctiva, and nose.
Gram-positive, coagulase-negative micrococci, including Staphylococcus epidermidis, are responsible for most cases of postoperative endophthalmitis.16,19,20,28,37,45,49,53 Kattan et al16 reported that 78% of organisms cultured following cataract surgery between 1984 and 1989 consisted of Gram-positive species. More cases were attributed to S. epidermidis than to any other species. In their group of all patients with culture-positive endophthalmitis from 1990 to 1994, Aaberg et al19 found 76.7% of patients grew Gram-positive organisms, 47.8% of which were S. epidermidis. In the Endophthalmitis Vitrectomy study (EVS), Gram-positive, coagulase-negative micrococci represented 70% of the 323 isolates meeting the study criteria for confirmed growth.53 This group consisted almost entirely of S. epidermidis with a single isolate of Micrococcus kristinae.53 The next most common group consisted of other Gram-positive species, including S. aureus (9.9%), 29 isolates of Streptococcus species (9.0%), and 7 isolates of Enterococcus species (2.2%). The least common group of organisms consisted of Gram-negative species, representing only 5.9% of isolates.53 Polymicrobial growth occurred in 9.3% of the EVS patients. In this group, two species of Gram-positive, coagulase-negative micrococci grew in more than half of patients.53 It is clear that organisms typically found colonizing the ocular adnexa are not as benign as once believed51,54 and play a significant role in the pathogenesis of postoperative endophthalmitis.
Methicillin-resistant Staphylococcus aureus (MRSA) endophthalmitis
S. aureus may develop resistance to antibiotics, primarily by the production of β-lactamase.55 Methicillin is not inactivated by β-lactamase and was traditionally used to treat resistant strains of S. aureus.56 However, in 1961 methicillin-resistant S. aureus (MRSA) were first detected,57 and today MRSA is a common cause of nosocomial infection.58 MRSA wound infection has been reported with clear cornea incisions in phacoemulsification, and one of the patients presented with endophthalmitis.59,60 Other studies have reported MRSA endophthalmitis following cataract extraction and vitrectomy.61,62,63
Diagnosis
One of the most important aspects in the prognosis of postoperative endophthalmitis is early diagnosis and prompt treatment. This requires maintaining a high level of suspicion for patients who are at greater risk preoperatively and for patients who have greater postoperative inflammation than expected. If postoperative inflammation is disregarded and other signs and symptoms of endophthalmitis go unnoticed, critical time may be lost and diagnosis delayed.
In the EVS, 24% of patients presented within 3 days of the surgical procedure; 61% presented within 1 week.45 These figures emphasize the importance of early and frequent postoperative examinations, especially in complicated surgical cases. Patients should be educated regarding the normal postoperative course, counseled about warning signs such as excessive pain and decreased vision, and instructed to call their physicians promptly if these symptoms develop.
In cases of suspected endophthalmitis, a careful history and examination are of utmost importance. Systemic diseases such as diabetes mellitus, which might put the patient at increased risk for the development of endophthalmitis or might point to a source of endogenous infection, should be noted. Factors such as antibiotic and steroid use and systemic immunosuppression should also be determined. Details regarding the operative course such as duration of the case, presence of capsular rupture, or vitreous loss are important.6,64 Physical examination should include a careful determination of visual acuity as this influences the immediate course of treatment.45 The pupils should be examined and any afferent pupillary defect noted. Findings such as the degree of anterior chamber inflammation, the presence of a hypopyon or corneal infiltrate, and clarity of the vitreous should be noted. Loss of vision, vitreous opacification, and the presence of a hypopyon are some of the findings consistent with acute postoperative infection.28 In general, if a hypopyon is present, one needs to assume that the source is infectious until proven otherwise. The integrity of the incision should be assessed by looking for signs of wound leak, vitreous wicks, and the presence of any suture material that might provide an infectious tract. The conjunctiva should be inspected for evidence of a bleb and the iris examined for rubeosis. IOL position, stability, and capsular integrity should be noted. The capsule should also be examined for evidence of white plaques, which may suggest an infectious process, particularly in late postoperative endophthalmitis. Retained lenticular material should also be noted, as this can result in inflammation, which may mimic infectious endophthalmitis. This is particularly important if the posterior capsule is no longer intact. Ultrasound can be useful in the diagnosis of retained nuclear material, which may not be visualized on clinical examination. Examination of the posterior segment should include noting the presence or absence of a red reflex, degree of retinal vascular visualization, retinal detachment, choroidal detachment, or vitreous membrane. Retinal detachments are difficult to repair in patients with underlying endophthalmitis and usually confer a poor visual prognosis.46 When the posterior segment cannot be properly visualized, B-scan ultrasound should be performed.
