Figs. 13.1 and 13.2
(a, b) Two examples of the geriatric nose, profile and frontal views. Note the skin changes and external structural effects of aging
Anatomic changes of the nose are, in part, related to changes of the cartilaginous structures as well as weakening of soft tissue attachments. The biochemical composition and mechanical properties of cartilage change with age, resulting in a tendency to collapse. For example, there is a decrease in the glycosaminoglycan content of nasal septal cartilage with increasing age, resulting in stiffening with decreased fluid flow through the tissue. Additionally, there appears to be a slight increase in hydroxyproline content with increasing age. These changes are similar to those seen in articular cartilage during the aging process. Although the physical stresses on articular cartilage are much different than that of septal cartilage, the similarity of their biochemical changes may represent systemic effects on cartilage with age (Rotter et al. 2002).
There are two practical considerations of these data for clinical practice. First, anatomic changes are likely to affect airflow through the nose in older patients, resulting in restrictions that may cause dryness, irritation, and obstruction. These problems may exacerbate existing conditions and contribute in a large way to patient symptoms. Secondly, older adults may seek septorhinoplasty to alleviate such functional problems and also for cosmetic desires. Careful consideration must be given to these patients as they may have underlying significant medical comorbidities, such as hypertension, coronary artery disease, or diabetes, placing them at higher risk for elective surgery. Cochran et al. noted that many older patients at the time of rhinoplasty had ossified septal cartilage, making it more frequently necessary for auricular or costal cartilage to be harvested (Cochran et al. 2007), potentially lengthening operative time. It would be prudent to have medical and anesthesia clearance prior to proceeding with elective surgery in any older patient and several guidelines exist to make this determination (Stefan et al. 2011; Palmer 2009). Nevertheless, nasal surgery in older subjects can be safe and effective when approached carefully (Ban et al. 2010). As with all rhinologic patients, psychological motivation for the procedure needs to be assessed. Geriatric patients frequently have to deal with significant life issues, including death of loved ones, lack of social support, financial challenges, and struggles with quality of life. One must assess whether older patients have taken ample time to consider any cosmetic surgery prior to proceeding. Most importantly, assessment of the functional consequences of rhinoplastic procedures on this patient subset is critical to maintain airflow.
13.3 Physiological Changes with Age
New research is helping to elucidate some of the subtle changes in the physiology of the nose over time that may lead to symptoms and/or disease in older adults. Although we are far from fully understanding comprehensively how the nose changes over time, further study of the known physiological changes will help us to provide better medical and surgical treatment regimens for these patients.
13.3.1 Alterations in Nasal Function
A common result of aging is nasal dryness, a frequent complaint in older patients. This condition may present with crusting, irritation, epistaxis, or obstruction. Although this is not well studied, etiologies include mucosal and glandular atrophy, vascular changes which reduce nasal humidification, and medication use (e.g., antihypertensives, which affect vascular regulation in the nose, or first-generation antihistamines, which inhibit cholinergic responses in the nose). Structural changes in the nose may also contribute by causing turbulent airflow which dries the mucosa. The mucosa itself is altered. Schrödter performed biopsies of the middle turbinate in 40 subjects of varying ages and found significant atrophy of the epithelium in the older subjects (Fig. 13.3) (Schrödter et al. 2003). This analysis found thin epithelium and also increased thickness of the basement membrane. Also, the percentage of normal ciliated respiratory epithelium declined in the older subjects. Similar studies using electron microscopy corroborate this finding (Toppozada 1988). Increased expression of caspase 3, an apoptotic marker, indicates that part of the defect may be in the ability of the epithelium to renew itself, and there are other functional aspects of the epithelium (toxin neutralization) that may be affected (Getchell et al. 1993). This matches what we see clinically, a thinner, atrophic epithelium in older subjects when examined with nasal endoscopy.
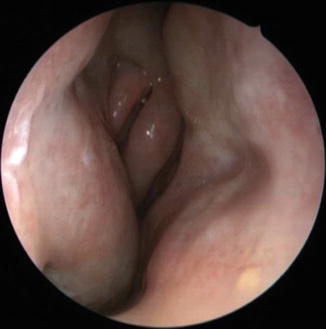
Fig. 13.3
Endoscopic view of geriatric patient with atrophy of endonasal mucosa
Another major alteration affecting this symptom is in one of the main functions of the nose: the ability to warm and humidify air (Naclerio et al. 2007). The two major mechanisms leading to the alteration in nasal conditioning capacity (NCC) are changes in the nasal mucosal temperature (NMT) and the volume of the nasal cavity. During inspiration of air, water evaporates from the nasal mucosa to condition inspired air, leading to heat loss and, consequently, a decrease in NMT (Cole 1954). Lindemann found that using in vivo air temperature and humidity measurements at the nasal valve and the region just anterior to the head of the middle turbinate, both temperature and humidity values were significantly lower in older subjects (mean age 70 years old) compared to younger subjects (median age 27 years old) (Lindemann et al. 2008). Additionally, nasal volumes appeared to be larger in the older age group. Increased turbulence of airflow may be one result causing the sensation of nasal obstruction despite larger space (paradoxical nasal blockage). Endonasal geometries are indeed enlarged in older subjects (Kalmovich et al. 2005). This suggests that NCC in older subjects is compromised. Thus, lower ability to warm and humidify air may be present in older subjects, potentially affecting lower airway function. This may also contribute to the increased dryness in this population.
We may take some analogy from changes in the oral mucosa. As with the nose, the lubrication of the oral cavity changes with age. Although there should not be a decrease in saliva production with age, unless altered by medications or systemic conditions, there are changes in the consistency of the saliva, including a decrease in mucin concentration and decreased secretion of protective IgA antibodies (Ritchie 2002). This may lead to increased caries, periodontal disease, and poorer nutritional status.
A second area of altered nasal physiology with age is in mucociliary clearance. Kim noted decreased ciliary beat frequency in vitro nasal epithelial cells taken from patients older than 60 years of age (Kim et al. 2007). Ho et al. also found decreased nasal mucociliary clearance by the crude saccharin clearance test and the more sensitive measure of ciliary beat frequency by a photometric test (Ho et al. 2001). However, there is likely significant interindividual variability in this phenomenon since Sakakura et al. showed that 70 % of patients older than age 60 retained saccharin transport times comparable to their younger counterparts (Sakakura et al. 1983). Diabetes and hypertension, both increased in prevalence in older subjects, are also associated with decreased ciliary function (de Oliveira-Maul et al. 2012). In summary, if the cilia are not moving as rapidly, this can lead to symptoms of mucus buildup, rhinitis, inflammation, or infection, from persistence of organisms and molecules trapped in the mucus layer (Fig. 13.4). Hence, both the structural components and the function of the epithelial lining of the nose in older patients demonstrate significant changes with age that may affect airflow, mucus quality and production, and mucociliary clearance.
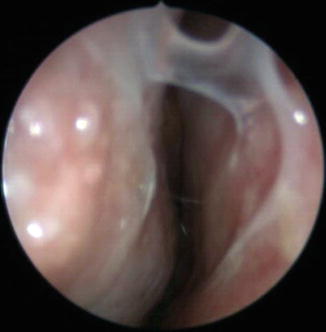

Fig. 13.4
Endoscopic view of geriatric patient with thickened nasal mucus, a common complaint in this patient population
A third way nasal function is altered with age is regulation of vascular responses in the nose. Nervous control of vascular tone in the nose is important in the regulation of the critical functions of the nose, to warm and humidify air, and also may affect diseases involving vasodilation such as allergic and nonallergic rhinitis. Tillmann found that vascular regulatory responses are reduced with age (Tillmann et al. 2009). Fifty-two subjects were acclimated to the laboratory environment for 5 min and baseline measures of perfusion by optical rhinometry were performed. The subjects were then moved to supine position for 30 min, which should alter blood flow. Older subjects had more rapidly increased perfusion, but did not return to baseline in contrast to younger subjects. This study suggests that autonomic control of nasal blood flow is altered in older subjects, potentially affecting humidification (as described above) and the other functions of the nose discussed above. Using liquid crystal thermography exhalation monitoring to measure nasal cycle, Doty found decreased regulation of the nasal cycle in older adults (Mirza et al. 1997). Overall, the proportion of subjects exhibiting the alternating rhythmicity associated with the classic nasal cycle decreased with age. No association was present between nasal cycle parameters and scores on the mini-mental state examination (MMSE). The results suggest that the classic nasal cycle may be a marker for age-related central nervous system changes. These phenomena may also be important in nonallergic rhinitis.
13.3.2 Olfaction and Nasal Sensitivity
Olfaction, mediated by the olfactory nerve, and sensitivity of the nose, controlled by the somatosensory system and mediated by the trigeminal nerve, both decrease with age. In 1994, the National Health Survey determined that 3.2 million people, or 1.65 % of adults in the United States, had chronic chemosensory problems involving smell and/or taste; 40 % of these adults were aged 65 years and older, with an exponential increase seen with increasing age (Hoffman et al. 1998). Adults over 65 years of age have approximately half the intensity of smell and irritant sensation as subjects between the ages of 18 and 25 (Stevens et al. 1982). In another study, the sensitivity of the nose was assessed by providing tactile stimulation with monofilaments of progressively larger sizes. Adults 50–65 years of age required significantly less stimulation to detect sensation of the monofilament as compared to adults aged 66 and above (Salzano et al. 2010).
In order to understand how the olfactory system is altered with age, we first need a better understanding of the normal olfactory system. The olfactory mucosa is distributed along the upper portion of the nasal septum, below the cribriform plate, and along the medial aspect of the superior turbinate, as well as along the roof of the middle turbinate (Paik et al. 1992). On histologic examination, there is a patchy distribution of olfactory epithelium, which becomes increasingly sparse with aging because olfactory epithelium becomes replaced with respiratory epithelium over time. Robinson et al. demonstrated in a murine model that there is an increase in apoptotic gene expression of olfactory neuron receptors with increasing age, likely resulting in increased cell death (Robinson et al. 2002). Over time, even small toxin exposures, including heavy metals, such as manganese, cigarette smoke, or volatile chemicals, may cause harm to the olfactory epithelium (Lafreniere and Mann 2009). In mice it has also been seen that there is decreased regeneration of the neuroepithelium of the olfactory bulb with increasing age (Suzukawa et al. 2011). Because olfactory stem cells regenerate with time, any alteration of this turnover may also contribute to age-related olfactory loss, either from insult or from inherent aging. All of these may contribute to a decrease in function of the olfactory system as one ages.
Age-related olfactory loss (presbyosmia) is an important public health problem worldwide (Hoffman et al. 1998; Wysocki and Gilbert 1989; Brämerson et al. 2004; Landis et al. 2004). In the USA, olfactory complaints from the approximately 14 million individuals over age 55 who are affected lead to over 200,000 physician visits annually (NIDCD 2010; NAMCS 1979). This sensory impairment of aging affects critical functions such as nutrition (Mattes and Cowart 1994; Mattes et al. 1990; Miwa et al. 2001), sensation of pleasure (Wolfe et al. 2008), detection of environmental hazards (Santos et al. 2004), mood, cognition, behavior (Herz and Schooler 2002; Schiffman et al. 1995a, b), sexuality (Bhutta 2007; Jacob et al. 2002), and general well-being (Neuland et al. 2011), and therefore, it poses a profound burden on older adults. Indeed, up to one-third of older subjects report dissatisfaction with their ability to smell (Wysocki and Pelchat 1993), and ~50 % are unable to detect the standard warning odor in natural gas (Cain and Stevens 1989). Importantly, decline in olfaction has been linked to several neurodegenerative conditions, mentioned above (Hawkes 2006; Handley et al. 2006; Kovacs 2004; Schubert et al. 2008; Wilson et al. 2006, 2007a, b, 2009). If this link involves shared genetic or environmental risk factors, then understanding the pathogenesis of presbyosmia may have broad implications for a wide array of problems, especially regarding other sensory impairments of aging (Li and Lindenberger 2002). Thus, olfactory sensory loss is related to factors that are critical to the physical well-being, social function, and quality of life of older adults. Because olfaction declines over time, the clinical impact will increase as our population ages.
Acquired chemosensory complaints are also important in Otolaryngology. In addition to traumatic injury or post-viral olfactory loss, cancer chemotherapy can affect olfaction. For example, patients undergoing palliative chemotherapy treatment for cancer frequently had complaints of dysgeusia and sensitivity to odors while undergoing treatment. Those patients with severe complaints were found to have lower calorie intake, increased weight loss, and lower quality of life scores, than in patients with less severe complaints (Hutton et al. 2007). Because the olfactory epithelium regenerates, one cause of this could be a decrease in stem cells with age in the olfactory mucosa, either inherently or after stimulation of regeneration by injury. Indeed, telomere shortening has been observed in the regenerative response to chemical injury of the olfactory epithelium in a mouse model, providing evidence that telomere shortening impairs the regenerative capacity of this tissue (Watabe-Rudolph et al. 2011). So, aging may lessen the ability of the olfactory mucosa to survive respiratory insult by toxins.
There are many systemic causes of chemosensory dysfunction. Olfactory deficits are seen in neurologic disorders, such as Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, and Korsakoff’s psychosis, all of which generally affect the elderly. It is important to counsel patients with hyposmia or anosmia, as they may be unaware of noxious materials in their surroundings. They should obtain natural gas detectors if they have natural gas stoves, oven, or heat at home as they may not be able to detect a fuel leak. Additionally, they should be aware of dates on food products, label leftovers, and dispose of foods reaching their expiration dates to avoid food spoilage. Lastly, smoke detectors should be checked regularly for function and efforts must be maintained to promote nutrition.
13.3.3 Immunosenescence
Immunosenescence is the term used to describe the decreased function of the immune system with age. Both the adaptive and innate immune systems are impaired (Wang et al. 2010; Shaw et al. 2011). Decreased immune response is believed to contribute to increased infections, autoimmune disease, and cancers in the elderly population.
There are several effects of immunosenescence in the nose. IgA is an immunoglobulin that is secreted along the respiratory and gastrointestinal tracts, where it helps to neutralize a variety of pathogens. Alford demonstrated that IgA levels can be measured in nasal secretions and comprise approximately 38 % of nasal wash proteins in normal patients. He also showed that IgA levels in nasal secretions decrease significantly with age (Alford 1968). In a murine model, it has been shown that mucosal immunity wanes prior to systemic immunity in studies where vaccination to cholera toxin was performed by mucosal and subcutaneous methods, respectively, in different ages of mice (Koga et al. 2000).
IgE levels also decrease with age. Mediaty and Neuber studied 559 individuals with atopic dermatitis, allergic rhinitis or asthma, and insect allergy. In all patients, except those with atopic dermatitis or those with high total serum IgE >300 kU/l, total and specific serum IgE levels were significantly decreased in patients aged greater than 60 compared to their younger counterparts. They hypothesized that there may be more robust mechanisms in atopic dermatitis or conditions resulting in high serum IgE that lead these patients to have a more persistent response. Additionally, the type of atopic disease and the age of disease onset may impact IgE levels (Mediaty and Neuber 2005).
Immunosenescence is poorly studied as it affects the upper airway. However, we can extrapolate findings from the lower airway (Busse and Mathur 2010). In that location, there are several immune alterations that might facilitate persistence of asthma, a related airway disease to rhinitis. These include changes in airway neutrophil, eosinophil, and mast cell numbers and function as well as altered antigen presentation, decreased specific antibody responses, and altered cytokine profiles. The sum of these changes might affect susceptibility to upper respiratory tract infections.
In summary, this is a complex area in which there is little data on the nose specifically, but age-related changes in immune function are likely to impact nasal physiology and disease in older patients.
13.4 Geriatric Rhinitis
13.4.1 Overview
Rhinitis is a pervasive complaint in physicians’ offices. It is estimated that 20–40 % of subjects living in Western countries are affected by rhinitis. Rhinitis may be allergic or nonallergic in nature, with approximately 50 % of rhinitis patients belonging to each group (Fokkens 2002). These forms of rhinitis affect older subjects as well, though because allergy wanes with age, nonallergic rhinitis tends to predominate. There are many potential causes of rhinitis in the geriatric population. Edelstein noted that six nasal complaints were more prominent in older adults: nasal drainage, postnasal drip, sneezing, coughing, olfactory loss, and gustatory rhinitis (Edelstein 1996). Regarding quality of life in an Italian study, geriatric patients with rhinitis underwent clinical evaluation and responded to the Rhinasthma questionnaire (Ventura et al. 2012). All patients also underwent skin prick testing, measurement of total IgE level, and nasal cytologic analysis. In the older patients the epithelial to goblet cell ratio was decreased. The quality of life in older people was more impaired than in young adults. These authors concluded that quality of life is more heavily impaired compared with young adults. However, one study showed that nasal-specific quality of life measures show no deterioration in older subjects and do not therefore correlate with changes in nasal function (Lindemann et al. 2010). Further investigation of this topic is warranted to elucidate this paradox.
13.4.2 Allergic Rhinitis
Allergy is defined as an immediate type IgE-mediated response to an allergen exposure, where IgE is bound to mast cells and its attachment to an allergen results in degranulation of the mast cell with histamine release. The diagnosis of allergy can be confirmed by skin, in vitro, or provocation testing. The Allergic Rhinitis and its Impact on Asthma (ARIA) consensus categorizes allergic rhinitis as either intermittent allergic rhinitis (IAR) or persistent allergic rhinitis (PAR). IAR denotes that symptoms are present less than 4 days per week or less than 4 consecutive weeks per year. PAR indicates that symptoms are present greater than 4 days per week and more than 4 consecutive weeks. These can be further categorized as mild or moderate/severe based on impact of quality of life (Bousquet et al. 2008).
Although allergy is well studied, the majority of studies performed to date involve either the pediatric population or young adults. In older adults, the incidence of allergic rhinitis actually decreases with age along with atopy as detected by skin prick tests. Karabulut et al. noted that only 50 % of older adults have positive skin prick tests as compared to 70 % of younger adults when matched for allergic symptoms including nasal, eye, pulmonary, and dermatologic symptoms (Karabulut et al. 2011). Subjects from French cohort aged 65 years and over (n = 352) found that respiratory allergy is present in older people and that there is an association between smoking and IgE level independent of allergic reactivity to common allergens in the elderly, highlighting the importance of environmental factors in rhinitis (Raherison et al. 2004). A recent longitudinal study analyzed allergic sensitivity 15 years after primary testing. Skin prick test (SPT) and evaluation of serum total and specific IgE and nasal eosinophils were conducted in 108 subjects from Palermo, Sicily, in Italy. In general, rhinitis symptoms tended to be milder at follow-up. All parameters examined decreased with time. However, the changes in rhinitis symptoms appear to be related to changes in the nasal eosinophils, independently of SPT and specific IgE (Di Lorenzo et al. 2013). Thus, in general allergic disease and markers decline with age.
One practical consideration for these patients is that because elderly patients frequently have atrophic or photodamaged integument, skin testing for allergy may actually be less reliable than in younger adults. There is evidence that there are decreased numbers of mast cells in atrophic skin, thereby making the possibility of a false-negative result higher. In older subjects, an area of skin that is protected from the sun can be considered for skin testing, a histamine control used so that the most dramatic obtainable response can be visualized, and consideration given to in vitro testing if no reliable area of skin is present (King and Lockey 2003). Both skin and in vitro testing must always correlate with patient history for accurate diagnosis. Allergic disease in the older patient population has been reviewed recently (Cardona et al. 2011).
13.4.3 Nonallergic Rhinitis
There are many causes of rhinitis which are not allergic in nature, but can be equally bothersome to patients and have a marked impact on quality of life. The data regarding this phenomenon are less developed, as compared to those for allergic rhinitis. Studies of patients presenting to allergists have demonstrated that 23–52 % of patients with rhinitis have the nonallergic form (Schroer and Pien 2012). Patients can also be sensitized to allergens at any time, thereby changing the categorization of their rhinitis. No good diagnostic techniques are available to distinguish between the subtypes of nonallergic rhinitis, but a diagnosis is based on history and symptoms and negative allergy testing. Additionally, there is no set classification system for nonallergic rhinitis. Further confusing the matter is allergic and nonallergic rhinitis can coexist (“mixed rhinitis”). Interestingly, it has been observed that 70 % of patients with nonallergic rhinitis present during adulthood (age > 20), whereas 70 % of patients with allergic rhinitis initially present during childhood (age < 20). There is also a greater propensity for females to be affected, compared to males (Togias 1990). Recent study of nonallergic rhinitis suggests that local IgE production may be involved. The only approved treatment for nonallergic rhinitis currently is intranasal antihistamines which are effective in some cases. Nonallergic rhinitis, therefore, is a major disorder of older adults. Due to its complex nature, however, our understanding of its pathophysiology in general and specifically in older adults is limited, resulting in limited ability to treat this problem effectively. A trial and error method of different nasal medications is usually pursued.
13.4.4 Vasomotor Rhinitis
Vasomotor rhinitis is one form of nonallergic rhinitis and is often viewed as an idiopathic diagnosis that is considered when a patient has no evidence of allergy, infection, eosinophilia, hormonal changes, or drug exposure (Lal and Corey 2004). Symptoms include nasal congestion, nasal drainage or postnasal drip, and perennial symptoms; however, pruritus is rare (Shah and McGrath 2012). There is also typically no cytological evidence of nasal mucosal inflammation (Settipane and Charnock 2007). Vasomotor rhinitis is a poorly understood condition but believed that the primary cause is autonomic nervous system dysfunction with either a diminished sympathetic drive or an elevated parasympathetic drive causing increased nasal congestion and resistance.
13.4.5 Drug-Induced Rhinitis
Many medications can have the adverse effect of causing rhinitis. This can especially affect the geriatric population, as they are often on numerous medications to control other comorbidities. It is important to consider polypharmacy as a cause of the patient’s symptoms and see if removing medications, rather than adding more of them, may help. Aspirin, or other nonsteroidal anti-inflammatory drugs (NSAIDs), which are commonly taken by older adults, may cause acute inflammation in the nose in susceptible patients via the inhibition of COX-1. The breakdown of arachidonic acid to the lipoxygenase pathway is favored causing decreased prostaglandin E2 and an increase in cysteinyl leukotrienes, which include LTC4 which is thought to be a lead contributor to aspirin-exacerbated asthma (Varghese et al. 2010). Many other medications can cause rhinitis in older patients. There are also neurogenic-type medications, such as alpha- or beta-adrenergic antagonists, which work by decreasing sympathetic tone. This causes primarily congestion but also rhinorrhea. Medications such as clonidine and methyldopa fall within this category of medication. Phosphodiesterase-5 inhibitors, which is used for erectile dysfunction, may reportedly impact the erectile tissues of the nose and have been associated with nasal stuffiness and epistaxis (Hicklin et al. 2002). Certain antihypertensives, psychotropic agents, and hormonal treatments, including estrogen therapy, can also cause rhinitis. Their mechanisms of action have not been fully elucidated. Lastly, rhinitis medicamentosa can affect adults who overuse nasal decongestants, causing rebound nasal congestion.
When medication use is thought to be the cause of rhinitis, a pharmacist or geriatrician may be useful in giving recommendations about changing the medication. Polypharmacy is frequently an issue in the geriatric population, and these patients need close monitoring for drug interactions, as well as adverse side effects.
13.4.6 Atrophic Rhinitis
Atrophy related to structural changes of the nasal structure and intranasal function can cause atrophic rhinitis which frequently affects the elderly with a mean age of occurrence of 52–56 years (Settipane 2011). Symptoms include dryness and crusting of the nasal mucosa, with possible underlying bony resorption (Liston and Siegel 1981). Anatomic and physiological changes may cause atrophic rhinitis, including structural changes of the mucosal glands, vasculature, and connective tissue of the nose as well as cholinergic hyperactivity. Patients with atrophic rhinitis complain of drying of the nose, nasal congestion, and rhinitis and sometimes fetor is noted. Thick nasal secretions increase worsening postnasal drip symptoms and lead to frequent clearing of the throat. Treatment of this condition is difficult as it is impossible to reverse or mitigate the physiological changes. Intranasal steroids, expectorant drugs, and nasal saline sprays are recommended though their efficacy in this group is understudied (Wallace et al. 2008). Aggressive use of nasal humidification, emollients, and saline irrigation is helpful.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


