Chapter 67 Parathyroid Management in the MEN Syndromes
Primary hyperparathyroidism (HPT) is most commonly a sporadic disease, but it can be part of a familial syndrome in a small subset of patients. These inherited disorders include multiple endocrine neoplasia (MEN) type 1, MEN 2A, hyperparathyroid-jaw tumor syndrome, familial isolated hyperparathyroidism, and benign familial hypocalciuric hypercalcemia (see Chapter 58, Principles in Surgical Management of Primary Hyperparathyroidism). This chapter focuses on the surgical management of HPT in patients with MEN 1 and MEN 2A.
Multiple Endocrine Neoplasia Type 1
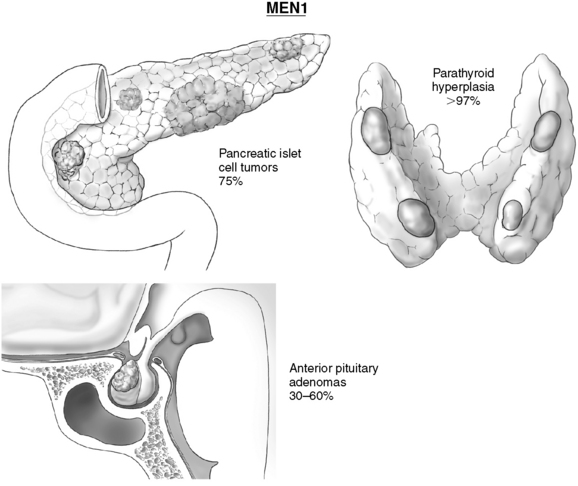
MEN 1 is an autosomal dominant disease with a prevalence of 2 to 3 persons per 100,000; it is equally common among men and women.1,2 MEN 1 is clinically defined as the presence of any two of the three most common tumors: primary HPT, pituitary tumors, and pancreatic endocrine (neuroendocrine) tumors of the pancreas (Figure 67-1). Other MEN 1–associated tumors include facial angiofibromas, lipomas, carcinoid tumors, thyroid neoplasms, adrenocortical adenomas, and pheochromocytomas.3,4 Familial MEN 1 is defined clinically as an index case of MEN 1 and at least one first-degree relative with tumor in one or more of these sites.
Genetic Testing in MEN 1
More than 400 unique germline MEN 1 mutations have been identified and have been found in 70% to 90% of families with MEN 1.1,5 Despite this diversity in mutations, studies of the MEN 1 germline mutation have supported the theory that the MEN 1 trait arises from the same chromosomal locus and, therefore, from the same gene.3 This heterogeneity of mutations has resulted in the lack of a population-level genotype-phenotype correlation (as opposed to, for example, MEN 2A) between a specific MEN 1 mutation and the development and biologic behavior of tumors, as well as between the clinical features of disease, such as age or sites of tumor development.1,4,6 A review of the literature of MEN 1 kindreds has demonstrated that different families with the same clinical manifestations have had diversity in mutations and, likewise, that different families with different clinical manifestations have been found to have the same MEN 1 mutation.4,7–9
However, the specific mutation type and location may be associated with the expression of a constant phenotype within an individual family of MEN 1 patients.4 Genetic testing allows patients with known mutations to be followed prospectively, allowing for earlier surgical intervention and potentially decreased morbidity and mortality. Furthermore, patients with a negative genetic test can be spared annual clinical testing and are assured that there is no risk of passing a mutation on to their children.1,4 The most recent consensus guidelines recommend MEN 1 germline mutation testing for any patient with a sporadic case of HPT with two or more MEN 1–related tumors or in a patient with a clinical history suspicious of MEN 1, such as the presence of multiple parathyroid tumors before age 30, recurrent HPT, or multiple islet cell tumors (functional or nonfunctional) at any age.3 MEN 1 germline testing should also be considered in patients with multigland HPT and endocrine tumors not commonly associated with MEN 1, such as pheochromocytoma or other adrenal cortical tumors, as MEN 1 germline mutations have been detected in patients with atypical endocrine neoplasia.10
Periodic screening for MEN 1–associated endocrine tumors in MEN 1 carriers is likely to help improve patient management. Age-related penetrance for all features is near zero in patients younger than 5 years of age, greater than 50% by age 20, and above 95% by age 40.3 Biochemical screening is recommended annually; for HPT, this includes serum calcium and intact parathyroid hormone (PTH) levels beginning by age 8.
MEN 1–Associated Hyperparathyroidism
Primary HPT is a disease of increased serum calcium levels associated with the inappropriate elevation of intact PTH. MEN 1–associated HPT accounts for 2% to 4% of all cases of primary HPT and characteristically is a multiglandular disease with the clonal, asymmetrical enlargement of all parathyroid glands.2,3,11,12 The typical age of onset of MEN 1–associated HPT is in the third or fourth decade and is often the first manifestation of disease; importantly, there is near complete penetrance of HPT by age 50.3,13,14 MEN 1–associated HPT occurs approximately 30 years earlier than in patients with sporadic primary HPT.
Symptoms in patients with MEN 1–associated HPT are similar to those of sporadic primary HPT, with the presence of nephrolithiasis, decrease in bone mineral density, polyuria, polydipsia, and neurocognitive symptoms of fatigue, muscle weakness, depression and mood changes, muscular aches and pains, and difficulty with memory and concentration. Patients with MEN 1–associated HPT may have a long asymptomatic phase but often have more aggressive disease; gene carriers have a detectable decrease in bone mass density by age 35.2,13,15 MEN 1 can cause simultaneous HPT and Zollinger-Ellison syndrome (gastrinoma), and hypercalcemia increases the secretion of gastrin from gastrin secreting tumors of the pancreas and duodenum.
Given the two-hit etiology of MEN 1–associated HPT, parathyroid gland involvement is often asynchronous and asymmetrical; macroscopically normal parathyroid glands have been reported in 12% to 55% of patients at the time of initial surgery, particularly in younger patients.2,5,13 It has been postulated that the prospect of having simultaneous second hits in each of the parathyroid glands is extremely low, but that the probability that the second hit would occur in a parathyroid gland increases with time; therefore, the likelihood of having any normal parathyroid gland decreases with age.16 Supernumerary (more than four) and ectopic parathyroid glands (up to 20%) have also been reported and may be found intrathymic, intrathyroidal, or in the anterior mediastinum; this may be related to the presence of parathyroid rests (embryonic parathyroid remnants) that are chronically stimulated.5,13 Malignant progression of MEN 1–associated HPT (parathyroid carcinoma) has not been reported.
Indications for Surgery
Indications for parathyroidectomy are no different in patients with MEN 1 than for those with sporadic primary HPT.3,5,11,12,17 Patients with symptomatic HPT should proceed to surgery. Specifically, patients with hypergastrinemia due to Zollinger-Ellison syndrome (even if asymptomatic) and biochemically confirmed HPT should undergo parathyroidectomy as successful surgery may markedly reduce gastrin secretion.18–20 Recent guidelines for the management of patients with asymptomatic primary HPT recommend surgery for patients with (1) serum calcium levels > 1 mg/dL above reference range, (2) glomerular filtration rate < 60 mL/min, (3) bone density findings consistent with a T score of -2.5 or worse at any site or a previous fragility fracture, and (4) age < 50 years.17
For patients with MEN 1–associated HPT, it is unclear if earlier surgical treatment reduces morbidity or mortality, particularly in those patients who are asymptomatic or minimally symptomatic. Early parathyroidectomy, especially in younger patients, would decrease the long-term effects of HPT, particularly with reference to bone loss; bone density evaluation should therefore play an important role in the planning and timing of surgery.11,13,15,21 Earlier intervention also may improve the symptoms of other concomitant endocrinopathies, such as gastrinoma.13 However, postponing surgery may facilitate the initial procedure by increasing the likelihood of finding parathyroid glands as they may be larger and easier to locate, including supernumerary/ectopic glands. Delayed intervention may also decrease the likelihood of persistent/recurrent disease and the need for future remedial surgery.13
Surgical Treatment of MEN 1–Associated HPT
The early and late outcomes of surgical treatment of MEN 1–associated HPT are inferior to those for sporadic HPT, because of the development of hyperplasia in either parathyroid glands that appeared macroscopically normal at initial surgery (and were left in situ) or in neoplastic remnants, which over time hypertrophy.13 The goals of initial surgery are to (1) successfully correct hypercalcemia with minimal risk of persistent or recurrent HPT, (2) avoid permanent hypoparathyroidism, and (3) facilitate the anticipated future surgical treatment of recurrent HPT. The initial surgery of choice for patients with MEN 1–associated HPT is subtotal parathyroidectomy with transcervical thymectomy or total parathyroidectomy with heterotopic autotransplantation of resected parathyroid tissue. Although some have advocated for the removal of only macroscopically enlarged glands (less than subtotal parathyroidectomy), this approach has shown to be associated with significantly higher rates of persistent/recurrent disease.20,22–25
Less Than Subtotal Parathyroidectomy
Less than subtotal parathyroidectomy is the removal of ≤ 2.5 parathyroid glands, leaving 1.5 presumably unaffected glands in situ and marked with titanium clips or nonabsorbable stitches to facilitate identification at a subsequent operation. Less than subtotal resection has been associated with an unacceptably high frequency of persistent or recurrent HPT. A study of 26 MEN 1 patients treated by less than subtotal parathyroidectomy reported persistent and recurrent HPT rates of 35% and 61%, respectively.22 These findings have been supported by subsequent studies demonstrating similarly high rates of persistent or recurrent disease. Dotzenrath et al. found a recurrence rate of 23% in 13 patients who underwent a limited resection, compared to 12% in 25 patients who underwent subtotal parathyroidectomy (p = 0.04).26 In a review of 79 patients by Arnalsteen and colleagues, the rate of reoperation in patients who underwent resection of only grossly enlarged glands at initial parathyroidectomy was 30%, compared to 7% in patients who underwent initial subtotal parathyroidectomy (p = 0.02).25
In a review of 92 patients with MEN 1 who underwent initial parathyroidectomy, 14% had less than subtotal parathyroidectomy; these patients had a shorter median recurrence-free survival rate compared to those patients who underwent either subtotal or total parathyroidectomy (7 versus 16.5 years; p = 0.03). The actuarial 1-, 5-, and 10-year recurrence-free survival was 92%, 69%, and 37%, respectively, for patients after less than subtotal parathyroidectomy and 100%, 80%, and 61%, respectively, after subtotal or total parathyroidectomy.23
Subtotal Parathyroidectomy
Subtotal parathyroidectomy is the removal of 3 to 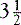 parathyroid glands, and remains the preferred operative approach at the time of initial surgery in patients with MEN 1–associated HPT.11,12,23,25,27,28 The goal of subtotal parathyroidectomy is to achieve long-term eucalcemia while minimizing the risks of permanent hypoparathyroidism. In one of the largest studies comparing the surgical approaches in 174 patients with MEN 1–associated HPT over three time periods, the rate of postoperative hypercalcemia decreased (47%, 15%, 19%; p < 0.0001) as the number of subtotal parathyroidectomies increased (25%, 59%, 51%; p = 0.0004); the increased rate of hypocalcemia (5%, 15%, 15%) was not significant.29
parathyroid glands, and remains the preferred operative approach at the time of initial surgery in patients with MEN 1–associated HPT.11,12,23,25,27,28 The goal of subtotal parathyroidectomy is to achieve long-term eucalcemia while minimizing the risks of permanent hypoparathyroidism. In one of the largest studies comparing the surgical approaches in 174 patients with MEN 1–associated HPT over three time periods, the rate of postoperative hypercalcemia decreased (47%, 15%, 19%; p < 0.0001) as the number of subtotal parathyroidectomies increased (25%, 59%, 51%; p = 0.0004); the increased rate of hypocalcemia (5%, 15%, 15%) was not significant.29
Elaraj and colleagues demonstrated that in a cohort of 79 patients who had at least three parathyroid glands resected at initial surgery, the rate of recurrent HPT was 33% after subtotal parathyroidectomy and 23% after total parathyroidectomy. The authors advocated subtotal parathyroidectomy as the initial procedure of choice rather than total parathyroidectomy. Total parathyroidectomy was associated with significantly higher rates of severe hypoparathyroidism requiring chronic treatment with calcium and vitamin D or delayed parathyroid autotransplantation in comparison to subtotal parathyroidectomy (46% versus 26%).23
A retrospective cohort study of 37 patients with MEN 1–associated HPT by Lambert et al. from MD Anderson Cancer Center found recurrent HPT in 20 patients (65%) at a median of 4 years following initial surgery; 16 patients (75%) had fewer than three parathyroid glands resected at the time of initial surgery and the remaining 4 patients had a subtotal parathyroidectomy. Of these 20 patients, 16 underwent an additional 24 parathyroid-related operations and recurrent HPT was found in 7 patients (35%) at the time of last follow-up. Of 25 parathyroid autografts performed in 22 patients, four patients remained permanently hypoparathyroid, highlighting the importance of finding a balance between the risks of recurrent HPT and permanent hypoparathyroidism, particularly in patients with MEN 1–associated HPT, which usually develops early in life.11
Salmeron et al. studied the causes, timing, and treatment of patients with recurrent MEN 1–associated HPT in 69 patients who underwent subtotal parathyroidectomy as initial surgery. Sixty patients also underwent cervical thymectomy. After surgery, 15 patients had a single normal gland remaining and 54 patients had a 50- to 70-mg remnant of an abnormal gland remaining. Nine patients (13%) had recurrent HPT within a mean of 85 months (range, 12 to 144 months). Five recurrences occurred in a parathyroid remnant, three in a previously “normal” gland, and one recurrence was in both a remnant and a supernumerary gland. Two patients had a second recurrence resulting from a supernumerary gland. Recurrence was associated with a longer follow-up time ([115 months versus 66 months, p = 0.005] demonstrating that HPT will recur in the neck or the site of an autograft as all parathyroids are genetically abnormal) and not performing a thymectomy at the time of surgery (p = 0.003).28
Total Parathyroidectomy with Forearm Autograft
Total parathyroidectomy includes the removal of all four parathyroid glands, including any ectopic/supernumerary glands; this aggressive approach to avoid cervical recurrence of HPT must be accompanied by autologous parathyroid autotransplantation to avoid permanent hypoparathyroidism. Advocates of total parathyroidectomy for MEN 1–associated HPT cite the low rates of recurrent disease, and when recurrence does develop in a forearm autograft, it can be “debulked” under local anesthesia—much simpler than a reoperative procedure on the neck. Tonelli et al. studied 51 patients, including 45 patients who underwent initial total parathyroidectomy. At the time of last follow-up, no patients had developed cervical recurrence of disease, and five recurrences (10%) in the forearm were observed after a mean of 7 years. Permanent hypoparathyroidism occurred in 10% of patients after initial surgery.21 Similarly, Lambert et al. found no cervical recurrences in five patients undergoing total parathyroidectomy.11
Parathyroid autotransplantation can be performed immediately upon completion of total parathyroidectomy from fresh autologous tissue or days later, if PTH levels are not detectable, using cryopreserved tissue.13,30,31 Parathyroid tissue that is to be autografted should be obtained from the most normal-appearing parathyroid gland in volume, color, and texture. The parathyroid tissue should be minced into small pieces. The optimal site of autotransplantation is within the brachioradialis muscle of the nondominant forearm to prevent a potential cervical reoperation if the graft becomes hyperfunctional. In follow-up, serum PTH levels obtained from right and left basilic veins can be used to monitor function of the autotransplantation. Briefly, after incision of the fascia of the muscle, the muscle fibers are gently divided to create a small pocket into which fragments of parathyroid tissue are embedded. The pocket is then sutured with a nonabsorbable suture (we use a blue 5-0 Prolene) and marked with a metallic clip. The reported optimal number of fragments to autograft has ranged from 5 to 25.30,32,33 If autografting is performed in a delayed setting, the vials containing the cryopreserved parathyroid fragments should be placed in a 37° C water bath until the medium is thawed and subsequently washed to eliminate toxicity of the preservation fluid. Prior to autotransplantation, a piece of tissue should be submitted for frozen section to confirm parathyroid tissue.
Outcomes from parathyroid forearm autotransplantation have varied, perhaps because of the experience of the surgeons and the varied techniques employed for autografting. Autografts may take years to develop function and up to 40% of autografts may fail, resulting in permanent hypoparathyroidism11,23,33—hence the concern with total parathyroidectomy as the initial treatment for MEN 1–associated HPT. Lambert and colleagues described their experience with 25 parathyroid autografts in 22 patients; 11 autografts were performed in 10 patients who underwent subtotal parathyroidectomy, and 14 autografts were performed in 12 patients who underwent resection of four or more parathyroid glands. At the time of last follow-up, 15 (60%) autografts were functional, 1 was nonfunctional, and 6 (24%) were unable to be assessed. Despite autografting, four patients (18%) were permanently hypoparathyroid; two patients required emergency treatment of hypocalcemia and frequent outpatient visits for assessment of serum calcium levels. After autograft revision with cryopreserved parathyroid tissue, one patient remained hypoparathyroid at the time of last follow-up.11 Patients are also at risk for recurrent HPT within the forearm autograft; when this occurs we debulk the autograft under ultrasound guidance.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree