and pulmonary complications. Elevated systemic blood pressure has been found in children with OSA (17, 18, 19, 20, 21). More importantly, children with elevated blood pressures are more likely to suffer from hypertension and metabolic syndrome as adults (22). Although previous studies evaluating the association between primary snoring and increases in blood pressure in children have been somewhat conflicting (23, 24, 25, 26), more recent data suggest that children with primary snoring have higher nighttime blood pressures when compared to those control patients without snoring (27). Further, a dose-response relationship exists between SDB and the severity of blood pressures in children (27). Although not fully understood, clear risks exist for those children with prolonged SDB.
TABLE 139.1 CONDITIONS CAUSING AN INCREASED RISK OF SDB AND OSA IN CHILDREN | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 139.2 FREQUENTLY WITNESSED SIGNS AND SYMPTOMS OF OSA IN CHILDREN | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
pathology. For example, a hyponasal voice can suggest enlarged adenoids, while a muffled voice can result from tonsillar hypertrophy. Care should be taken to perform a thorough cardiovascular exam including auscultation for any murmurs. Systemic findings including failure to thrive, cor pulmonale, and pectus excavatum should be considered (33). Special consideration is given to obesity and body habitus, especially in the adolescent patient. Finally, genetic and syndromal dysmorphisms should also be noted.
TABLE 139.3 SYSTEMATIC APPROACH TO THE PHYSICAL EXAMINATION IN A CHILD WITH OSA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
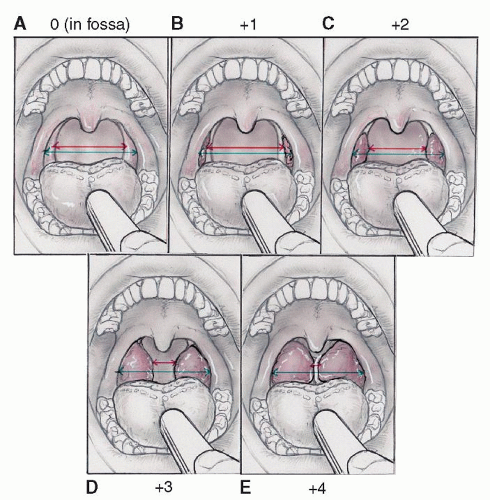
and 4 for tonsils that “kiss,” or approximate, in the midline. An assessment using the modified Mallampati score (36, 37, 38) can also be performed by evaluating the view of the uvula and posterior pharyngeal wall while asking the child to open his/her mouth completely while the tongue is in a resting position. Scoring is grade I when the entire uvula is visible, grade II for a partial view of the uvula, grade III when none of the uvula is visible but some of the soft palate is visible, and grade IV for a view of the hard palate only. Flexible laryngoscopy is recommended in all children under 1 year of age with OSA and when further evaluation is needed in older children. Flexible laryngoscopy allows direct visualization of the base of tongue, pharyngeal walls, vallecula, epiglottis, hypopharynx, vocal folds, and potentially, a portion of the subglottis. This procedure is useful in the evaluation of possible laryngomalacia, lingual tonsil hypertrophy, and persistent OSA despite previous tonsils and adenoids (T&A), and is also useful to document vocal cord mobility and to rule out pharyngeal/hypopharyngeal masses.
TABLE 139.4 PARAMETERS RECORDED DURING NOCTURNAL PSG | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Sleep Medicine (AASM), recommends that PSG is indicated when OSA is suggested by clinical assessment, and all children being considered for adenotonsillectomy for OSA should undergo PSG (48). These guidelines also recommend that PSG be used prior to decannulation in children that required tracheostomy placement, be used postoperatively in children with symptoms of persistent OSA after adenotonsillectomy as well as those with moderate to severe preoperative OSA, and those with high risk for persistent disease such as those with obesity or craniofacial abnormalities (48).
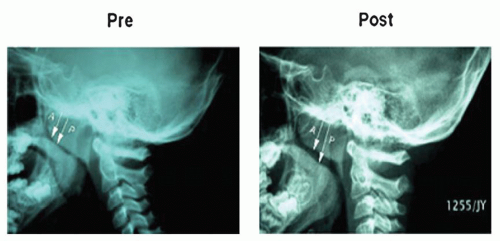 Figure 139.2 Lateral neck films showing effects of a 16-week treatment of Montelukast on adenoid size. (Reprinted from Goldbart AD, Goldman JL, et al. Leukotriene modifier therapy for mild sleep-disordered breathing in children. Am J Respir Crit Care Med 2005;172(3):364-370, Figure 1, with permission.) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree