Chapter 51 Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer
Introduction
The majority (75% or more) of follicular cell–derived thyroid carcinomas (FCDTCs) retain the capacity of the normal thyroid follicular cell to take up and to concentrate iodine.1 This iodine concentration is less efficient than that seen in the normal thyroid gland, because of the abnormal architecture of the follicular structures within the cancer, which impairs organification and retention of the isotope, explaining why cancers are typically seen as “cold” nodules on isotopic imaging.2 Nevertheless, this retained differentiated function permits radioactive isotopes of iodine to be used for both the localization and treatment of residual thyroid carcinoma.3,4
When administered orally, all isotopes of iodine are absorbed rapidly and reliably from the upper gastrointestinal tract, circulate transiently in the bloodstream, and are concentrated in tissues that express a functional sodium-iodide transporter (NIS).5 The remaining isotope is filtered and excreted through a renal route, with consequent radiation exposure to the entire urinary tract. Tissues that actively concentrate iodine include normal and cancerous thyroid tissue, salivary gland, breast (particularly during lactation), stomach, kidney, and colon.5,6 The uptake of iodine into both normal and malignant thyroid tissue, though not most other tissues, depends on the activity of the thyroid-stimulating hormone (TSH) receptor, which up-regulates expression and increases the activation of NIS in thyroid tissue.7 In addition, thyroid tissue is capable of iodine organification onto thyroglobulin, a reaction that requires at least a partially intact follicular structure.8 Such organification increases the effective biologic half-life of the iodine, increasing exposure of thyroid tissue to the irradiation and enhancing radiation-induced cell injury and cell death.8
Those radioactive isotopes of iodine in clinical use (131I, 123I) emit γ rays,9 which can be detected using appropriate detection apparatus (a gamma-camera), allowing imaging of iodine-concentrating tissues and thereby the detection and localization of residual or metastatic thyroid cancer, following stimulation with TSH. This technique of whole-body scanning became the mainstay of postoperative surveillance for thyroid cancer in North America in the 1980s and 1990s, though has become less commonly used in recent years, as improved ultrasound technology, cross-sectional imaging, and measurements of basal or stimulated thyroglobulin have proved more sensitive and more specific.10 Nevertheless, the introduction of single-photon emission computed tomography (SPECT) scanning, in particular, to more accurately localize areas of iodine concentration ensures that isotope imaging continues to play a useful role in the evaluation of patients with known or suspected residual thyroid cancer.11
Although γ rays are high energy, their tissue absorption is low and the majority of these particles do not interact with the cell in which the iodine is concentrated or with surrounding tissue.12,13 Although this is optimal for imaging purposes, providing good image resolution, γ rays are not particularly effective in the treatment of residual thyroid carcinoma, which instead depends on beta-particle emission, the major emitted particle released by the decay of 131I but not 123I.13 The moderately high energy beta particles emitted from 131I have a short median path length in human tissues, traveling, on average, only 0.5 cm before interacting with surrounding tissue.14 The resulting ionization causes DNA damage, including both single- and double-strand DNA breaks.15 This DNA injury is detected by the cell, activating the p53 pathway, which is commonly intact in differentiated thyroid carcinoma cells.16 In the face of minor DNA damage, cellular repair mechanisms are activated, usually restoring the cell to normal health, though with the potential for the induction of additional mutations or chromosomal rearrangements. However, with more extensive DNA damage, activation of p53 triggers apoptosis (programmed cell death) of the affected cell.17,18 Because cancer cells—including thyroid cancer—often lack efficient mechanisms to repair double-strand DNA breaks,19 there is reason to believe that residual thyroid cancer is susceptible to the effects of beta-irradiation, more so than the surrounding normal tissue, though no in vitro, clinical, or outcomes data yet exist to support this hypothesis.
Hypothyroidism stimulates up-regulation of NIS expression in the kidney, increasing iodine reabsorption from the glomerular filtrate20,21 and consequently increasing whole-body iodine retention. Once again this increases the biologic half-life of radioactive iodine, with augmented recirculation and increased opportunity for iodine-concentrating tissues to be exposed to the isotope, but also an increase in whole-body radiation exposure.22 This fact may favor use of recombinant human TSH (rhTSH; Thyrogen) when radioactive iodine (RAI) is being used for either scanning purposes or for ablation of normal thyroid tissue, rather than treatment of thyroid cancer. When treating residual or metastatic cancer, on the other hand, it may actually be preferable to induce hypothyroidism, to maximize whole-body iodine retention, and to maximize the effective radiation dose.
Pretreatment with lithium has a similar impact on iodine half-life, through both a thyroid and possibly a renal mechanism, and has been recommended by some experts as a means to augment the effect of radioactive iodine treatment of thyroid cancer patients.23 The evidence for benefit is scanty, however, and current thyroid cancer treatment guidelines from the American Thyroid Association (ATA), the American Association of Clinical Endocrinologists (AACE), and the National Cancer Cooperative Network (NCCN)24–26 do not recommend this approach.
The question of who should and who should not receive radioactive iodine as adjuvant therapy following surgery for thyroid cancer remains the subject of considerable debate. Unfortunately, that debate is poorly informed by data, almost all of which are limited to anecdotal, small case series and large retrospective reviews, with all of their intrinsic biases.27 Uncertainty remains, also, on the subjects of how best to prepare a patient for such treatment, optimal dosing, use of dosimetry, frequency of administration, and cumulative dose maximums, and few of these topics have high-quality data available to inform the discussion.
Radioiodine Remnant Ablation
Postoperative ablation of the thyroid remnant by radioactive iodine is often used to “complete” the initial surgical treatment in FCDTC.28 Defined as the destruction of residual, normal thyroid tissue following thyroidectomy,”29 there remains controversy about whether this approach is effective in reducing recurrence, whether it is necessary to enable accurate follow-up using measurements of (basal or stimulated) thyroglobulin, and whether those benefits (if they exist) outweigh the risks (if they exist) of the radiation exposure. Consequently, strong disagreements continue to complicate decisions about who should receive radioactive iodine, how best to administer it, and what dose of radioactive iodine is appropriate to use.30 There has often been a lack of clarity in these discussions about the distinction between “radioiodine remnant ablation” (RRA: destruction of normal thyroid tissue) and efforts to treat actual or presumed residual cancer (true adjuvant therapy). Such a distinction is, in my opinion, important, because the risk-benefit ratio of treating a patient with known (or highly likely) residual cancer is quite different from that of using adjuvant therapy in a patient whose cancer has been effectively eliminated by surgery alone. If the potential for benefit of RRA is low, then even a low risk of adverse consequences needs to be considered. If, on the other hand, the cancer represents a real and present danger and if there is some reason to think that radioactive iodine might be helpful, then some significant risks may be worth tolerating in pursuit of effective cancer treatment. “The punishment should fit the crime.”31
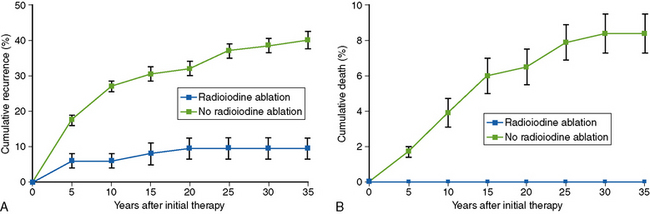
The initial suggestion that RRA could improve outcomes in FCDTC arose from the seminal studies of Mazzaferri and colleagues, whose pioneering work was based, like almost all the studies that underpin our current practice in thyroid oncology, on the analysis of a large retrospective cohort of patients who had undergone surgery for thyroid cancer either in the U.S. Air Force or at Ohio State University.32,33 In his early analyses, the outcome of patients treated by the combination of total thyroidectomy, radioactive iodine, and thyroid hormone suppressive therapy was clearly superior—both in terms of mortality and recurrence risk—than that of patients who were not so treated.33 In these studies, the 30-year recurrence rates, in patients with tumors larger than 1.5 cm, were halved (from 38% to 16%) in 350 patients who received ablation with 131I, compared to 802 who did not receive that treatment (Figure 51-1, A).34 Similarly, cause-specific 30-year mortality rates fell from 8% to 3%, with no deaths observed in patients who received 131I in whom there was no evidence of residual disease following surgery (Figure 51-1, B). These findings, published from the late 1970s onward,32–41 proved immensely influential, changing how patients with thyroid cancer were treated, monitored, and followed and making RRA a de facto “standard of care,” strongly supported by several other groups.42–45
However, the studies on which the Mazzaferri publications were based included patients with a variety of pathologies (follicular, Hurthle cell, and papillary thyroid cancer), who had undergone radically different surgeries: lobectomy in some, subtotal thyroidectomy in others, total thyroidectomy in many. The more aggressive (and potentially more likely “curative”) surgeries were most likely to be performed by the expert surgeons at Ohio State University, particularly late in the series, when radioactive iodine was also increasingly being used. The studies also included patients with extensive disease, including metastatic disease, as well as much lower-risk patients; the original report, from 1977, included patients with gross residual disease and metastatic disease, with true RRA being performed in only 33 patients.33 Because of these confounding variables, the true benefit of RRA as adjuvant therapy, following adequate removal of the primary tumor, resection of nodal disease, and virtually complete extirpation of the thyroid gland, cannot be fully assessed from these data. Nevertheless, the data were sufficiently compelling that there followed a fundamental shift in the management of thyroid cancer across the country, leading to a new de facto standard of care, including pressure on surgeons to undertake a “total” thyroidectomy—to facilitate the use of RRA—and on endocrinologists to use the effective treatment to effect a “cure” for their patients.
Hay and colleagues struck a contrary note throughout the rise of the RRA approach during the 1980s and 1990s. Along with other authorities, Hay argued that the outcome for the majority of patients with papillary thyroid cancer (PTC) is excellent following adequate surgery; that RRA does not improve outcome in otherwise low-risk patients (defined by a variety of staging and prognostic scoring systems); and that exposure to radioactive iodine in these patients incurs for them unnecessary expense, inconvenience, and risk of side effects in the unjustified and inappropriate pursuit of a “clean slate” on iodine scanning, an undetectable basal thyroglobulin and an almost undetectable stimulated Tg.46–55
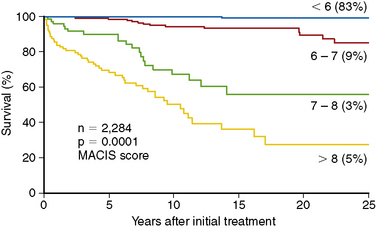
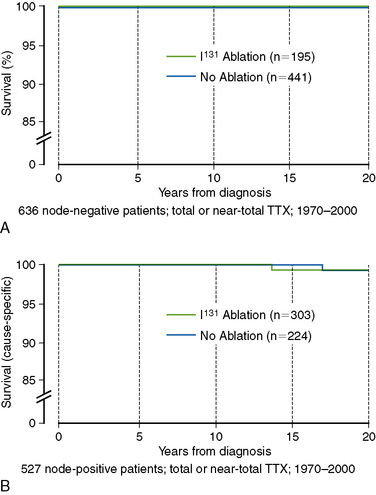
The most fundamental distinction between the original Mazzaferri studies and the data reported by Hay et al. was Hay’s focus specifically on patients at low risk of mortality. Based on his previously published “MACIS” scoring system (metastases, age, completeness of resection, invasiveness, and size), over 80% of the Mayo Clinic’s PTC patients could be classified as “low-risk,” with MACIS scores of < 6, carrying a 25-year mortality risk of < 0.5% (Figure 51-2).56 Most of these patients are young (< 45 years), with locoregional disease, but this category also includes patients over 45 years of age with intrathyroidal (noninvasive) disease, completely resected at surgery. Because nodal status is not a factor in calculating the MACIS score, many of these patients are node positive, which carries an increased risk of recurrence but not of mortality in most,43,56–63 though not all, studies.64–66 Restricting the analysis to patients with PTC (excluding other histologies), at low risk of mortality (the majority), who have undergone a similar extent of surgery (near-total or total thyroidectomy, with targeted neck dissection based on preoperative ultrasound), who do not have distant metastatic spread or gross residual disease (both indications for radioactive iodine treatment, rather than RRA), and matched for nodal status allows the impact of RRA to be examined as accurately as is possible in a retrospective study. Figure 51-3, A, shows the mortality impact of RRA on these low-risk, node-negative patients with PTC; clearly there is no detectable improvement in the already zero risk of mortality in this group. Mortality for the node-positive patients (Figure 51-3, B) is similarly low and is not improved by RRA. Based on these data, the use of RRA cannot be justified on the grounds of improved mortality in low-risk patients with PTC.49
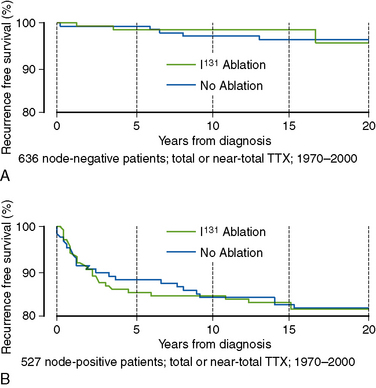
Recurrence rates in low-risk PTC are determined primarily by nodal status, with overall recurrence rates of < 5% in initially node-negative patients, rising to 15% to 20% in node-positive patients.49 Almost all such recurrences are detected in cervical lymph nodes. In the Mayo Clinic series, those recurrence rates are not impacted by RRA in either the node-negative (Figure 51-4, A) or node-positive (Figure 51-4, B) but otherwise low-risk PTC patients. Consequently, these studies have failed to show any detectable benefit of “routine” 131I ablation in low-risk PTC patients following adequate primary surgery, including selective node dissection, when no evidence of residual disease exists postoperatively.55
The recurrence rates reported in the Mayo Clinic series48 are considerably lower than those reported in some other studies, even in the absence of routine 131I ablation. This almost certainly reflects the completeness of surgical excision of the primary tumor, achieved in this highly specialized surgical center, and emphasizes the importance of the primary surgery in determining outcome. It has been argued that these data are unique for that sole reason: excellent surgeons operating in a highly specialized center can achieve better outcomes, whereas everyone else needs RRA to achieve the same outcome.
