11

Pattern Scan Laser (PASCAL) Photocoagulation
Raul Velez-Montoya  Gerardo García-Aguirre
Gerardo García-Aguirre  Mark Blumenkranz
Mark Blumenkranz  Hugo Quiroz-Mercado
Hugo Quiroz-Mercado
INTRODUCTION
Thirty years ago, in the 1970s, the Diabetic Retinopathy Study (DRS) established the foundation of modern panretinal photocoagulation (PRP). The study results demonstrated a benefit from the application of laser photocoagulation to the retina by reducing the risk of severe visual loss in patients with proliferative diabetic retinopathy (PDR) by 50% (1). Later, the Early Treatment Diabetic Retinopathy Study (ETDRS) likewise showed similar results by decreasing the risk of moderate visual loss in patients with nonproliferative diabetic retinopathy from 16.3% to 6.4%, and up to 50% in patients with diabetic macular edema (2). Since then, the treatment of the retina with laser photocoagulation has become a fundamental part of the everyday medical practice for ophthalmologists and is the treatment of choice for ischemic and neovascular diseases (1–4).
Panretinal, direct focal, and macular grid photocoagulation have been the leading techniques of laser treatment to the retina for nearly 40 years. In ischemic diseases, patients usually receive 1200 to 1800 laser burns, divided into two to four sessions, each one separated at intervals of 2 to 4 weeks and with each session lasting 10 to 15 minutes. This treatment scheme has remained virtually unchanged for over 20 years (5–7).
Since solar and xenon arc lamp photocoagulation were introduced in the late 1950s by Meyer-Schwickerath as a therapeutic method in ophthalmology, the indications for their use as well as the equipment used for their administration have evolved. The first device, successfully used in clinical practice, was the xenon arc lamp (8). This light source had the disadvantage of being composed of a polychromatic wavelength. This made it very inefficient in terms of the amount of energy absorbed by the tissue, and unpredictable in terms of tissue interaction. With the next generation of laser photocoagulators, more efficient equipment appeared, providing monochromatic wavelength (e.g., the ruby, argon, and krypton laser). Since then, advances in technology have focused on designing devices more convenient for the physician than for the patient. The emergence of systems with air-cooling modules and the advent of fiber optic allowed companies to significantly reduce the size of the equipment. However, despite the fact that today we have more compact and efficient solid-state lasers, the way in which the treatment is administered remains unchanged since the mid-1970s (8). In short, the procedure is tedious for the patient and physician and varies between uncomfortable and painful for the patient, occasionally requiring the administration of retrobulbar anesthetic injections.
LASER MECHANISM OF ACTION
The mechanism through which the laser exerts its therapeutic action is still undetermined. When photocoagulation was applied for the first time in PDR, it was thought that the mechanism of action was the photocoagulation and destruction of areas of neovascularization, thus preventing vitreous hemorrhage and tractional retinal detachment. In later observations, it was clear that the clinical effect of photocoagulation seemed to depend more on applying laser to healthy retina than to areas of neovascularization (9).
The first theory about the effectiveness of laser was based on the initial observation by Michaelson, in which he proposed the existence of a “growth factor X,” responsible for neovascularization of the hypoxic retina (10). Patz et al. took up this idea and suggested that the therapeutic effect of laser was exercised by destroying sick and hypoxic tissue. In such a way, the source of production of the “factor X” was destroyed and hence its intraocular levels were lowered. The problem with this theory was, in the case of PDR, that hypoxia is located in the inner retinal layers, being primarily a problem of microangiopathy and occlusion of the microcirculation, while the outer retinal layers remain apparently normal. Histological evidence demonstrated that mild to moderate laser burns destroy the outer layers and the retinal pigment epithelium (RPE), leaving the inner layers intact, which was the alleged source of the “growth factor X” (9, 10).
Another theory, based on histological observations, stated that laser destruction of RPE cells produces the release of protective factors that slows the process of neovascularization. This theory was reinforced by Mori et al., who demonstrated the protection that a platelet-derived factor produced on delaying neovascularization of the retina and choroid. However, a direct relationship between the application of laser and the release of these factors has never been proven conclusively (9, 11).
The oxygen theory suggested that photocoagulation destroys the photoreceptors, decreasing oxygen demands of the outer retina and improving oxygenation of the inner layers. This, in turn, decreases the production of growth factors and alters retinal hemodynamics. The acceptance of this theory is based in the fact that it explains the effect of laser not only in proliferative diseases but also on macular edema (9, 12).
Under normal circumstances, oxygen and nutrients diffuse through the choriocapillaris to the retina to be consumed by photoreceptors, which have a high number of mitochondria and hence a high level of oxygen consumption. After a laser burn, the photoreceptor layer is replaced by glial tissue. This tissue then has fewer mitochondria and therefore lowers the overall oxygen demand (9, 12). This means that laser injuries serve as “windows” in the outer retina, where oxygen consumption is low and may spread further to the inner retina, increasing the oxygen tension in this space (Fig. 11-1). Besides lowering the total consumption of oxygen, laser photocoagulation also decreases the production of growth factors (e.g., vascular endothelial growth factor [VEGF]) by destroying hypoxic tissue. Aiello et al. and Agustin et al. published that levels of VEGF were significantly elevated in the vitreous of patients with PDR and that these decreased significantly after laser treatment (13).
An important proof that laser treatment improves the effects of hypoxia in the retina is the observed changes in the vascular diameter. Under conditions of low oxygen tension, the arterioles are dilated, which increases local blood flow and thus the hydrostatic pressure within capillaries. When oxygen levels return to normal, the arterioles contract. This in turn decreases the blood flow and the hydrostatic pressure. In the ETDRS, fundus photographs were taken of all patients before and after the laser treatment. The analysis of this material proved that blood vessel caliber decreased 10% to 15% after laser treatment with xenon arc or argon laser photocoagulation (9, 14).
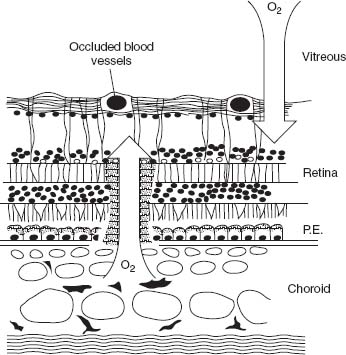
Figure 11-1. Laser burns serve as a “window” for oxygen diffusion, allowing the molecule to pass directly from the choroid to the inner retina and vitreous, without being consumed by highly active cells like retinal pigment epithelium cells and photoreceptors. (Reprinted with permission from Blackwell Publishing. Stefánsson E. Acta Ophthalmol. Scand 2001;79:435–440.)
Vasoconstriction, secondary to the increased oxygen tension, in turn, reduces the distention of capillary endothelial cells, which can reduce their growth potential. This is due to a decreased captation of thymidine and VEGF by the endothelial cell, limiting cell proliferation. In summary, laser diminishes neovascularization through two pathways: (a) by decreasing the production of VEGF secondary to hypoxia and (b) through contraction of capillaries, which limits their growth potential (12).
In the case of macular edema, laser therapy produces a therapeutic effect through two different mechanisms. Arteriolar vasoconstriction decreases the hydrostatic pressure in capillaries and venules, while the oncotic pressure remains constant. According to Starling’s law, this reverses the flow of fluid toward the capillary lumen. Alternatively, the correction of hypoxia reduces VEGF production by retinal tissues, reducing capillary permeability (15).
PATTERN SCAN LASER PHOTOCOAGULATOR PRINCIPLES
The first attempt to make photocoagulation a completely automated procedure involved a series of complex robotic equipment, image recognition software, and special tracking devices (16). This allowed the production of retinal burns in real time, based on laser shots of 100 ms duration. These systems had the disadvantage of requiring a previous retinal image as well as a shot calibration system, to establish the proper depth of the lesion (17). The complexity of these systems prevented their commercial introduction (16–18).
The pattern scan laser photocoagulator (PASCAL, OptiMedica Corp. Santa Clara, CA) is a semiautomatic system based on a frequency-doubled Nd:YAG laser, with a wavelength of 532 nm, which can apply from a single burn (single or continuously), up to 56 burns per second, with depression of the foot pedal (Figs. 11-2 and Figs. 11-3) (8). The system has an automated table, which allows height adjustment; the slit lamp has magnification factors of 7x, 12x, and 20x. The control of laser parameters is performed by means of a touch screen graphic user interface, facilitating navigation among the different patterns of photocoagulation. Control of power can be achieved in two ways: through the display or through a small knob that is located at the side of the ocular arm of the slit lamp. This control allows changes in 50 mW steps, allowing the adjustment of the intensity of the shots without the need to shift the clinician’s gaze from the patient to the display. The laser is activated by depressing the foot pedal. It is necessary to keep the pedal depressed until the entire pattern is completed, in a manner similar to the way in which an excimer laser is activated. If a complication occurs, the physician can deactivate the foot pedal, and the laser will stop. The application of the laser is made via contact lens for laser treatment (Figs. 11-4A to D and 11-5).
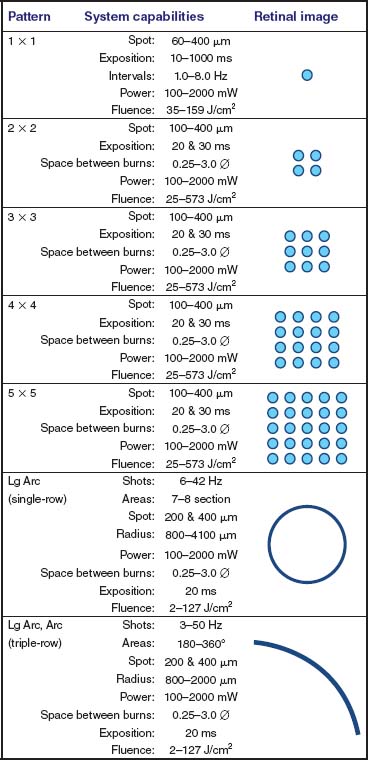
Figure 11-2. Peripheral patterns: system capabilities. For most treatments, we use a combination of patterns and parameters, as reported in Figure 11-1. The figure also shows a representation of the patterns imaged on the retina. The arc pattern is formed by a triple row of laser shots.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


