Purpose
To investigate the influence of photodynamic therapy (PDT) and combination of PDT and ranibizumab on aqueous humor levels of vascular endothelial growth factor (VEGF) in polypoidal choroidal vasculopathy (PCV).
Design
Prospective randomized clinical trial.
Method
We included 20 eyes with treatment-naïve PCV and 20 eyes undergoing cataract surgery as controls. PCV eyes were randomized to treatment with PDT alone or to a combination of ranibizumab and PDT on the same day. During 3 months, retreatment was not performed. Aqueous humors were collected at baseline and at 1 week, 1 month, and 3 months after treatment in the PCV group and during cataract surgery in the control group. VEGF levels were measured using multiplex bead immunoassay.
Results
At baseline, VEGF levels were significantly increased in PCV eyes compared with control eyes. A significant decrease in VEGF levels was found at 1 week after PDT treatment (n = 8) and at all time points after combination treatment (n = 12). With combination treatment, VEGF levels were decreased to values below the detection limit in all eyes at 1 week and 1 month and in 7 of 12 eyes at 3 months. There was no difference in the clinical profiles among the 2 treatment groups at each time point.
Conclusion
Decreased levels of VEGF detected 1 week after PDT for PCV seems to reflect acute damage of vascular endothelial cells, one of the VEGF expression sites in PCV. Concomitant ranibizumab resulted in a further decrease in VEGF to negligible levels, but this result did not affect the clinical results for 3 months.
It is still unclear whether polypoidal choroidal vasculopathy (PCV) is a subtype of neovascular age-related macular degeneration (AMD) or a unique choroidal vascular abnormality. The results of several histopathologic and optical coherence tomography (OCT) studies on PCV were found to be conflicting in terms of pathogenic mechanisms. The role of vascular endothelial growth factor (VEGF) in the pathogenesis of PCV has also been investigated. Strong expression of VEGF positivity was found in vascular endothelial cells and in retinal pigment epithelial (RPE) cells in 2 studies using surgical specimens, while a lack of positivity was reported in 1 study. In studies measuring ocular VEGF in eyes with PCV, the concentration of VEGF in aqueous humor was higher when compared with normal controls; however, VEGF levels were much lower in comparison to eyes with neovascular AMD. It seemed that VEGF had a lesser contribution to the pathogenesis of PCV compared to neovascular AMD. Nonetheless, these observations provide a biologic rationale for the treatment of PCV with anti-VEGF agents.
Various trials have shown that anti-VEGF agents are effective in reducing leakage, resolving fluids, and improving visual acuity, but are ineffective for polyp regression. Continuous injections are usually required to prevent recurrence of fluid collection. Photodynamic therapy (PDT) is highly effective for the regression of polyps and for the resolution of associated exudates, with excellent visual outcomes at 6- or 12-month follow-up. However, recurrence seems to be inevitable because of the persistence of a branching vascular network from which recurrent polyps and/or a more typical choroidal neovascularization (CNV) may develop. Repeated PDT exposure may culminate in extensive choroidal nonperfusion and VEGF upregulation. Reduced-fluence PDT has been attempted to decrease this inherent risk, with quite promising results. Recently, combination treatment has gained an important role in the treatment of PCV. Use of PDT with angio-occlusive actions along with an anti-VEGF agent with antiangiogenic and antipermeability effects may be synergistic. In particular, adding an anti-VEGF agent can counteract VEGF upregulation resulting from PDT-induced tissue hypoxia.
The histology of PDT-treated human eyes has demonstrated an occluded choriocapillary layer within the treated area, which has been confirmed by indocyanine green angiography (ICGA), showing an early and long-lasting choroidal nonperfusion. This hypoxia can lead to a counterproductive angiogenic response predisposing to recurrent neovascularization. Enhanced VEGF expression by RPE cells was observed 3 days after PDT in a surgical specimen of CNV membrane secondary to AMD. However, no information is available regarding the changes of VEGF after PDT in PCV, either at the tissue level or in ocular fluid. There have been no studies investigating the biologic response of VEGF after PDT in PCV or the impact of concomitant anti-VEGF agents on this profile. This type of investigation would provide valuable information in determining the optimal treatment method as well as the optimal mode, dosage, and timing of anti-VEGF agents in combination treatment.
We carried out a prospective study in patients with PCV to analyze the concentration of VEGF in aqueous humor and to identify characteristic changes following PDT alone and combined with ranibizumab. In addition, baseline VEGF levels were compared with those of unaffected control eyes to investigate their potential role in the pathogenesis of PCV.
Methods
This prospective randomized clinical trial was performed at the Department of Ophthalmology in Seoul St. Mary’s Hospital, Catholic University of Korea. This study was approved by the institutional review board of the Catholic Medical Center and conducted according to the Declaration of Helsinki. Patients gave written informed consent to participate in this research study after a detailed explanation of the study design and purpose. This study is registered at www.clinicaltrials.gov under the identifier NCT01360151 .
Participants
A diagnosis of PCV was established by the presence of polypoidal dilations, with or without a branching vascular network, on ICGA using a confocal scanning laser ophthalmoscope (Heidelberg Retina Angiograph; Heidelberg Engineering, Heidelberg, Germany). Patients with symptomatic PCV, considered as macular fluid on OCT and definite subfoveal, juxtafoveal, or extrafoveal macular polyps on ICGA, were included. Exclusion criteria were any previous treatment for PCV, ocular disease apart from PCV and cataracts, previous ocular surgery other than cataract surgery performed at least 3 months prior, diabetes mellitus, and use of immunosuppressive drugs. Eligible patients were randomized to receive PDT or combination treatment of PDT and 0.5 mg ranibizumab injection, using the table of random sampling numbers.
Age-matched patients who were to undergo cataract surgery were included as a control group. Exclusion criteria for the control group were any ocular disease other than cataracts, systemic diseases including diabetes mellitus, previous vitrectomy, laser treatment, and use of immunosuppressive drugs.
Study Treatments
After screening to determine patients’ eligibility and obtain baseline data, patients in the PCV group were randomized to receive PDT alone or a combination of PDT and ranibizumab. PDT was performed largely according to the protocol from the Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) study. Standard-fluence PDT was used, but the laser spot size was selected based on ICGA findings to cover the entire PCV lesion, including the polypoidal lesions and the branching vascular network. Patients in the combination group were administered an intravitreal injection of 0.5 mg ranibizumab within 1 hour after PDT; intravitreal injections were performed using a standard sterile procedure.
Study Visits and Assessment
All patients in the PCV group were assessed at 1 week, 1 month, and 3 months after treatment. Comprehensive ocular examinations, including best-corrected visual acuity (BCVA) testing and fundus examination with slit-lamp biomicroscopy, were conducted at baseline and at each follow-up visit. OCT (Stratus OCT; Carl Zeiss Meditec, Dublin, California, USA) was performed at each visit; fluorescein angiography (FA) and ICGA (Heidelberg Retina Angiograph; Heidelberg Engineering) were performed at baseline and 3 months after treatment. BCVA results were converted to logarithm of the minimal angle of resolution (logMAR) visual acuities.
Sample Collection and Measurement of Cytokines
Undiluted aqueous humor (100-200 μm) was collected from the PCV group using anterior chamber paracentesis at baseline and at each follow-up visit (1 week, 1 month, and 3 months) after treatment. At baseline, sample collection was performed within 1 hour after PDT in the PDT group and just prior to injection in the combination group. At 1-week, 1-month, and 3-month visits, sample collection was performed after ocular examinations. All sample collections were done using a standard sterile procedure similar to intravitreal injection. Undiluted aqueous humor (100-200 μm) was also collected from the control group during cataract surgery to use as a reference. Samples were immediately frozen and stored at −80 C. The concentration of VEGF was measured in all samples using Luminex multiplex bead immunoassay (Human Cytokine LINCOplex kit; LINCO Research, St. Charles, Missouri, USA). The assay was performed according to the manufacturer’s instructions with Luminex laser-based fluorescent analytical test instrumentation.
Statistical Analysis
Statistical analyses were performed using SPSS statistical software (version 12.0; SPSS Inc, Chicago, Illinois, USA). The Wilcoxon signed-rank test was used to evaluate changes in concentrations of VEGF, logMAR BCVA, and central foveal thickness (CFT) after treatment in the PCV group. The Mann-Whitney U test was used to compare the concentrations of VEGF, logMAR BCVA, and CFT between the 2 treatment groups and to compare the baseline VEGF concentrations of PCV and control groups. The χ 2 test was used to compare noncontinuous variables.
Results
Patient Baseline Characteristics
Forty Korean patients were enrolled in the study: 20 in the PCV group and 20 in the control group. All 20 patients in the PCV group were randomized to treatment; 8 patients were in the PDT group and 12 patients were in the combination group. The baseline characteristics of the 2 treatment groups were well balanced in terms of age, BCVA, and CFT. Also, there was no significant difference in laser spot size for PDT ( Table ).
| PCV Group (N = 20) | Age (y) (Mean ± SD) | Sex (M:F) | Mean LogMAR BCVA (Mean ± SD) | Mean CFT Measured With OCT (μm) (Mean ± SD) | Mean Spot Size of PDT (Mean ± SD) |
|---|---|---|---|---|---|
| Combination treatment a group (n =12) | 63.68 ± 8.78 | 11:1 | 0.46 ± 0.30 | 391.13 ± 130.25 | 2900.00 ± 1169.86 |
| PDT group (n = 8) | 66.33 ± 7.85 | 5:3 | 0.53 ± 0.35 | 481.33 ± 128.46 | 2791.67 ± 800.52 |
| P b | .427 | .851 | .208 | .910 |
a Combination treatment consists of PDT and intravitreal injection of ranibizumab on the same day.
Baseline VEGF Concentrations
At baseline, concentrations of VEGF were significantly increased in PCV eyes compared with control eyes ( P < .001). The concentrations of VEGF were 62.3 ± 37.14 pg/mL in the PCV group and 19.48 ± 20.08 pg/mL in the control group.
Post-treatment VEGF Concentrations
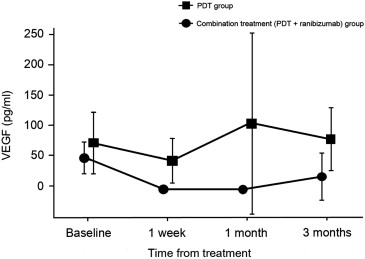
In the PDT group, VEGF levels significantly decreased to 41.75 ± 36.11 pg/mL 1week after treatment ( P = .025) and then rebounded to 103.61 ± 150.14 pg/mL at 1 month and to 76.99 ± 51.45 pg/mL at 3 months. These 1- and 3-month values were not significantly different from baseline values (70.91 ± 50.81 pg/mL; P = .779 and P = .208, respectively).
In the combination treatment group, VEGF levels were reduced from a mean of 56.56 ± 25.49 pg/mL to values below detection limit (<3.20 pg/mL) in all patients at 1 week and 1 month after treatment. At 3 months, VEGF levels remained below physiologic limits in 7 of 12 eyes, and the mean VEGF level (24.46 ± 38.96 pg/mL) was significantly lower than the baseline level. The VEGF concentration was significantly higher in the PDT treatment group compared to the combination treatment group at each time point ( P = .002, P = .002, and P = .041 at 1 week, 1 month, and 3 months, respectively) ( Figure ).