Purpose
To identify the change in clinical and microbiological profiles of pediatric microbial keratitis in Taiwan between 1998–2002 and 2008–2012.
Design
Retrospective, observational study.
Method
setting : Chang Gung Memorial Hospital, a referral center in Taiwan. patient population : We retrospectively reviewed the medical records of 68 eyes of 67 children aged 16 years or younger who were diagnosed with microbial keratitis and treated at our hospital between July 2008 and December 2012. main outcomes and measures : Predisposing factors, isolated organisms, antibiotic susceptibility, and clinical outcomes. The findings were compared with the results of our previous study conducted between July 1998 and December 2002.
Results
As in 1998–2002, the leading risk factor for microbial keratitis during 2008–2012 was contact lens use, and the infection rate significantly increased from 40.7% to 52.9% ( P = .024), which was mainly attributable to the recent increase in the rate of orthokeratology-related keratitis from 9.9% to 19.1% ( P = .011). Pseudomonas aeruginosa remained the most commonly isolated organism (30.6%), but the number of isolated coagulase-negative Staphylococcus cases increased significantly in the 2008–2012 cases ( P = .04). Antibiotic susceptibility of organisms did not change significantly between the 2 study periods. By using multiple linear stepwise regression analysis, we found that gram-negative bacterial infection played a crucial role in poor visual outcome.
Conclusions
Contact lens–related microbial keratitis increased in Taiwanese children over time, especially because of the use of overnight orthokeratology. Clinicians must understand the infection background and pay further attention to contact lens use in pediatric patients.
Among ocular surface diseases, infectious keratitis poses one of the greatest threats to vision, and delayed or inappropriate treatment of infectious keratitis can lead to severe visual impairment. Microbial keratitis occurs infrequently in childhood and accounts for up to 13% of all microbial keratitis cases ; and pediatric patients with the infection must receive extra attention because the disease history is often understood poorly, the patients do not seek professional care in a timely manner, examining the patients and administering topical medication is challenging, and the patients face the risk of secondary amblyopia. Over the past 20 years, studies conducted in the United States, India, Taiwan, and China have enhanced the understanding of pediatric microbial keratitis.
Epidemiologic patterns of infectious diseases often vary according to the geographic location and population groups and may change over time. The high prevalence of myopia in Asian populations has led to an increase in the popularity of using soft contact lenses or overnight orthokeratology in recent years. Previously, we reported that contact lens use rather than trauma was the main predisposing factor for pediatric microbial keratitis in Taiwan between 1998 and 2002. Because people have become more educated about infectious keratitis since that period and have gained information about the infection from schools and media such as the Internet, people can be expected to have become more careful in using contact lenses and, therefore, more successful in avoiding infectious keratitis.
Whereas a considerable amount of attention has been paid to the clinical profiles of pediatric microbial keratitis, little is known about the in vitro antibiotic susceptibility patterns of the microorganisms that affect the pediatric population. Moreover, frequent and occasionally indiscriminate use of antibiotics has led to the development of resistance to several commonly used antimicrobial agents. Thus, antibiotic susceptibility should be tested periodically to ensure that the antibiotics available during a given period provide adequate protection against clinical isolates of pathogenic bacteria.
Therefore, we reexamined pediatric microbial keratitis between 2008 and 2012 in the same tertiary referral center as that used in 1998–2002 to determine the recent predisposing factors, microbiological spectrums, drug susceptibility, and treatment outcomes and thereby identify changes in disease trends after the 10-year interval.
Methods
Prior to initiation of the study, this retrospective study was approved by the Institutional Review Board of Chang Gung Memorial Hospital (CGMH; 101-1024B), which granted a waiver of consent because patient anonymity was maintained by the data source. We reviewed medical records of patients at CGMH, a tertiary referral center in northern Taiwan, from July 1, 2008, to December 31, 2012. We identified patients by using a computerized diagnostic code search for all inpatients and outpatients who were aged ≤16 years and had been diagnosed with microbial (nonviral) keratitis. Patients who had been administered topical antibiotics before referral and showed negative culture results were included if the clinical findings were consistent with the diagnosis of an infectious ulcer.
The clinical evaluation involved visual acuity testing (when possible) and slit-lamp examination. An ulcer was defined as being central if it encroached within 2 mm of fixation, peripheral if it involved a zone within 2 mm from the limbus, and paracentral if it was in the intervening zone. Corneal ulcers were defined as being small (<2 mm), medium (2–6 mm), and large (>6 mm).
Smears and cultures from corneal scrapings for bacteria, mycobacteria, and fungi were performed. The specimens were inoculated in blood agar, chocolate agar, modified Sabouraud agar, Lowenstein-Jensen agar slant, and thioglycolate broth. Non-nutrient agar seeded with Escherichia coli for Acanthamoeba was used for patients with suggestive clinical characteristics. In vitro susceptibility was determined using the disk-diffusion method based on the Clinical and Laboratory Standard Institute (CLSI) standards for testing antimicrobial susceptibility based on the serum standards. Intermediate susceptibilities were categorized as antibiotic resistant in the organisms. Bacterial isolates were tested for susceptibility to a cephalosporin (ceftazidime), aminoglycosides (gentamicin and amikacin), a fluoroquinolone (ciprofloxacin), oxacillin, piperacillin, and vancomycin.
Levofloxacin (0.5%) alone or a combination of 2 fortified antibiotics (cefazolin 25 mg/mL and amikacin 25 mg/mL) was administered topically once per hour and modification of antibiotics was based on the laboratory results and clinical response. Topical natamycin or amphotericin B (0.1%) was applied hourly for mold or yeast infection, respectively. Topical polyhexamethylene biguanide (0.02%) was used for Acanthamoeba keratitis.
We assessed visual outcome based on visual acuities measured at the last follow-up visit. Snellen visual acuity was converted to a logarithm of minimal angle of resolution for analyses.
Categorical variables were analyzed using the χ 2 test and continuous variables were analyzed using a Student t test. Simple linear regression for univariate analysis was used to identify the factors associated with visual outcome. Multiple linear stepwise regression was performed after univariate analysis. A P value < .05 was considered statistically significant. All statistical analyses were performed using SPSS software, version 20 (IBM, Armonk, New York, USA).
Results
Demographics and Clinical Features
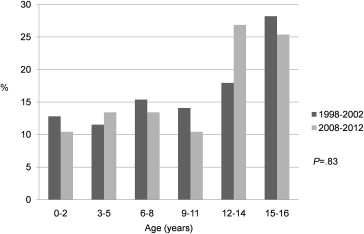
Sixty-eight eyes of 67 patients, of which 29 were outpatients (43.3%) and 38 were inpatients (56.7%), were included in this study. The Figure and Table 1 show the age distribution and the profiles of patients, 25 (37.3%) of whom were boys and the remaining 42 (62.7%) girls; patients had a mean age of 10.3 ± 5.1 years. The percentage of patients in various age groups was similar in the 1998–2002 and 2008–2012 periods ( P = .83), but disease prevalence was higher in the adolescent group in the recent period. The laterality of eye, sex, and age were not significantly different between the 2 study periods.
| 1998–2002 (n = 78) | 2008–2012 (n = 67) | P Value | |
|---|---|---|---|
| N (%) | N (%) | ||
| Age, mean (SD), y | 10.3 (5.3) | 10.3 (5.1) | .94 |
| Sex | .15 | ||
| Male | 36 (46.2) | 25 (37.3) | |
| Female | 42 (53.8) | 42 (62.7) | |
| Eye involved | .24 | ||
| Right | 37 (47.4) | 38 (56.7) | |
| Left | 38 (48.7) | 28 (41.8) | |
| Both | 3 (3.9) | 1 (1.5) | |
| Size | (n = 81) | (n = 68) | <.001 |
| Small | 49 (60.5) | 26 (38.2) | |
| Medium | 30 (37.0) | 35 (51.5) | |
| Large | 2 (2.5) | 7 (10.3) | |
| Location | .31 | ||
| Central | 37 (45.7) | 26 (38.2) | |
| Paracentral | 32 (39.5) | 33 (48.5) | |
| Peripheral | 12 (14.8) | 9 (13.3) |
The infiltrate was small in 26 of the 68 eyes (38.2%), medium in 35 eyes (51.5%), and large in 7 eyes (10.3%). The location of the corneal infiltrate was central in 26 of the 68 eyes (38.2%), paracentral in 33 eyes (48.5%), and peripheral in 9 eyes (13.3%). The prevalence of medium- and large-sized infiltrates was significantly increased ( P < .001) in the recent study period.
Predisposing Factors
The predisposing factors for pediatric microbial keratitis in the 2 study periods are compared in Table 2 . As in the previous study period, contact lens use was the dominant risk factor for microbial keratitis in the recent study period, occurring in 36 of 68 eyes (52.9%) and accounting for 28 patients (28 eyes, 87.5%) among 32 patients aged >12 years. The proportion of contact lens use increased from 40.7% to 52.9% ( P = .024), which was mainly attributable to the recent increase in the rate of orthokeratology use from 9.9% to 19.1% ( P = .011). The patients with orthokeratology were significantly younger than those with soft contact lens (11.9 years vs 14.8 years, P = .001). Trauma remained the second most common risk factor, identified in 11 (16.2%) of 68 eyes and distributed among all age groups; however, in the recent period, systemic disease increased to the same prevalence level as trauma and was also the leading risk factor for patients aged ≤12 years. Three (4.4%) of 68 eyes presented at least 2 predisposing factors.
| Predisposing Factor | 1998–2002 (n = 81) | 2008–2012 (n = 68) | P Value |
|---|---|---|---|
| N (%) | N (% a ) | ||
| Contact lens | 33 (40.7) | 36 (52.9) | .04 |
| Soft contact lens | 25 (30.8) | 23 (33.8) | .60 |
| Orthokeratology | 8 (9.9) | 13 (19.1) | .01 |
| Trauma | 17 (21.0) | 11 (16.2) | .33 |
| Ocular disease | 12 (14.8) | 7 (10.3) | .30 |
| History of corneal ulcer | 0 | 2 (3.0) | |
| Recurrent corneal erosion | 0 | 1 (1.5) | |
| Congenital glaucoma | 0 | 1 (1.5) | |
| Peter’s anomaly | 0 | 1 (1.5) | |
| Orbital rhabdomyosarcoma | 0 | 1 (1.5) | |
| Trichiasis | 6 (7.4) | 1 (1.5) | |
| Other b | 6 | 0 | |
| Systemic disease | 9 (11.1) | 11 (16.2) | .18 |
| Stevens-Johnson syndrome | 0 | 1 (1.5) | |
| Sinusitis | 0 | 1 (1.5) | |
| Hypoxic encephalopathy | 6 (7.4) | 5 (7.4) | |
| Rhabdomyolysis | 0 | 1 (1.5) | |
| Crouzon syndrome | 0 | 1 (1.5) | |
| Congenital heart disease | 1 (1.2) | 1 (1.5) | |
| Prematurity | 0 | 1 (1.5) | |
| Other c | 2 | 0 | |
| Previous ocular surgery | 5 (6.2) | 3 (4.4) | .54 |
| Leukoma cornea | 0 | 1 (1.5) | |
| Neurotrophic keratopathy | 0 | 1 (1.5) | |
| Ptosis | 2 (2.5) | 1 (1.5) | |
| Other d | 3 | 0 | |
| Unknown | 5 (6.2) | 3 (4.4) |
a Total is greater than 100% because 3 patients had multiple risk factors.
b Other included blepharitis (2), epidemic keratoconjunctivitis (3), and phthisis bulbi (1).
c Other included Marshall-Smith syndrome (1) and cerebral palsy (1).
Isolated Organisms
Thirty-six patients received topical antibiotics before referral. Out of the 63 eyes, 36 (57.1%) were culture-positive, and 16 of 27 culture-negative eyes (59.3%) had been previously treated with antibiotics; 4 eyes showed polymicrobial infection. The various isolated microorganisms are listed in Table 3 . The most commonly isolated organism was Pseudomonas aeruginosa (30.6%), followed by coagulase-negative Staphylococcus (CNS, 13.9%) and Propionibacterium acnes (11.1%). The number of isolated CNS and P acnes increased significantly ( P = .04 and P = .02, respectively) in the recent study period.
| Organism | 1998–2002 | 2008–2012 | P Value |
|---|---|---|---|
| N (% a of Positive Culture) | N (% a of Positive Culture) | ||
| Gram-positive cocci | 17 (36.2) | 11 (30.6) | .59 |
| Coagulase-negative Staphylococcus | 1 (2.1) | 5 (13.9) | .04 |
| Staphylococcus aureus | 9 (19.1) | 1 (2.8) | .02 |
| Others | 7 b | 5 c | |
| Gram-positive bacilli | 1 (2.1) | 7 (19.4) | .01 |
| Propionibacterium acnes | 0 | 4 (11.1) | .02 |
| Others | 1 d | 3 e | |
| Gram-negative bacilli | 32 (68.1) | 16 (44.4) | .03 |
| Pseudomonas aeruginosa | 21 (44.7) | 11 (30.6) | .19 |
| Others | 11 f | 5 g | |
| Fungi | 3 (6.4) | 5 (13.9) | .25 |
| Acanthoamoeba | 1 (2.1) | 2 (5.6) | .40 |
| Polymicrobial | 7 (14.9) | 4 (11.1) | .61 |
| Positive cultures | 47 | 36 | .91 |
a Percentage sum was greater than 100% because of polymicrobial infections.
b Others included Streptococcus pneumoniae (5), α-hemolytic streptococci (1), and micrococcus (1).
c Others included Streptococcus pneumoniae (2), viridans streptococcus (2), and Staphylococcus epidermidis (1).
d Others included clostridium (1).
e Others included corynebacterium (2) and unidentified (1).
f Others included Serratia marcescens (4), Haemophilus influenzae (2), Acinetobacter baumanni (1), Citrobacter species (1), and unidentified (3).
g Others included Serratia marcescens (2), glucose nonfermenting group (2), and Haemophilus influenzae (1).
The spectrum of organisms varied depending on the risk factors. Rates of gram-positive and gram-negative isolates were similar among trauma, ocular disease, and systemic disease. Gram-negative bacteria remained the most common microorganisms in contact lens users, but more gram-positive bacteria were isolated in the 2008–2012 period than in the 1998–2002 period ( P = .033).
Antibiotic Susceptibility
For gram-negative bacteria, 87.5% of the isolates were susceptible to gentamicin and amikacin in the 1998–2002 period, and 100% were susceptible in the 2008–2012 period. All P aeruginosa isolates were sensitive to gentamicin, amikacin, ciprofloxacin, ceftazidime, and piperacillin. All the gram-positive bacteria were susceptible to vancomycin. The susceptibility of gram-positive and gram-negative bacteria to various antibiotics did not change significantly between the 2 study periods; susceptibilities of the bacteria to antibiotics are shown in Table 4 .
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


