Purpose
To compare visual, refractive, topographic, and corneal higher-order aberration outcome at the 2-year follow-up after customized photorefractive keratectomy (PRK) followed by cross-linking (CXL) as a single procedure versus CXL alone in eyes with progressive keratoconus.
Design
Prospective, interventional, nonrandomized clinical trial.
Methods
Seventeen patients (34 eyes) with progressive keratoconus were assigned to 2 groups: the worse eye (17 eyes) was assigned to the PRK plus CXL group and the better eye (17 fellow eyes) was assigned to the CXL group.
Results
In the PRK plus CXL group, uncorrected distance acuity improved significantly, from a mean ± standard deviation of 0.63 ± 0.36 logarithm of the minimal angle of resolution (logMAR) units to 0.19 ± 0.17 logMAR units ( P < .05) and best distance acuity from 0.06 ± 0.08 logMAR to 0.03 ± 0.06 logMAR ( P < .05). Manifest refraction spherical equivalent and spherical and cylindrical power improved significantly ( P < .05). Simulated keratometry, flattest, steepest, average, cylindrical, apex keratometry, and inferior–superior value decreased significantly ( P < .05). Total and coma-like aberrations significantly decreased for all pupil diameters ( P < .05). In the CXL group, uncorrected distance acuity improved, but not significantly, from 0.59 ± 0.29 logMAR units to 0.52 ± 0.29 logMAR units, and best distance acuity improved from 0.06 ± 0.11 logMAR units to 0.04 ± 0.07 logMAR units ( P > .05). Manifest refraction spherical equivalent and cylindrical power improvement was not significant ( P > .05), unlike spherical power ( P < .05). Steepest simulated keratometry, average simulated keratometry, and inferior–superior value significantly decreased ( P < .05), unlike flattest simulated keratometry, cylindrical simulated keratometry, and apex keratometry ( P > .05). Total and coma-like aberrations were not decreased significantly for all pupil diameters ( P > .05). No significant endothelial changes were observed in either group.
Conclusions
The PRK plus CXL procedure may be a good option to reduce corneal aberrations and stabilize corneas with progressive keratoconus.
Corneal cross-linking (CXL) using riboflavin and ultraviolet A has been used to stabilize the cornea with progressive keratoconus or ectatic corneal disorders after corneal refractive procedures. Further investigations are being conducted regarding its use for corneal melting and bullous keratopathy. Although the potential advantage of CXL over penetrating keratoplasty is well recognized in keratoconic corneas, the former procedure raises concerns about visual and topographic outcomes in treated eyes. Studies with a mean follow-up ranging from 10.69 to 36 months demonstrated that the simultaneous wavefront optimized approach of photorefractive keratectomy (PRK) plus CXL offers improvements in uncorrected distance acuity (UDA), best distance acuity (BDA), and topographic irregularity, even if the surgical goal is not a refractive end point.
The aim of this prospective, nonrandomized, single-center study was to evaluate and compare the results obtained by topographic PRK with minimal ablation using the iVIS Suite platform (Ligi Tecnologie Medicali, Taranto, Italy) followed by CXL as a single procedure with those obtained by CXL treatment alone. Visual acuity, refractive, topographic, corneal higher-order aberration (HOA), and tomographic outcomes up to 24 months after surgery were analyzed.
Methods
Seventeen patients (4 women and 13 men) with progressive keratoconus in both eyes were enrolled in this study. Mean age was 31.17 ± 8.12 years (range, 21 to 46 years). Inclusion criteria were a documented progression of keratoconus in the previous 12 months, a corneal thickness of at least 450 μm at the thinnest point in the worse eye, hard contact lens and full spectacle correction intolerance because of blurred or distorted vision or subjective perception of comet-like asymmetric starbursts, and age older than 18 years. The diagnosis of keratoconus was made based on corneal topography results (Orbscan IIz; Bausch & Lomb, Rochester, New York, USA) as an asymmetric bowtie pattern, with or without skewed axes, and a paracentral inferior–superior dioptric difference of more than 1.4 diopters (D). Progression of keratoconus was defined, based on serial differential topography, as an increase in the apex keratometry by more than 1.0 to 1.5 D and a corresponding change (> 1.0 to 1.5 D) in the refractive cylinder in the previous 6 months. Exclusion criteria were a corneal thickness of less than 450 μm at the thinnest point in the worse eye, corneal scarring or Vogt striae, any history of other ocular disease or eye surgery, any systemic disease, and pregnancy. Patients who failed to attend the follow-up visits also were excluded.
Based on the treatment received, the 34 eyes were assigned to 1 of 2 groups: all patients underwent the combined procedure, namely PRK immediately followed by CXL as a single procedure (17 eyes) in the worse eye, and CXL alone in the fellow eye (17 eyes). At least 3 months elapsed between surgery on the first and then the fellow eye. The 34 eyes were graded as stage I to III according to the Aliò and Shabayek classification, using the C.S.O. EyeTop Topographer Corneal Aberrometry Program (Compagnia Strumenti Oftalmici, Firenze, Italy) for 6-mm pupil diameter; grade I keratoconus is characterized by a mean keratometry value of less than 48.00 D and a coma-like root mean square (RMS) value of between 1.50 and 2.50 μm for a 6.0-mm simulated pupil. Grade II keratoconus is characterized by a mean keratometry value of less than 53.00 D and a coma-like RMS value of between 2.5 and 3.5 μm. Grade III keratoconus is characterized by a mean keratometry value of less than 55.00 D and a coma-like RMS value of between 3.5 and 4.5 μm. In the PRK plus CXL group, 9 eyes had keratoconus stage I, 5 eyes had keratoconus stage II, and 3 eyes had keratoconus stage III. In the CXL alone group, 10 eyes had keratoconus stage I, 4 eyes had keratoconus stage II, and 3 eyes had keratoconus stage III.
Patient Assessment
At baseline and at each follow-up (1, 3, 6, 12, 18, and 24 months), a full ophthalmic examination was performed, including assessment of UDA and BDA (JANG vision tester; Sifi Diagnostic, Treviso, Italy), slit-lamp microscopy examination, optical and Scheimpflug-based topography and tomography (Orbscan II; Orbtec, Bausch & Lomb; Precisio; Ligi Tecnologie Medicali, Taranto, Italy; respectively), measurement of corneal HOA (C.S.O. EyeTop Topographer; Compagnia Strumenti Oftalmici, Firenze, Italy), pachymetry using optical coherence tomography (OCT; Carl Zeiss Meditec Inc, Dublin, California, USA), and confocal microscopy (Corneal Confocal Microscope CS4; NIDEK Technologies, Erlangen, Germany). To improve the reliability of topographic, tomographic, aberrometric, and interferometric measurements, at least 3 consecutive scans were performed by the same examiner for each eye, and the mean values of all scans were used for data analysis.
Surgical Technique
All treatments were performed with the iVIS Suite platform by the same experienced surgeon (G.A.) at the Department of Ophthalmology of Bari University Hospital from June 26, 2008, through July 8, 2009. The iVIS Suite custom ablation system combines the Scheimpflug-based topographer and tomographer Precisio, the dynamic pupillometer pMetrics, the topography-based ablation Corneal Interactive Programmed Topographic Ablation (CIPTA) planning software, and the high-resolution 1-KHz flying spot laser iRES. The steps of both topographic-guided PRK plus CXL as a single procedure (worse eye) and the CXL procedure alone (better eye) are reported below briefly.
The first part of the PRK plus CXL treatment consisted of acquisition of the corneal shape by means of the Precisio tomographer, which uses a rotating Scheimpflug camera to generate a 3-dimensional model of the cornea and anterior segment, with a repeatability of less than 3 μm. The elevation data from 50 high-resolution images with 39 000 measurement points on the anterior and posterior corneal surface from all captured images were combined with the patient’s subjective refraction and were imported into the CIPTA planning software. Variations in pupil size were not taken into consideration.
Transepithelial topography-guided custom ablation with CIPTA software has been described elsewhere. This is a single-step, no-touch ablation technique involving the corneal epithelium and stroma; the uninterrupted, customized ablation procedure was planned to remove the corneal epithelium and the stroma. To minimize the tissue consumption and to assure the smoothness of the transition zone toward the untreated area, the CIPTA software provides the option of the Restored Morphological Axis strategy. The goal is to achieve an aspherical surface with a basic curvature corresponding to the flattest meridian, defined by the Precisio tomographer elevation map. The center of this surface is fitted to the corneal morphologic axis, which is not the visual axis, but rather a computer-generated axis of the restored corneal optical symmetry that approximates the best match between the axis of the ideal shape and that of the true shape of the cornea. In this way, the postoperative corneal surface is symmetrical with respect to the preoperative morphologic features and the change of corneal curvature allows the maximum reduction of irregularities with the minimum tissue consumption. The center of the planned ablation was the corneal apex in all treated eyes. The optical zone ranged between 2.16 and 5.45 mm; the diameter of the transition zone ranged between 6.00 and 8.75 mm. Ablation stromal depth was planned between 18 and 49 μm (mean, 31.1 ± 9.5 μm). Because of the transepithelial treatment, a supplementary depth of the ablation for the epithelium was added, but in keratoconus eyes, the epithelial thickness profile was altered, so the value was selected arbitrarily as 50-μm in all cases by the surgeon. The corneal epithelium was removed by laser within a 9-mm diameter in all cases. Because of variations in the epithelial thickness map in keratoconic corneas, at the end of the photoablative procedure, possible remaining cells at the base of the cone were removed mechanically by the surgeon using a blunt spatula. The ablation plan was transferred to the iRES excimer laser, which is a 193-nm dual flying-spot laser, with the fluence adjusted to 250 mJ/cm 2 , a maximum frequency of delivery of 1 KHz, and an infrared active-passive eye tracker for x, y, and cyclotorsional tracking. After topical anesthesia with 4% lidocaine drops, the laser beam, generated within the iRES laser, was delivered to the corneal surface. Within the selected optical zone, the aim was to normalize the anterior corneal surface as much as possible, rather than treating the refractive error itself.
After PRK, 2% pilocarpine drops were instilled to induce myosis of the pupil, and a CXL procedure was performed, according to the methodology described by Wollensak and associates. Riboflavin 0.1% solution (10 mg riboflavin-5-phosphate in 20% dextran T500 solution; Ricrolin; SOOFT Italia S.p.A., Montegiorgio, Italy) was applied to the cornea every 2 minutes for 20 minutes. Using a slit lamp with blue filter inserted, the surgeon confirmed the presence of a yellow flare in the anterior chamber before the cornea was exposed to the ultraviolet A emitter Vega CBM X-linker (SOOFT Italia S.p.A.) for 30 minutes. During ultraviolet A exposure, riboflavin drops were applied again every 2 minutes.
The eye with lesser keratoconus staging received only CXL treatment. After removing the central 9.0 mm epithelium by mechanical debridement, using a blunt spatula (Desmarre corneal knife; John Weiss & Son Ltd, Milton Keynes, UK), the procedure was performed according to the steps described above. Before every CXL treatment, the intended irradiance (3 mW/cm 2 at 3 cm) of the VEGA CBM X-linker was checked with a LaserMate Q ultraviolet A meter (Laser 2000, Wessling, Germany).
The postoperative medical treatment was the same for both groups of eyes. After surgery, a soft bandage contact lens was applied until re-epithelialization was complete and patients were instructed to use 0.3% ofloxacin (Exocin; Allergan, Roma, Italy) and 0.1% indomethacin (Indocollirio; Baush+Lomb, Vimodrone, Milano, Italy) eye drops for the first 5 days, then, after complete re-epithelialization and removal of contact lens, patients received 0.1% topical fluorometholone (Flumetol; Thea Farma, Settimo Milanese, Italy) 4 times daily for the first month. The dosage was tapered by 1 drop monthly over the next 3 months. Postoperative follow-up was scheduled at 1, 3, 6, 12, 18, and 24 months after each eye surgery. Follow-up was 24 months for all eyes of both groups included in the study. All data were collected in an Excel spreadsheet (Microsoft, Redmond, Washington, USA) and were reported as mean ± standard deviation. The paired 2-tailed Student t test was performed to analyze the postoperative outcome changes compared with baseline values and to analyze the postoperative outcome changes over time. The unpaired t test was performed to compare outcome data at the last follow-up visit between the PRK plus CXL group and the CXL alone group. The level of statistical significance was set at P < .05.
Results
Visual Acuity and Refractive Outcome
Table 1 summarizes preoperative and postoperative visual and refractive data for both groups: PRK plus CXL and CXL alone. UDA and BDA changes over time are shown in Figures 1 and 2 . In the PRK plus CXL group, the eyes with UDA of 20/40 or better increased from 3 (17.6%) before surgery to 14 (82.3%) at the last follow-up examination; in the CXL only group, the eyes with UDA of 20/40 or better were 3 (17.6%) before surgery, and these remained unchanged at the last postoperative visit.
| PRK plus CXL Group | CXL Group | P Value b | P Value c | |||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 24-Month Follow-up | P Value a | Baseline | 24-Month Follow-up | P Value a | |||
| UDA (logMAR) | 0.63 ± 0.36 (1.6 to 0.18) | 0.19 ± 0.17 (0.5 to 0.00) | 4.77 × 10 −4 | 0.59 ± 0.29 (1.18 to 0.18) | 0.52 ± 0.29 (1.00 to 0.00) | .07 | .72 | 4.03 × 10 −4 |
| BDA (logMAR) | 0.06 ± 0.08 (0.3 to 0.00) | 0.03 ± 0.06 (0.18 to 0.00) | .198 | 0.06 ± 0.11 (0.3 to 0.00) | 0.04 ± 0.07 (0.18 to 0.00) | .05 | 1.00 | .019 |
| MRSE (D) | −1.94 ± 1.30 d (–3.25 to +2.00) | −0.19 ± 0.65 d (–1.75 to +1.25) | 9.15 × 10 −6 | −1.54 ± 1.37 e (–3.25 to +0.75) | −1.09 ± 0.87 e (–2.75 to +0.50) | .148 | .38 | .003 |
| Spherical power (D) | 0.95 ± 1.49 (–2.00 to +4.00) | +0.11 ± 0.46 (–0.75 to +1.25) | .02 | −0.13 ± 1.02 (–1.5 to +2.00) | +0.32 ± 0.65 (–1.00 to +1.50) | .03 | .07 | .285 |
| Cylindrical power f (D) | −2.48 ± 1.01 (–0.75 to –4.50) | −0.88 ± 0.79 (0.00 to –2.50) | 8.18 × 10 −5 | −2.78 ± 1.27 (–0.50 to –4.00) | −2.34 ± 1.25 (0.00 to –4.00) | .059 | .45 | 1.73 × 10 −4 |
a Differences between preoperative and last postoperative values.
b Differences between PRK plus CXL and CXL alone values before surgery.
c Differences between PRK plus CXL alone values 24 months after surgery.
d In the PRK plus CXL group, preoperative MRSE was positive in 4 cases, negative in 11 cases, and 0 in 2 cases; 24-month postoperative MRSE was positive in 2 cases, negative in 10 cases, and 0 in 5 cases.
e In the CXL alone group, preoperative MRSE was positive in 1 case, negative in 14 cases, and 0 in 2 cases; 24-month postoperative MRSE was positive in 1 case, negative in 13 cases, and 0 in 3 cases.
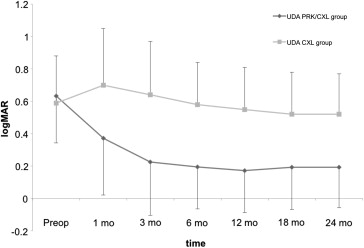
The stability of UDA, defined as 1-line (Snellen) difference in UDA between follow-up visits, was analyzed at different time points after treatment for both groups. In the PRK plus CXL group, from the 1-month to the 3-month examination, 6 (35.3%) eyes gained more than 1 Snellen line of UDA, and 11 (64.7%) eyes remained stable. From the 3-month to the 6-month examination, 7 (41.2%) eyes gained 1 or more than 1 Snellen line of UDA and 10 (58.8%) eyes remained stable. From the 6-month to the 12-month examination, 6 (35.2%) eyes gained 1 Snellen line of UDA and 11 (64.7%) eyes remained stable. From the 12-month to the 18-month examination, 2 (11.7%) eyes gained 1 Snellen line of UDA, and 13 (76.4%) eyes remained stable. From the 18-month to the 24-month examination, all the eyes (100%) were stable. No eye lost Snellen lines of UDA.
In the CXL only group, from the 1-month to the 3-month examination, 3 (17.6%) eyes gained 1 or more than 1 Snellen line of UDA, and 14 (82.3%) eyes remained stable. From the 3-month to the 6-month examination, 3 (17.6%) eyes gained 1 or more than 1 Snellen line of UDA, and 14 (82.3%) eyes remained stable. From the 6-month to the 12-month examination, from the 12-month to the 18-month examination, and from the 18-month to the 24-month examination, all the eyes (100%) were stable. No eye lost Snellen lines of UDA.
At the last postoperative examination, in the PRK plus CXL group, no eye lost lines of BDA and 6 eyes (35.2%) gained 1 to 3 lines of BDA, as compared with preoperative data; in the CXL only group, no eye lost lines of BDA and 3 eyes (17.6%) gained 1 line of BDA, as compared with preoperative data ( Figure 3 ).
Spherical error was present before surgery in 15 (88.2%) eyes in the PRK plus CXL group and in 13 (76.4%) eyes in the CXL only group. All patients had astigmatic error. At the last follow-up visit, spherical error was present in 7 (41.1%) eyes in the PRK plus CXL group and in 10 (58.8%) eyes in the CXL group.
In both groups, at different time points after surgery, mean manifest refraction spherical equivalent was stable within a 1.00-D difference ( Figure 4 ). Also, vector analysis of astigmatic correction according to the Alpin method, based on refractive data, was performed in both groups ( Table 2 ).
| PRK plus CXL Group | CXL Group | |
|---|---|---|
| Targeted induced astigmatism | 2.53 ± 1.1 | 2.68 ± 1.2 |
| Surgically induced astigmatism | 2.13 ± 1.1 | 1.61 ± 1.1 |
| Difference vector | 0.63 ± 0.67 | 1.69 ± 2.27 |
| Angle of error | 6.8 ± 17 | 7.76 ± 30.5 |
| Magnitude of error a | −0.4 ± 0.6 | −1.17 ± 1.39 |
| Index of success b | 0.3 | 0.7 |
a The negative value of the magnitude of error indicates an undercorrection.
b The index of success is directly proportional to the difference vector and inverse to the targeted induced astigmatism vector; the ideal result is 0, implying that the targeted astigmatism correction has been achieved.
Topographic Results
Table 3 summarizes topographic and keratoconus indices as measured with the C.S.O. Topographer at baseline and 24 months after surgery for both groups. In the entire cohort, the following anterior corneal surface parameter was evaluated with the Precisio tomographer: maximum anterior corneal elevation above the best fit sphere in the central 3-mm diameter area before surgery and at last follow-up time. After surgery, in each case, the sphere reference was fitted to the preoperative best fit sphere value. The results are reported in Table 4 .
| PRK plus CXL Group | CXL Group | P Value b | P Value c | |||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 24-Month Follow-up | P Value a | Baseline | 24-Month Follow-up | P Value a | |||
| SimK-1 | 44.83 ± 11.67 | 43.17 ± 2.97 | 6.23 × 10 −8 | 44.92 ± 2.76 | 43.85 ± 1.38 | .23 | .97 | .39 |
| SimK-2 | 47.41 ± 12.44 | 44.94 ± 3.33 | 3.33 × 10 −9 | 47.17 ± 3.74 | 46.08 ± 3.21 | 1.3 × 10 −3 | .94 | .31 |
| SimK-AVG | 46.12 ± 12.04 | 44.05 ± 3.11 | 6.66 × 10 −8 | 46.01 ± 3.18 | 44.86 ± 1.92 | .04 | .97 | .36 |
| SimK-Cyl | 2.70 ± 1.56 | 1.58 ± 1.29 | 3.9 × 10 −3 | 2.29 ± 1.00 | 2.25 ± 0.94 | .76 | .36 | .09 |
| AK | 54.46 ± 14.3 | 50.00 ± 4.60 | 3.68 × 10 −7 | 54.14 ± 2.79 | 52.79 ± 3.36 | .07 | .92 | .05 |
| IS | 6.31 ± 3.34 | 3.28 ± 2.72 | 6.46 × 10 −6 | 5.86 ± 1.40 | 5.08 ± 1.46 | 1.28 × 10 −6 | .61 | .02 |
a Differences between preoperative and last postoperative values.
b Difference between the PRK plus CXL and CXL alone groups before surgery. The baseline 6 CSO indices in the PRK plus CXL group were not significantly different from the same baseline measurements in the CXL alone group.
c Difference between the PRK plus CXL and CXL alone groups 24 months after surgery. All 6 CSO indices were decreased, but the difference between the 2 groups was not significant for SimK-1, SimK-2, SimK-AVG, and SimK-Cyl, whereas the difference was significant for AK and IS values.
| Baseline | PRK plus CXL Group | CXL Group | P Value a | P Value b | P Value c | ||
|---|---|---|---|---|---|---|---|
| 24-Month Follow-up | P Value a | Baseline | 24-Month Follow-up | ||||
| 39.47 ± 18.07 (5 to 88) | 3.21 ± 29.90 (–48 to 64) | 4.18 × 10 −7 | 38.98 ± 16.5 (6 to 57) | 32.52 ± 11.4 (3 to 35) | .76 | .87 | 4.32 × 10 −5 |
a Differences between preoperative and last postoperative values.
b Differences between the PRK plus CXL and CXL alone groups before surgery. The preoperative maximum anterior corneal elevation was not significantly different between the 2 groups.
c Differences between the PRK plus CXL and CXL alone groups 24 months after surgery. Maximum anterior corneal elevation was significantly different between the 2 groups.
The topographic stability was analyzed at different time points after treatment for both groups, stability being defined as a 1.00-D difference in average simulated keratometry, apex keratometry, and inferior–superior value indices between follow-up visits. In both groups, mean average simulated keratometry, mean apex keratometry, and mean inferior–superior values were stable within a 1.00-D difference ( Figures 5 and 6 ).