Once endophthalmitis is suspected and a thorough history and physical examination completed, diagnosis is confirmed by completion of appropriate cultures. External cultures of the eyelid margins and conjunctiva may be obtained. Both aqueous and vitreous cultures should be obtained before injection of any antibiotics or steroids. A vitreous sample is especially important because many aqueous samples demonstrate negative growth, even in cases of confirmed infectious endophthalmitis. In the EVS, confirmed growth on culture was demonstrated from 54.9% of vitreous samples compared with only 22.5% of aqueous samples.42 In cases in which a vitreous sample was positive, Bode et al41 demonstrated negative growth in up to 57% of aqueous cultures. Given these findings, a vitreous sample is imperative for adequate diagnosis.
Vitreous specimens can be obtained either by trans–pars plana needle aspiration or by vitreous biopsy with a vitrectomy unit. In the EVS, vitreous was cultured by one of three methods: needle aspiration, vitreous biopsy with the vitrector, or vitreous biopsy followed by vitrectomy (vitreous effluent was also cultured in these cases). No statistically significant difference was found in the yield among the three groups.42 However, some authors have suggested that a vitreous culture using a vitrector unit may be superior because a higher inoculum is obtained and a larger specimen is collected.65 This may aid in culture positivity as well as assist in clinical recovery by removing more infectious organisms from the eye. In addition, aspiration of vitreous with a syringe is often difficult and can place traction on the retina.28 If needle aspiration is attempted and fails, a vitreous biopsy using a vitrectomy unit should be performed.
The EVS concluded that patients who presented with initial vision of only light perception who underwent a vitrectomy had a three times greater chance of achieving 20/40 final visual acuity (33% vs. 11%) compared with patients who had only a vitreous tap or biopsy. There was no difference in outcome between vitrectomy and vitreous tap in patients with endophthalmitis who presented with initial visual acuity of hand motions or better.45
When vitrectomy is performed for biopsy and culture, an initial undiluted specimen should be obtained. This may be achieved in the following manner. The vitrector is set up but not primed. The infusion cannula is inserted but not turned on. The vitrector is connected to a 5-cc syringe, and set at low cut rate (e.g., 600–800 cpm) to minimize damage to cells. As the surgeon is performing the vitrectomy, the assistant slowly aspirates into the syringe to collect the pure vitreous aspirate (no infusion); 1.5 cc to 2.0 cc of vitreous can often be obtained before collapse of the globe occurs. Once the pure vitreous aspirate has been obtained, the infusion can be turned on. Additional vitrectomy (core or complete) can be performed at regular speed (e.g., 1,200–1,500 cpm). The vitreous washing (collected through the vitrectomy system) may also be sent for analyses. This same technique may be used when doing diagnostic vitrectomy for other indications, such as suspected intraocular lymphoma.
The fluid from the cassette may be processed through filter paper31 or be centrifuged to produce a concentrated specimen.28 After specimens have been collected, samples are cultured on appropriate media. These include blood, chocolate, thioglycolate broth, and Sabouraud dextrose agar, all incubated at 37°C. Gram and Giemsa staining should also be performed. The presence of a positive result on Gram stain has been associated with a higher incidence of culture positivity.42 In the EVS, the positive predictability of the Gram stain was greater than 94%.42 A positive culture is often defined as at least semiconfluent growth on a solid medium, any growth on two or more media, or growth on one medium with a positive Gram stain.45,66
Detection of bacterial DNA using polymerase chain reaction (PCR) may be considered for cases in which infectious endophthalmitis is clinically suspected but in which negative cultures have been obtained.67,68 Okhravi et al67 reported a 100% concordance rate between PCR- and culture-positive bacterial samples and demonstrated that the technique is useful for detecting infection in cases that are culture negative.
In a study by Chiquet et al,69 conventional culture and PCR were performed on samples of aqueous humor and vitreous in 30 patients with endophthalmitis. Aqueous cultures were positive in 32% of cases and PCR in 61% of cases. Microbiologic identification was successful in 71% of cases.
PCR analysis offers several advantages over culture; it allows for faster microbiologic diagnosis than conventional culture, has higher sensitivity, can be helpful in patients previously treated with antibiotics, and is useful for the identification of resistant microbes.70
Treatment
The aims in treating patients with postoperative endophthalmitis include sterilization of the eye and control of inflammation.
Intravitreal antibiotics are the initial treatment of choice for postoperative endophthalmitis.28 Two drugs are typically used. One should have broad and effective activity against Gram-positive species, specifically coagulase-negative staphylococci, and the other should protect against Gram-negative bacilli. In the EVS, intravitreal vancomycin and amikacin were administered to all patients and demonstrated effective antimicrobial activity against most pathogens.45,64 Vancomycin is the traditional drug of choice for Gram-positive coverage. It is the only agent tested to which all Gram-positive species in the EVS were susceptible.64 Development of vancomycin-resistant strains of staphylococcal and streptococcal species has prompted the Centers for Disease Control and Prevention (CDC) to recommend reserving use of vancomycin for treatment of serious infections produced by b-lactam–resistant Gram-positive microorganisms.71 Cefazolin (Ancef) has activity against many Gram-positive organisms. However, in a rabbit model of S. aureus endophthalmitis, cefazolin was less effective than vancomycin and clinical resistance of Gram-positive organisms is frequent.72,73 In light of the absence of data suggesting an effective alternative to vancomycin, and the potentially devastating consequences of endophthalmitis, vancomycin remains the agent of choice for the coverage of Gram-positive organisms in cases of postoperative endophthalmitis. Based on susceptibility data from the EVS, vancomycin in combination with amikacin, ceftazidime, or ciprofloxacin are appropriate antibiotic choices.64 Many practitioners have moved away from using gentamicin because it is more toxic than amikacin in experiments using rabbit eyes. Additionally, macular infarction has been reported after its administration in patients.74,75
Current recommendations for empirical therapy are vancomycin 1.0 mg/0.1 mL and ceftazidime 2.25 mg/0.1 mL. Fourth-generation fluoroquinolones may also be used. The dosage calculated from experimental studies on rabbits to be used intravitreally in human eyes is 400 μg/0.1 mL of moxifloxacin or gatifloxacin.76,77 These drugs are broad spectrum but are most active against Gram-negative bacteria. Since Gram-positive bacteria in endophthalmitis have shown increasing resistance rates (40%),78 fourth-generation fluoroquinolones are used in concert with an antibiotic effective against Gram-positives (e.g., vancomycin).
Periocular antibiotics may be used as an adjunct to intravitreal therapy.45 Subconjunctivally injected antibiotics have poor intravitreal penetration79 but can achieve high concentrations in the anterior segment. Similarly, topical antibiotics provide poor intravitreal penetration but can achieve high concentration in the anterior segment and help sterilize wound sites.64 It is still unclear whether subconjunctival and topical antibiotics offer additional benefit in the treatment of postoperative endophthalmitis.
The role of corticosteroids in the treatment of postoperative endophthalmitis is still somewhat controversial. In the EVS, corticosteroids were administered to all patients subconjunctivally, topically, and orally, although the study was not designed to determine the efficacy of this adjunctive therapy. Corticosteroids play an important role in controlling the inflammatory response after infection, which may be further damaging to the eye. Corticosteroid use intravitreally may help control intraocular inflammation, especially after bacterial death from antimicrobials. Unless fungal endophthalmitis is suspected, some practitioners recommend the use of intravitreal steroids in conjunction with intravitreal antibiotics.28,80 Several studies have suggested that administering intravitreal corticosteroids may be beneficial,81,82,83 although at least one study found that intraocular steroid use was not associated with a significant effect on visual acuity.19 No prospective, controlled clinical trials have yet proved the efficacy of intravitreal corticosteroids in the management of postoperative endophthalmitis.
