38
Periocular Steroids
Beatriz S. Takahashi  Jason S. Slakter
Jason S. Slakter  Howard F. Fine
Howard F. Fine
INTRODUCTION
Periocular steroids were first used clinically in the 1950s. Delivering the medication locally maximizes the effect of the drug while minimizing systemic side effects. There are different ways of delivering periocular corticosteroids (1):
 Subconjunctival injection: the drug is introduced between the conjunctiva and Tenon’s capsule. The procedure is simple and safe to perform, but less of the drug is absorbed.
Subconjunctival injection: the drug is introduced between the conjunctiva and Tenon’s capsule. The procedure is simple and safe to perform, but less of the drug is absorbed.
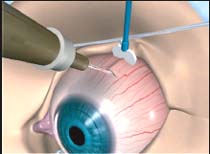
 Anterior sub-Tenon’s injection: the drug is injected between Tenon’s capsule and the sclera. This can be performed in any quadrant; the drug is visible after delivery.
Anterior sub-Tenon’s injection: the drug is injected between Tenon’s capsule and the sclera. This can be performed in any quadrant; the drug is visible after delivery.
 Posterior sub-Tenon’s injection: the drug is injected in a similar fashion to an anterior sub-Tenon’s injection, with the patient looking away from the quadrant chosen to allow more posterior access. The drug typically cannot be visualized, and the minor risk of accidental globe perforation increases slightly.
Posterior sub-Tenon’s injection: the drug is injected in a similar fashion to an anterior sub-Tenon’s injection, with the patient looking away from the quadrant chosen to allow more posterior access. The drug typically cannot be visualized, and the minor risk of accidental globe perforation increases slightly.
 Retrobulbar injection: a standard retrobulbar injection with a shorter needle is performed.
Retrobulbar injection: a standard retrobulbar injection with a shorter needle is performed.
TRIAMCINOLONE ACETONIDE
Triamcinolone acetonide (TA), or 9α-fluoro-16α-hydroxyprednisolone, is an intermediate-acting corticosteroid. It is supplied as a crystalline suspension (Bristol-Myers Squibb, Princeton, NJ), which allows it to dissolve in and around the eye in a sustained-release fashion. The intravitreal use of triamcinolone is a well-established procedure (2–4).
As with other corticosteroids, the use of periocular TA has also been associated with local sequelae including sterile conjunctival ulceration (5), intraocular pressure rise and cataracts (6, 7), central serous chorioretinopathy (8), as well as ptosis and orbital fat prolapsed (9–11), cutaneous hypopigmentation (12) and periocular abscess (13). The location of the drug and its proximity to the macular area is important for the outcome. When sub-Tenon’s injection is performed, a superiotemporal procedure is typically preferred (14).
Cytotoxicity in vitro has been reported (15, 16). The preservation vehicle used for triamcinolone (0.99% benzyl alcohol, 0.75% carboxymethylcellulose, 0.04% polysorbate 80) has also been studied for toxicity to human retinal pigment epithelium cells (ARPE19). The viability of the cells was the greatest in preservative free formulations, followed by the vehicle alone and then by the trade formulation (17). The vehicle used with TA has also been showed to have toxicity to the retina and to the lens after intravitreal injection in an animal model (18).
TA was shown to reduce the expression of ICAM-1 when modulated by phorbol 12-myristate 13-acetate in a human bladder carcinoma-derived epithelial cell line in vitro (19) as well as to inhibit choroidal endothelial cells migration and permeability in a dose dependant manner (20). Another experiment with oxygen-induced retinopathy in rats showed decreased vascular leakage with 20 mg/ml triamcinolone (21), a smaller area of neovascularization, a lower concentration of vascular endothelial growth factor (VEGF) and blockage of upregulation of proinflammatory factors such as TNFα, IL-1β, ICAM-1, p-p38 MAPK, p-JNK MAPK, p-ERK MAPK, p-IkB, NFkB, HIF-α (22). Neovascularization has a dose dependant response to TA. Other experiments with triamcinolone showed less leukocyte accumulation in ischemia induced eyes (23), smaller retinal endothelial cell nuclei counts compared to controls in a rat model of retinopathy of prematurity (24), inhibition of corneal neovascular response after subconjunctival injection of the corticosteroid (25), degenerative changes on fibroblasts, and more macrophages within as well as adjacent to an area where the drug was injected subconjunctivally before glaucoma surgery (26).
In an experimental study with laser treated rats, 2 mg or 0.5 mg of posterior sub-Tenon’s triamcinolone was administered (27). Animals treated with 2 mg of triamcinolone showed statistically significantly lower leakage than 0.5 mg treated and controls. When treatment was given, the choroidal neovascularization (CNV) membranes were thinner than the control eyes. Similar results were observed in laser treated models (28).
Periocular triamcinolone has been successfully used to treat macular edema secondary to branch vein occlusion associated with retinal arteriovenous malformation (29). Changes in the central retinal thickness were also observed in patients treated for refractory cystoid macular edema for diabetic retinopathy with retrobulbar triamcinolone. In those patients, intraocular pressure elevation was observed, but all cases could be controlled with topical glaucoma medications (30).
A prospective study evaluating the subconjunctival injection of triamcinolone demonstrated the successful treatment of 10 patients with recalcitrant anterior scleritis; six of those patients were able to discontinue systemic therapy. The symptoms of pain, redness, and scleral inflammation resolved in all but one patient. One patient developed high intraocular pressure which was stabilized with topical medication and another had subconjunctival hemorrhage (31). Nonnecrotizing anterior scleritis could also be controlled with subconjunctival triamcinolone (32, 33).
In a clinical trial comparing posterior sub-Tenon’s injection of triamcinolone and orbital floor methylprednisolone for noninfectious posterior uveitis with cystoid macular edema and/or vitritis, improvement rates were not statistically different between groups. Both groups enjoyed a statistically significant improvement in visual acuity at 6 weeks and at 3 months. Of 30 eyes that received sub-Tenon triamcinolone, 2 patients in the triamcinolone group had lid ptosis and another 2 developed raised intraocular pressure (34–38).
Posterior sub-Tenon’s triamcinolone was used in a prospective case control study to evaluate its effects before panretinal photocoagulation in diabetic retinopathy (34). Eyes treated with triamcinolone had less increase in their retinal thickness and better final visual acuity than eyes not treated with the steroids one week before the laser procedure. Pretreatment of diabetic macular edema with sub-Tenon’s triamcinolone before grid pattern laser photocoagulation showed that foveal thickness at 24 weeks was thinner in treated patients than in controls, but no difference was observed in vision (35). Another randomized study showed no advantage on ocular coherence tomagraphy (OCT) thickness or final visual acuity in patients treated with peribulbar TA with and without focal laser; all randomized groups showed reduction in retinal thickening and no benefit on visual acuity (36).
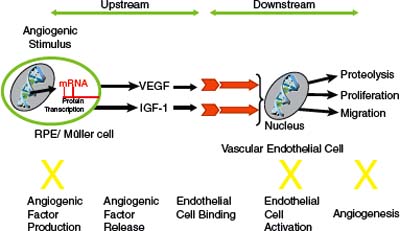
Figure 38-1. Anecortave acetate inhibits the angiogenic proteolytic cascade by inhibiting the expression of urokinase plasminogen activator and matrix metalloproteinases and up-regulating the expression of the urokinase plasminogen activator inhibitor plasminogen activator inhibitor-1. In addition, anecortave acetate inhibits vascular endothelial cell proliferation and migration. IGF, insulin-like growth factor; VEGF, vascular endothelial growth factor. (Adapted from Slakter JS. Anecortave acetate for treating or preventing choroidal neovascularization. Ophthalmol Clin North Am 2006;19(3):373–380).
Intravitreal triamcinolone (IVTA) was compared to posterior sub-Tenon’s triamcinolone in a study for diffuse diabetic macular edema by Cardillo. Central macular thickness of eyes treated with IVTA was statistically thinner than eyes treated with posterior sub-Tenon’s triamcinolone at 1 month and 3 months, but the difference not confirmed at 6 months. For visual acuity, both treatments raised the acuity, but IVTA was statistically better at 3 months, again not statistically significant at 6 months. None of the study eyes, previously treated with topical 1% prednisolone, showed increase in intraocular pressure (IOP) (39). Another randomized clinical trial for refractory diabetic macular edema showed statistically significant results only in eyes treated with intravitreal triamcinolone both for thickness of the retina and visual acuity (40). Eyes treated with sub-Tenon’s triamcinolone for diabetic macular edema that did not respond to this approach can still respond when the same corticosteroid is injected intravitreal (41). Similar responses for visual acuity and retinal thickness were observed in groups treated with sub-Tenon’s and with intravitreal injections (42).
A comparison between sub-Tenon’s injection of TA and prednisolone acetate 1% eye drops for controlling inflammation after vitrectomy was reported and no differences in symptoms nor in observable inflammation was seen (37). A case of benign lymphoid hyperplasia was successfully treated with 20 mg/ 0.5 ml subconjunctival injection of triamcinolone close to a salmon patch area (43).
ANECORTAVE ACETATE
Anecortave acetate 15 mg (RETAANE®; Alcon, Forth Worth, TX, USA) is an angiostatic cortisone that inhibits pathologic neovascularization at multiple steps within the angiogenic cascade, and does not show typical ocular glucocorticoid-induced side-effects such as cataract formation and increased intraocular pressure. Anecortave acetate can suppress (a) the expression of some extracellular proteinases (44), (b) endothelial cell proliferation and differentiation, and (c) the synthesis of proangiogenic growth factors and their receptors (Fig. 38-1). Preclinical efficacy pharmacology studies showed that anecortave acetate significantly inhibited corneal, retinal, and choroidal neovascularization as well as tumor growth in various species (44, 45–51). Moreover, it inhibited the growth of the pathologic newly forming vessels, and did not affect preexisting normal vessels. In order to maintain its angiostatic activity and eliminate typical untoward glucocorticoid effects, a hydroxyl group in cortisol was replaced by a double bond at the C9–11 position, and a 21-acetate was added to enhance drug penetration and prolong duration of the depot (Fig. 38-2) (45). Anecortave acetate has one major active metabolite, anecortave desacetate (AL-4940); this metabolite is formed by the deacetylation of the parent molecule. Because anecortave desacetate binds only moderately to plasma proteins and does not inhibit human hepatic cytochrome P-450 isozymes, it is unlikely to participate in drug-drug interactions (46). Genotoxicity, carcinogenicity, and reproductive toxicity studies in vivo revealed no significant ocular or systemic toxicities (47). During clinical trials, the drug was safe and well tolerated both as a primary therapy and as adjunctive therapy to VISUDYNE® photodynamic therapy (PDT) (48).
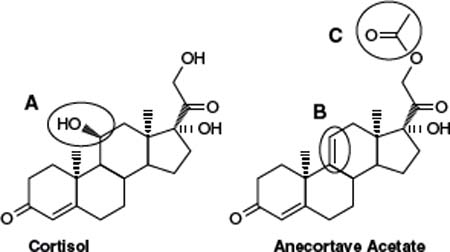
Figure 38-2. Three modifications made to cortisol to generate anecortave acetate. The 11B-hydroxyl group, which is essential for glucocorticoid activity, was removed from cortisol. A double bond between C9 and C11 was added to prevent in vivo enzymatic rehydroxylation at C11, and acetate group was added at C21 to enhance ocular penetration and provide ideal physical-chemical properties for administration of a slow-release depot. (Adapted from Slakter JS. Anecortave acetate for treating or preventing choroidal neovascularization. Ophthalmol Clin North Am 2006; 19(3):373–380.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