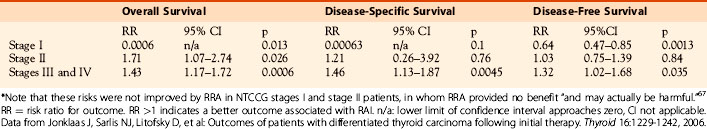
However, in 2007, the National Thyroid Cancer Cooperative Group (NTCCG) published an analysis of the impact of RRA in its large, multicenter, prospectively collected data set encompassing ~ 26,000 patients, followed for an average of 5 years, at multiple centers across North America.67 As reproduced in Table 51-1, the relative risks for overall mortality for cause-specific mortality and for recurrence-free survival were not improved by RRA in the stages I and stage II patients. Details of their unique staging system are shown in Table 51-2: it is worth noting that stages I and II include patients with tumors up to 4 cm, including both those with local extrathyroidal invasion and those with cervical nodal involvement, though without distant metastatic spread, overlapping significantly with Hay’s low-risk group. Demonstrating no evidence of benefit from 131I remnant ablation, Jonklaas et al. concluded that remnant ablation provided no benefit “and may actually be harmful” to these low-risk patients.67 In contrast, their patients with more advanced and therefore higher-risk disease (NTCCG stages III and IV) did see improved outcomes (overall survival, cause-specific survival, and disease-free recurrence) following RRA, suggesting that patients with residual malignancy after primary surgery or those with a high risk of such residual cancer do indeed benefit from adjuvant therapy with 131I. Support for the selective use of RRA also comes more recently from the Sloan-Kettering group who have analyzed outcomes in low-risk PTC patients and have found excellent outcomes following adequate initial surgery without any requirement for RRA.68
Table 51-1 Relative Risks for Overall Mortality for Cause-Specific Mortality and for Recurrence-Free Survival*
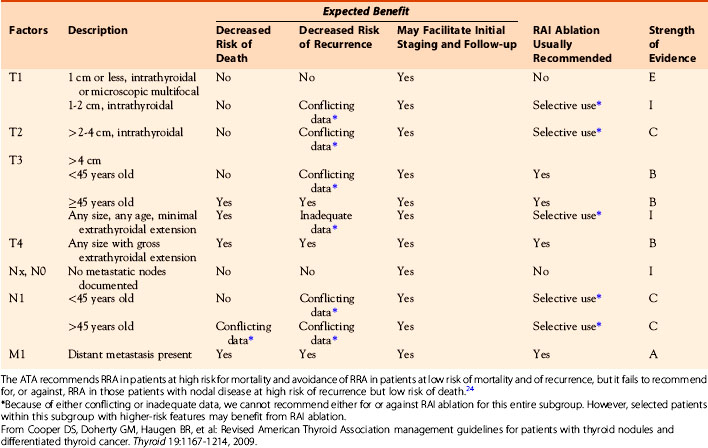
The American Thyroid Association cancer management guidelines, first published in 2006, supported the selective use of 131I ablation in low-risk patients with PTC,69 although the association still endorsed its near-routine use in patients with larger tumors, nodal involvement, or extrathyroidal spread. Following the publication of the Jonklaas paper in 2007, however, both the revised ATA 2009 Guidelines24 and the NCCN Guidelines26 have taken a more complex and more nuanced approach to patient selection for RRA. The ATA, in particular, has provided guidance (reproduced in Table 51-3) that recommends the use of I-131 ablation in patients at high risk for mortality (strong recommendation), and avoidance of 131I in patients at low risk of mortality and of recurrence (strong recommendation), but it fails to recommend for, or against, 131I therapy in those patients with nodal disease at high risk of recurrence but low risk of death.24 This lack of guidance reflects the reality that the available data on outcomes after 131I therapy largely fail to address the risk-benefit ratio of 131I ablation in patients divided into these prognostic strata. However, the Hay, Jonklaas, and Tuttle/Memorial data all argue that RRA is ineffective in lowering the risk of recurrent disease or death in low-risk patients, defined either by the MACIS scoring system,55 by AJCC stage,27,70 or by the NTCCG staging system.67 Because these groups encompass the majority of patients with thyroid cancer, it seems appropriate for RRA to be restricted to those patients in whom there exist either evidence for, or a significant likelihood of, residual disease following adequate primary surgery. In these individuals, radioactive iodine should be regarded as adjuvant therapy for the treatment of residual disease, rather than being used solely for ablation of a normal thyroid remnant.
Table 51-3 Guidelines from the American Thyroid Association Concerning the Use of 131I Remnant Ablation
Despite those ATA recommendations, Hay’s data, which have proved so controversial since he first presented them,54 remain a source of concern for endocrinologists even today. The differences between Mazzaferri’s data demonstrating the efficacy of RRA and Hay’s data showing the lack of efficacy have been the fodder for many heated debates, and those debates continue. Realistically, however, the differences between these groups of studies lie in the selection of patients and the adequacy of their initial surgical therapy. Those patients whose disease has been adequately treated by surgery do not benefit from RRA, whereas those with residual disease may benefit from effective adjuvant therapy with radioactive iodine. Ablation of the normal thyroid remnant does not change outcomes, whereas radioactive iodine treatment of residual thyroid cancer may. The key is to identify those patients with residual cancer so that they can be effectively treated, while avoiding unnecessary therapy in the majority of patients whose cancer has already been adequately treated by surgery alone.
RRA as a Requirement for Postoperative Surveillance in Thyroid Cancer
Elimination of the postsurgical remnant may carry an additional advantage by simplifying the interpretation of postoperative serum thyroglobulin (Tg) measurements. In most cases, a “total” thyroidectomy does leave some small remnant of thyroid tissue—in the pyramidal lobe/thyroglossal duct tract, in the region of the parathyroid glands, or the tubercle of Zuckerkandl and ligament of Berry regions—which some surgeons prefer to leave in situ rather than risk injury to the recurrent laryngeal nerve in that area.71 That residual normal thyroid tissue will contribute, to some degree, to the production of Tg, particularly in the stimulated state (i.e., with elevated TSH following thyroid hormone withdrawal or Thyrogen stimulation)72 and may easily be misinterpreted as an indication of residual malignancy rather than residual normal thyroid tissue. That is particularly the case for thyroglossal duct remnants, which also may be misinterpreted as indicating nodal disease on isotopic imaging, because of its location outside the thyroid bed.73
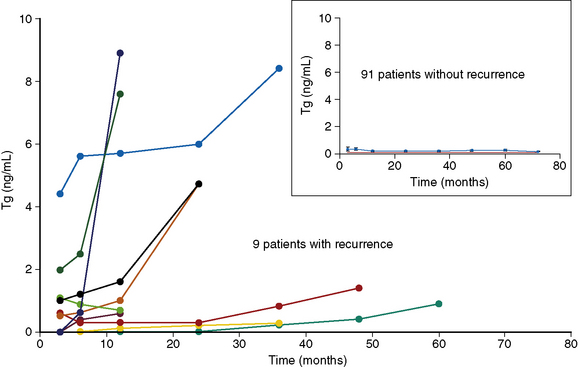
Even in the absence of significant residual thyroid tissue, it is an oft-repeated truism that measurements of basal (far less, stimulated) Tg cannot be interpreted unless the normal thyroid remnant is completely eliminated using RRA after a “total” thyroidectomy. The data that support this statement are scanty, however and are not based on data related to patient outcomes. We (Plaskowski et al., unpublished data) have reviewed the use of basal Tg to follow low-risk (MACIS < 6) patients with PTC following total thyroidectomy, without RRA. Of the 100 patients so far assessed in this series, 58 were node positive and 5 exhibited extrathyroidal extension, though complete surgical resection was achieved in all cases (negative surgical margins) and none of these patients showed evidence of distant metastatic spread. Patients with detectable Tg antibodies were excluded from the study for the purpose of this analysis. Following thyroidectomy alone, the mean basal Tg was 0.3 ng/mL, with 70% of patients exhibiting undetectable Tg (< 0.1 ng/mL) while taking thyroid hormone in minimally suppressive doses. During a median follow-up of 7 years (range 5 to 10 years), 9 patients developed recurrent disease, detected through a combination of rising basal Tg and high-resolution neck ultrasound scan. In all 9 cases, the unstimulated Tg was either elevated from the outset or rose progressively during follow-up (Figure 51-5). No patient in this series developed a recurrence without a rising basal Tg, and all 9 patients whose Tg rose progressively had residual disease eventually confirmed on ultrasound. These data confirm that the low-risk majority of PTC patients can safely be managed by removal of the primary tumor, a total thyroidectomy, and resection of involved lymph nodes, but without the need for RRA, either to alter outcomes or for the purpose of postoperative surveillance, a conclusion with which others are beginning to agree.68,70
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree