Purpose
To investigate changes of whole eye axial biometry during accommodation using ultra-long scan depth optical coherence tomography (UL-OCT).
Design
Prospective, observational case series.
Methods
Twenty-one adult subjects were enrolled. Using UL-OCT, the left eye of each subject was imaged with relaxed diopters (0 D) and accommodative stimuli (+6 D). Full eye biometry included central corneal thickness (CCT), anterior chamber depth (ACD), lens thickness, vitreous length, and axial length (AL).
Results
During accommodation (+6 D), the axial biometry of the whole eye changed significantly. Compared to the rest state, ACD at the accommodative state decreased significantly from 3.128 ± 0.305 mm to 2.961 ± 0.298 mm (paired t test, P < .001). The lens thickness increased significantly from 3.723 ± 0.237 mm to 3.963 ± 0.234 mm ( P < .001). The vitreous length decreased significantly from 17.129 ± 0.864 mm to 17.057 ± 0.848 mm ( P < .001). AL was 24.519 ± 0.917 mm at the rest state and increased to 24.545 ± 0.915 mm with +6 D accommodation stimulus. The elongated AL of 26.1 ± 13.4 μm between the rest and accommodative states was significant ( P < .001).
Conclusions
During accommodation, whole eye axial biometry changed, including a decrease in ACD and vitreous length and an increase in lens thickness and AL. UL-OCT provides an alternative method that is suitable for full eye biometry during accommodation.
Human accommodation is a dynamic process that compensates the retinal defocus for near working vision. During accommodation, the accommodation apparatus makes various responses for sharp focusing. With the classical theory, the axial biometry of the whole eye changes, including a decrease in anterior chamber depth (ACD) and vitreous length and an increase in lens thickness. However, changes in the axial length (AL) during accommodation remain controversial, possibly owing to the uncertainty of the crystalline refractive index for calculating AL or the technical limitations of performing full eye biometry during accommodation.
Whether the refractive index changes during accommodation remains unsettled. Research evidence and mathematical modeling have demonstrated that the refractive index may change during accommodation. For instance, Gullstrand’s intracapsular mechanism of accommodation assumed that the average refractive index of crystalline lens increased by 0.0015 per diopter during accommodation. Le Grand considered the average refractive index of the crystalline lens to increase 0.001 per diopter during accommodation. Dubbelman and associates demonstrated that the average refractive index of the crystalline lens increased 0.0013 per diopter during accommodation. On the contrary, other studies showed that the gradient refractive index remained constant with accommodation.
Measurement techniques for full eye biometry during accommodation need to be based on some assumptions to yield results. With A-scan ultrasonography, no change in AL was observed during accommodation, whereas other studies reported an increase. Using partial coherence tomography, including the IOLMaster (Carl Zeiss Meditec, Dublin, California, USA) and Lenstar (Haag-Streit AG, Koeniz, Switzerland), AL has been found to increase during accommodation. With advances in whole eye imaging using ultra-long scan depth optical coherence tomography (UL-OCT), each compartmental dimension of the whole eye can be measured. This may provide an alternative way for revisiting axial elongation during accommodation. The repeatability of our UL-OCT method for measuring the whole biometry has been validated, and the AL results agreed well with the IOLMaster results. The goal of this study was to investigate changes in axial biometry of the whole eye during accommodation using UL-OCT.
Methods
Twenty-one healthy adult subjects (10 male and 11 female) were recruited into this study. The mean age was 27.4 ± 4.5 years (ranging from 19-35 years). The mean refractive error was −0.9 ± 1.14 diopters (ranging from 0 to −3.00 diopters). There were 10 myopic subjects with a mean refractive error of 1.95 ± 0.88 diopters and 11 emmetropic subjects. The exclusion criteria included subjects older than 35 years; those with any systemic disease or anterior or posterior segment pathology of the eye, including a history of laser treatment; and those with trauma or who had received eye surgery. Approval was obtained from the institutional review board for human research at the University of Miami. All subjects participating in this prospective study signed consent forms and were treated in accordance with the tenets of the Declaration of Helsinki.
The use of UL-OCT has been reported previously and the details of the system have been documented elsewhere. Briefly, the system included a superluminescent diode light source (SLD; InPhenix, IPSDD0808, Livermore, California, USA). The center wavelength is 840 nm with a bandwidth of 50 nm. The spectrometer consisted of a line array complementary metal–oxide–semiconductor (CMOS) camera (Basler Sprint spL4096-140k; Basler AG, Ahrensburg, Germany), a collimating lens (f = 50 mm; OZ Optics, Ottawa, Ontario, Canada), a 1800 line/mm transmission grating, and an image enlargement lens (f = 240 mm; Schneider Optics, Hauppauge, New York, USA). A switchable reference arm with 4 mirrors was used to sequentially acquire 4 images. The scan depth of each single reference arm was 12.57 mm. Overlaying the first and second reference arms for the anterior segment, the 4 switchable reference arms obtained an equivalent scan depth of 37.71 mm in air without an image gap. By imaging a mirror at different depths we obtained the point spread function (PSF). The full width at half-maximum intensity of the PSF was determined as the resolution of the OCT system in air. The measured axial resolution was 7.7 μm in the air. The resolution for ocular biometry was 5.6 μm, assuming a refractive index of 1.38.
A Badal system mounted into the sample arm provided the vision targets and accommodative stimulus during imaging as detailed elsewhere. The vision target of a white Snellen letter “E” was displayed by a light-emitting diode (LED) screen on a black background. The measured eye fixed on the vision target, and the other eye was sheltered. The OCT measuring beam was congruent to the fixation target, which ensured that biometry was conducted on the fixation/visual axis. The spherical refractive error of each subject was corrected by the Badal target system before the subject was imaged. The left eye of each subject was imaged at a baseline status of 0 D and an accommodative status of +6 D stimulus. The measurements were repeated twice for both states during a single study visit. All measurements were performed by the same researcher (Y.S.).
An automated custom software was used to reconstruct full eye images; this software has been validated against a manual method as previously reported. Refractive indices of the cornea, aqueous humor, crystalline lens, and vitreous were 1.387, 1.342, 1.408, and 1.341 at 840 nm wavelength, respectively. Structural boundary locations were detected along the central axis up to the retinal pigment epithelium (RPE). Optical path lengths of the central corneal thickness (CCT), ACD (distance between corneal endothelium and anterior lens surface), lens thickness, and vitreous length were obtained from a longitudinal reflectivity profile processed from central 190 A-scans after unusable A-scans owing to the specular reflex on the apex were removed ( Figure 1 ). The geometric lengths of every compartment were calculated based on the cited refractive indices. The locations of the anterior and posterior poles of the crystalline lens were measured as the distances from the anterior corneal surface to each of these poles.
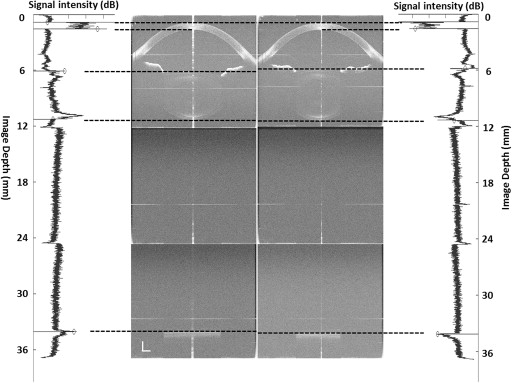
The method for whole eye biometry using our ultra-long scan depth OCT has been validated. In the published study, a model eye (OEMI-7; Ocular Instruments Inc, Bellevue, Washington, USA) was imaged with an ultra-long scan depth OCT to validate our method for OCT full eye biometry. The measurement obtained with the ultra-long scan depth OCT was highly correlated with the geometry of the model eye in all measured parameters. Repeated measurements were performed in a group of 37 subjects. The coefficients of repeatability (CoRs) of CCT, ACD, lens thickness, vitreous length, and AL were 0.021 mm, 0.067 mm, 0.071 mm, 0.084 mm, and 0.075 mm, respectively. The CoR% ranged from 0.3%–3.9% and the intraclass correlation coefficient ranged from 0.946–0.999. Measurements obtained from the UL-OCT in that study were compared with measurements obtained using the IOLMaster and a strong relation was documented.
A statistical software package (SPSS for Windows 17.0; SPSS Inc, Chicago, Illinois, USA) was used for descriptive statistics and data analysis. All data are presented as the mean ± standard deviation. Paired t tests were used to access changes in each parameter between the baseline and accommodative conditions. P < .05 was considered as significantly different between measurements.
Results
The geometric lengths of all elements of the whole eye biometry at rest and accommodative states were obtained ( Table 1 , Figure 2 ). Compared to the rest state, ACD at the accommodative state decreased significantly from 3.128 ± 0.305 mm (median 3.149) to 2.961 ± 0.298 mm (median 2.987) ( P < .001). Lens thickness increased significantly from 3.723 ± 0.237 mm (median 3.661) to 3.989 ± 0.234 mm (median 3.964). Vitreous length decreased significantly from 17.129 ± 0.864 mm (median 16.946) to 17.057 ± 0.848 mm (median 16.794) ( P < .001). AL was 24.519 ± 0.917 mm (median 24.335) at the rest state and increased to 24.545 ± 0.915 mm (median 24.353) in the +6 D accommodative stimulus. The elongated AL of 26.1 ± 13.4 μm between rest and accommodative states was significant ( P < .001). During accommodation, the anterior crystalline lens pole moved forward by 0.167 mm, which was approximately 1.7 times the backward movement of the posterior crystalline lens pole. There were significant correlations between the increase in lens thickness and the total decrease in ACD ( r = −0.72, P < .05) and vitreous length ( r = −0.64, P < .05). However, there was no correlation between the increase in lens thickness and an increase in AL ( r = −0.15, P > .05). No significant change was found in CCT during accommodation ( P > .05). Between myopic and emmetropic subjects, no significant differences were found for changes in ACD, crystalline lens thickness, vitreous length, or AL ( P > .05).
| 0 D | +6 D | P a | Δ b | |||||
|---|---|---|---|---|---|---|---|---|
| OPL | Mean | Median | OPL | Mean | Median | |||
| CCT | 0.747 ± 0.035 | 0.539 ± 0.025 | 0.538 | 0.747 ± 0.034 | 0.538 ± 0.024 | 0.538 | .416 | 0.000 ± 0.003 |
| ACD | 4.198 ± 0.409 | 3.128 ± 0.305 | 3.149 | 3.973 ± 0.400 | 2.961 ± 0.298 | 2.987 | <.001 | −0.167 ± 0.078 |
| LT | 5.242 ± 0.334 | 3.723 ± 0.237 | 3.661 | 5.616 ± 0.330 | 3.989 ± 0.234 | 3.964 | <.001 | 0.265 ± 0.104 |
| VL | 22.970 ± 1.159 | 17.129 ± 0.864 | 16.946 | 22.874 ± 1.138 | 17.057 ± 0.848 | 16.794 | <.001 | −0.072 ± 0.076 |
| AL | 33.158 ± 1.221 | 24.519 ± 0.917 | 24.335 | 33.210 ± 1.2019 | 24.545 ± 0.915 | 24.353 | <.001 | 0.026 ± 0.013 |
b Change between the baseline (0 D) and the accommodative condition (+6 D).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