Before the results of the EVS, systemic antibiotic use was considered a standard treatment for postoperative infectious endophthalmitis.45 The EVS concluded, however, that there was no difference in visual acuity or media clarity outcome with or without the use of systemic antibiotics.45 These results were important for several reasons. Systemic antibiotics may have significant adverse effects, and their use is not without risk. Systemic antibiotics are also expensive. Not only do the drugs incur expense, but the agents used to treat endophthalmitis used to require IV administration, and therefore required either admission to a hospital or home-based nursing for their administration. The results of the EVS may need to be re-evaluated, however, in light of new fluoroquinolone antibiotics that have better intraocular penetration compared with that of other antibiotics.9,84 Orally administered fluoroquinolones such as the fourth-generation moxifloxacin and gatifloxacin can reach levels in the aqueous and vitreous that are higher than the minimal inhibitory concentration (MIC) for most microbes implicated in endophthalmitis.85,86 These agents may therefore have a role as adjunctive therapy of postoperative endophthalmitis.
Of utmost importance in the treatment of postoperative endophthalmitis is prompt initiation of therapy. This point cannot be overemphasized. Endophthalmitis is an ocular emergency, and initiation of treatment should reflect this.
Prognosis
Despite the fear provoked by a diagnosis of postoperative endophthalmitis, final visual acuity can be surprisingly good. In general, the worse the acuity at initial presentation, the worse the final visual outcome.64 In the EVS, 41% of patients achieved visual acuity of 20/40 or better at 3 months. By the end of the study, more than half of patients (53%) achieved visual acuity of 20/40 or better and 74% achieved 20/100 vision or better. Only 11% of patients had acuity worse than 5/200.45 Other studies have found similar results. Kattan et al16 found that approximately one third of patients achieved a final visual acuity of 20/60 or better following endophthalmitis after cataract extraction with or without IOL placement. Speaker and Menikoff37 found 64% of patients with endophthalmitis after intraocular surgery had a visual acuity of 20/100 or better after treatment.
Final visual acuity after postoperative endophthalmitis varies according to surgical type. Aaberg et al19 found the median visual acuity after endophthalmitis treatment was 20/200 overall. Post–cataract extraction endophthalmitis patients had a median final acuity of 20/133, and the best median visual acuity, 20/40, was obtained after secondary IOL placement. Patients with post-PPV endophthalmitis fared worst, with a median acuity of no light perception.
The EVS and other studies have found certain risk factors associated with a worse visual acuity outcome after treatment for postoperative endophthalmitis. According to the EVS, the most significant independent risk factor for decreased vision was visual acuity of light perception or worse at initial presentation.45 Other independent risk factors included advanced age, diabetes, presence of a corneal infiltrate and/or ring ulcer, intraoperative violation of the posterior capsule, low or high intraocular pressure, presence of an afferent pupillary defect, rubeosis, and absent red reflex.45,64 Studies have demonstrated that more virulent organisms, including S. aureus, Streptococcus species, and Gram-negative rods are often associated with worse visual prognosis.20,28,87 A shorter time interval between the onset of symptoms and initiation of treatment also plays a role in visual outcome following postoperative endophthalmitis.87 In the EVS, initiation of treatment was begun within 6 hours of the initial exam.45 Visual results after treatment were better in the EVS than in many other studies.16,19,37,87 This may have resulted in part from efficient initiation of treatment, underscoring the importance of prompt diagnosis and treatment. The first few hours in the management of postoperative endophthalmitis are critical.
Prevention
Preventive measures play an important role in reducing the incidence of postoperative endophthalmitis. Preparation of the surgical field with sterile drapes does not eliminate the risk of infection, but it can reduce it.88 Adhesive drapes, which cover the eyelid margin and lashes, may be particularly important in reducing the chance of infection from microbial contaminants of the ocular adnexa.
Various topical antiseptics have been used for the preoperative preparation of the eye. Argyrol, a mild silver protein solution, was used for many years. In 1983, however, Isenberg et al89 demonstrated that the chemical preparation was no more effective than no treatment in reducing the number of bacteria. In addition, the same investigators evaluated the effect of conjunctival irrigation in the preoperative chemical preparation of the eye.90 They determined that both the bacteria colony count and species count increased following irrigation with saline solution compared with nonirrigated eyes.
Povidone-iodine has been shown to have significant antimicrobial effect.37,91,92,93,94,95,96 Povidone (polyvinylpyrrolidone) is a high-molecular-weight, water-soluble polymer that forms a complex with iodine.95 After instillation, free iodine is released, which has been credited with the compound’s antimicrobial effect.95,97 Povidone-iodine possesses minimal tissue toxicity and has a broad antimicrobial effect with activity against bacteria, viruses, fungi, protozoa, and spores.94,95 Maumenee and Michler98 first studied the use of iodine solution in ophthalmic surgery in the early 1950s. They found it possessed an antimicrobial effect on the ocular adnexa when the solution was used concomitantly on the skin. In 1984, Apt et al95 demonstrated that eyes treated with half-strength povidone-iodine solution preoperatively had a 91% decrease in bacteria colony counts and a 50% decrease in the number of species compared with controls. The solution did not cause significant conjunctival hyperemia, and no allergic or adverse reactions were reported. Speaker and Menikoff37 demonstrated that the preoperative application of 5% povidone iodine to the ocular surface significantly lowered the incidence of culture-positive endophthalmitis after intraocular surgery compared with eyes prepared with a silver protein solution. Among more than 3,000 cases in which povidone-iodine was used, the solution was well tolerated and no adverse reactions were noted.37
In most studies of povidone-iodine, a 5% solution is prepared by diluting full-strength 10% solution 1:1 with saline solution.28,37,95 Two drops of the solution are instilled in the conjunctival sac, and the eyelids are gently manipulated to distribute the solution over the surface of the eye. The solution bathes the eye while the rest of the preparation is completed. The solution should remain in contact with the eye for several minutes to exert its antimicrobial effect. The skin, eyelid margins and lashes are then scrubbed with povidone-iodine–soaked gauze or cotton swabs. Finally, the eye is irrigated with saline solution at the end of the preoperative preparation. This step is important to avoid intraocular toxicity with the solution.37 Povidone-iodine solution should not be confused with povidone-iodine scrub. The latter contains a detergent, which is toxic to the eye and should be avoided.37
The use of perioperative antibiotics in ophthalmic surgery remains controversial. Short-course topical antibiotics administered before surgery reduce the amount of lid and conjunctival bacteria.96,99,100 The amount of pathogen reduction depends on the type of antibiotic used, the frequency and duration of its use, and the type of bacteria present and the susceptibilities of the organism.100
Inasmuch as the major infectious source for postoperative endophthalmitis is the patient’s own eyelids and conjunctiva,9 it is reasonable to attempt to reduce the number of organisms with prophylactic antibiotics. Up to three quarters of patients carry organisms on the ocular adnexa capable of causing postoperative endophthalmitis.51 In 1964, Allen and Mangiaracine101 suggested that preoperative topical antibiotics reduced the incidence of endophthalmitis. At that time, chloramphenicol and gentamicin were routinely used as antibiotics. Studies using these antibiotics have shown variable efficacy.102,103,104 More recently, studies using fluoroquinolone antibiotics have reported favorable results. This family of antibiotics has broad-spectrum antimicrobial activity, low rates of resistance, and minimal toxicity.105 Donnenfeld et al105 demonstrated that patients given topical 0.3% ofloxacin 30 and 90 minutes preoperatively had aqueous humor drug levels that approached the minimal inhibitory concentration of 90% of Staphylococcus epidermidis, S. aureus, and most Gram-negative organisms. Von Gunten et al84 reported similar results. Snyder-Perlmutter et al106 reported that patients receiving topical antibiotics every 5 minutes for three doses had a statistically significant reduction in bacterial colony-forming units following treatment with ciprofloxacin but not ofloxacin.
An online survey of the members of the American Society of Cataract and Refractive Surgery showed that most surgeons prefer preoperative and postoperative topical antibiotic prophylaxis with the latest generation of topical fluoroquinolones.107
A recent study on the use of topical antibiotics used as prophylaxis in cataract surgery reported the outcome of 20,013 uncomplicated cataract extractions. 81% received perioperative gatifloxacin and 19% perioperative moxifloxacin. The overall rate of endophthalmitis was 0.07%, with no statistical difference between the two agents in rate of endophthalmitis.108
Although studies suggest that topically applied antibiotics decrease intraocular bacterial load, the low incidence of endophthalmitis makes it difficult to prove that a direct correlation exists between topical antibiotic use and a reduction in the incidence of infectious postoperative endophthalmitis.
Subconjunctival antibiotics have been used since the 1950s for infectious prophylaxis following ocular surgery.109 During this time their use has been a source of continued controversy. Administration of subconjunctival antibiotics circumvents the corneal epithelial barrier and permits a high concentration of drug to penetrate the corneal stoma and diffuse into the aqueous humor.9 Studies have demonstrated good penetration of antibiotics into the anterior chamber following subconjunctival administration.64,110 A subconjunctival injection of antibiotics can achieve bactericidal levels in the anterior chamber thus eliminating most infection.9 It has been suggested, however, that high intraocular levels of antibiotic achieved with subconjunctival injection might only partially treat an infection in some cases and delay onset of symptoms and diagnosis leading to a poor outcome.111 Additionally, the concentration of anterior chamber antibiotic decreases rapidly after administration, and vitreous penetration following subconjunctival administration of antibiotics is also poor, limiting its efficacy.9,28,79,100
Studies evaluating the efficacy of subconjunctival antibiotics in decreasing the incidence of endophthalmitis have been inconclusive. In 1967, Cassady38 published an uncontrolled series of 1,212 cataract extractions in which subconjunctival antibiotics were used. No cases of postoperative endophthalmitis were reported. Cassady attributed his success to the use of subconjunctival antibiotics and justified their usage after every cataract extraction. During that same year, a controlled, prospective study was reported evaluating subconjunctival antibiotic use following cataract surgery.112 Two cases of endophthalmitis were reported in both the control and treatment groups. Unfortunately, the small number of infections prevented any conclusions from being drawn regarding the statistical significance of antibiotic prophylaxis. In another study, Kolker et al113 reported a 1.42% incidence of postoperative endophthalmitis in patients given no prophylaxis versus 0.21% in a similar group given subconjunctival penicillin and streptomycin. In two series reviewing postoperative endophthalmitis, all but one patient had received postoperative subconjunctival antibiotics.16,37
Aminoglycosides were commonly used for subconjunctival injection, but such usage has fallen into disfavor. Gentamicin has been associated with more conjunctival toxicity and greater patient discomfort than cefuroxime.114 Aqueous penetration of gentamicin is poorer than that of cephalosporins, and more species that are implicated in the etiology of postoperative infectious endophthalmitis are sensitive to cephalosporins than to aminoglycosides.114 Retinal toxicity has also been demonstrated following aminoglycoside use. In 1991, a survey of members of the Retina, Macula and Vitreous Societies revealed 93 cases of macular infarction following administration of gentamicin sulfate with 8 additional cases secondary to other aminoglycosides.75 Most reports followed planned intravitreal injection of antibiotic, but 23 cases occurred in eyes treated with prophylactic subconjunctival antibiotics after routine ocular surgery. Of these cases, most occurred following known or presumed ocular penetration; at least two cases were thought to be associated with drug leakage into the eye through the cataract incision following injection. Aminoglycosides have an extremely narrow therapeutic window, making their use for routine surgical prophylaxis unwise.75 If even a small amount of gentamicin dosed for subconjunctival use gains entry to the eye, retinal toxicity may occur.109 Compochiaro and Conway75 pointed out that endophthalmitis is treatable whereas macular infarction is not; they thus recommended that use of prophylactic subconjunctival aminoglycosides be abandoned following routine ocular surgery and subconjunctival use of other antibiotics be administered only after serious consideration. Subconjunctival injections of antibiotics are rarely performed following cataract surgery in the era of clear cornea surgery.
Intracameral antibiotic use, including antibiotic administration in irrigating solutions and through direct injection, is another method of infectious prophylaxis. Preliminary toxicity studies have been done in animals, but difficulty has been encountered when equivalent antibiotic doses are applied in humans.115,116 This has resulted in disagreement regarding clinical doses and formulations of prophylactic intracameral antibiotics.117,118,119 An uncontrolled study by Gills117 reported one case of endophthalmitis in 20,000 cataract extractions following gentamicin use in irrigating solution and one case in 9,928 after intracameral vancomycin. No cases were reported following 25,000 operations in which both gentamicin and vancomycin were used in irrigating solution. In another uncontrolled study, Gimbel et al119 reported no cases of endophthalmitis following 4,684 cataract extractions in which both intracameral gentamicin and vancomycin were used. Those authors reported no adverse effects on corneal endothelium when cases were compared to similar series following phacoemulsification with lens implantation and no intracameral antibiotics, but endothelial damage may have been avoided because vancomycin was delivered into the capsular bag by intracameral injection after lens placement rather than continuously infused with the irrigating solution. Beigi et al118 reported that the addition of gentamicin and vancomycin to irrigation solution during phacoemulsification significantly reduced contamination of anterior chamber aspirates with bacteria. No patients in the study developed postoperative endophthalmitis, however, making it impossible to determine whether intracameral antibiotic use decreased the incidence of postoperative infection. One should also note that the CDC has cautioned against the routine use of vancomycin.71
The European Society of Cataract and Refractive Surgeons (ESCRS) performed a large randomized controlled study evaluating risks for postcataract endophthalmitis. One variable was use of intracameral cefuroxime, which resulted in a significant decrease in the incidence of postoperative endophthalmitis (0.07% with intracameral cefuroxime, 0.34% in the control group).120
In another study conducted in Manila, Philippines, prophylactic 0.1 mL intracameral moxifloxacin 0.5% ophthalmic solution resulted in no signs of toxicity on visual function, anterior chamber reaction, pachymetry, and corneal endothelial cell density.121
There are currently no commercially available preparations of antibiotics for intracameral use, which raises concern for many surgeons. A survey of members of the American Society of Cataract and Refractive Surgery (ASCRS) published in 2007 reported that most surgeons use preoperative and postoperative topical antibiotic prophylaxis, with most opting for topical fluoroquinolones. Seventy-seven percent of respondents did not routinely inject intracameral antibiotics, but 82% reported that they would likely do so if a commercial preparation were available.122
Delayed-Onset Bacterial Endophthalmitis
The signs and symptoms of delayed-onset endophthalmitis can be subtle, often mimicking other causes of inflammation and making diagnosis difficult.
Definition
Many terms have been used to describe this entity including delayed-onset, chronic, and late postoperative endophthalmitis. A consensus regarding the time period in which this disorder occurs has also been disputed. Some authors have suggested that symptoms starting more than 1 month postoperatively should be considered chronic endophthalmitis, whereas other authors have defined chronic endophthalmitis as requiring two or more episodes of low-grade postoperative infectious uveitis regardless of the time of onset.123,124,125 The Endophthalmitis Vitrectomy Study considered postoperative endophthalmitis acute if it occurred within 6 weeks after surgery.45 For our purposes, we will define delayed-onset postoperative infectious endophthalmitis as uveitis that develops more than 6 weeks after surgery with evidence of an infectious process. This definition is somewhat arbitrary and merely serves as a guideline for classification purposes.
Epidemiology
Most data estimate incidence of acute postoperative infectious endophthalmitis or do not distinguish between acute and chronic infections when reporting statistics of incidence.16,19,20,22,123 Unfortunately, this makes it difficult to assess the frequency with which delayed-onset endophthalmitis is encountered. In addition, studies have evaluated risk factors associated with acute postoperative endophthalmitis, but data are lacking with regard to infection of delayed onset.6,123
Etiology
Propionibacterium acnes was the first bacterial organism reported to cause chronic low-grade inflammation after intraocular surgery.123,126 In 1986, Meisler et al127 characterized a new syndrome of delayed-onset endophthalmitis and reported six cases of chronic postoperative endophthalmitis masquerading as chronic iridocyclitis from which Propionibacterium species were cultured. Other bacteria can cause a similar picture of chronic low-grade inflammation, including coagulase-negative Staphylococcus,128,129,130 Staphylococcus aureus,131,132 anaerobic Streptococcus,133 Corynebacterium species,128 Actinomyces,134,135 and Nocardia species.136 Various fungi, particularly Candida species, can produce a similar picture.128,137,138 Despite the array of organisms implicated in delayed-onset postoperative endophthalmitis, P. acnes is most often encountered.
P. acnes is a Gram-positive, non–spore-forming anaerobic pleomorphic bacillus that has been implicated in a wide variety of systemic infections.124 It is a ubiquitous organism, which inhabits the human sebaceous follicle.134 It has been cultured in up to 43.8% of normal human conjunctivae.54 In addition to chronic endophthalmitis, ocular and periocular infections from P. acnes have included preseptal cellulitis, canaliculitis, dacryocystitis, conjunctivitis, and keratitis.139 Several cases of P. acnes–induced acute postoperative endophthalmitis have also been described.125,134
P. acnes cell wall resists digestion by macrophages and polymorphonuclear cells, which allows it to persist sequestered in inflammatory cells within the eye.123,133,140 P. acnes can also live within the lens capsule, protected from inflammatory cells and attempts to sterilize the eye with antibiotics.141 Release of organisms sequestered within the capsule may explain the onset of endophthalmitis that has been reported after neodymium/yttrium-aluminum-garnet (Nd:YAG) capsulotomy.127,141,142
Clinical Features
Patients with delayed-onset postoperative endophthalmitis may present with signs and symptoms typically found in patients with acute endophthalmitis. Most often, however, they present with low-grade chronic and often recurrent inflammation that may simulate noninfectious uveitis. Such inflammation occurs over the course of months compared with the abrupt inflammatory presentation seen in acute postoperative infections. Patients may initially show some improvement following corticosteroid treatment, which hinders the correct diagnosis of endophthalmitis. Another finding that separates this entity from acute endophthalmitis is development of granulomatous precipitates on the corneal endothelium and IOL.124,127,142,143
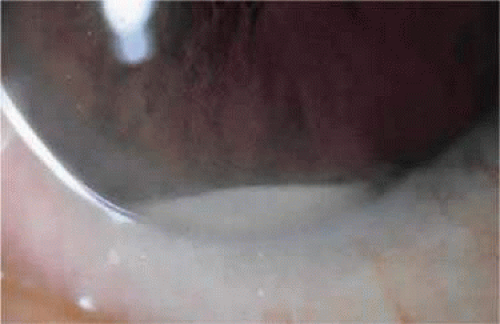
The finding of white plaques on the posterior capsule is suggestive of P. acnes endophthalmitis,123,124,133,144,145 although similar plaques have also been described in other causes of bacterial and fungal endophthalmitis.146,147 The plaque is usually located between the IOL and the posterior capsule or peripherally on the capsule, requiring adequate dilation for visualization124,133 (Fig. 55.2). Histologically, the plaque consists of a mixture of lens material and colonies of microorganisms.124,133 Other clinical findings may include hypopyon, a fibrinous reaction in the anterior chamber manifested as beaded fibrin strands, as well as vitritis and cystoid macular edema.127,134,141,142 Patients usually develop symptoms between 1 and 10 months after surgery, although the time elapsed to culture-proven infection is often delayed owing to initial misdiagnosis.127,128,134,141,142,148
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree