Chapter 18 Papillary Thyroid Cancer
An Overview of the Epidemiology and Initial Management of Papillary Thyroid Cancer
Introduction
This chapter summarizes the epidemiology and management of papillary thyroid cancer (PTC) and analyzes the changing incidence of PTC and the etiologic factors that may be responsible for this trend. This chapter also explores the various prognostic factors that may shape the risk for recurrent disease and thyroid cancer mortality and analyzes the initial treatment strategy of PTC, with an emphasis on the impact of therapy on long-term outcome (see Chapter 19, Papillary Thyroid Microcarcinoma).
Initial Approach to the Patient
When a diagnosis of thyroid cancer has been made, the patient’s physician and surgeon must carefully provide clear information about features of the disease and treatment, including expectations of the therapy and its potential complications. Also, patients should be encouraged to attend support groups that network with thyroid cancer patients and provide important practical information for patients who have newly diagnosed thyroid cancer (see Chapters 14, Preoperative Radiographic Mapping of Nodal Disease for Papillary Thyroid Carcinoma, and 15, Pre- and Postoperative Laryngeal Exam in Thyroid and Parathyroid Surgery).
Epidemiology
Changing Thyroid Cancer Incidence
The incidence of thyroid cancer has been increasing for decades. In fact, the incidence of thyroid cancer during this period has risen at a higher rate than any other cancer in the United States.1 A study of the Surveillance, Epidemiology, and End Results (SEER) database by Davies and Welch found that 3.6 thyroid cancers were diagnosed per 100,000 in the United States in 1973 and the incidence increased to 8.7 per 100,000 in 2002, representing a greater than 2.4-fold change.2 This trend has also been noted in many other countries across Europe, Asia, Oceania, and South America.3 The only countries to notice a decline in thyroid cancer rates in Europe were Sweden (18% reduction for both men and women), Norway (5.8% reduction for women), and Spain (25.9% reduction for women).3 Other countries studied in Europe noted an increase of 5.3% (Switzerland) to 155.6% (France).3 The greatest increase occurred in Southern Australia where the rates from 1973 to 1977 through 1998 to 2002 increased 177.8% in men and 252.2% in women.3
The incidence of PTC is also increasing in children and adolescents at a rate of 1.1% per year, according to SEER registry data.4 At the time of diagnosis, the mean age of children with thyroid cancer in the United States is 15.9 years, and the female-to-male ratio is 4 to 1,4 but it may be lower in certain tumors such as medullary thyroid cancer. A study of adolescents in England also found an increasing incidence rate of PTC from 1979 through 2003,5 when the increase occurred at an even greater rate of 3% per year across Europe.6
Although the rates of thyroid cancer are increasing, PTC remains a relatively rare tumor, representing only 2% of all cancers.7 In 2009, 37,200 new cases were diagnosed in the United States and 1630 deaths were attributed to thyroid cancer during this period.7 The disease is threefold greater in women than in men and occurs at all ages. However, its peak incidence occurs at approximately 40 to 50 years in women, as compared with 10 to 20 years later in men.7
Davies and Welch found a 2.4-fold increase in all thyroid cancers in the United States from 1973 through 2002,2 which was mainly caused by a 2.9-fold increase in PTC. Nearly half of the new cancers measured 1 cm or smaller, and 87% were 2 cm or smaller. Thus, virtually the entire increase in thyroid cancer was attributed to PTCs, whereas the incidence rates of follicular, anaplastic, and medullary cancers were stable, and there was almost no rise in tumors larger than 5 cm. Moreover, these authors found that the mortality rates from thyroid cancer were stable during the period under study. Based on the observation that up to 36% of autopsy specimens may contain occult thyroid carcinomas that were unrecognized throughout life,8 some have concluded that the rising trend in thyroid cancer could be attributed to increased detection of clinically irrelevant incidental tumors2,9 (see also Chapter 11, The Evaluation and Management of Thyroid Nodules).
Factors That May Contribute to Thyroid Cancer
Increased Diagnostic Scrutiny
Although the theory of increased diagnostic scrutiny is compelling, there is evidence that other factors may be contributing to the increasing incidence of thyroid cancer.10 Chen et al. found that the incidence is rising for all tumor sizes, including tumors 4 cm or larger and as well as distant metastases.11 Likewise, another analysis of the SEER database by Enewold et al. revealed that tumors of all stages were increasing in frequency, prompting the authors to argue that if the change in thyroid cancer was solely due to increased detection, then one would expect this trend to occur only in low-risk, smaller tumors.10
The relatively recent recognition of follicular variants of PTC has resulted in a shift to an increased proportion of PTCs rather than follicular cancers.12 However, this tumor reclassification should not affect the overall numbers of new cancers; the increase in PTCs should have reached a plateau a few years after the change in tumor classification. Furthermore, one would expect a decreasing trend in the incidence of follicular thyroid carcinoma (FTC), as more tumors are being categorized as follicular variant PTC (FVPTC). Instead, the incidence rates of FTC have been stable.2,10,11
Exposure to Radiation
Exposure to ionizing radiation during childhood has perhaps been the most extensively studied causative factor in the development of PTC and, less frequently, FTC. Factors that increase the risk for developing PTC after radiation exposure include female gender, radiation for childhood cancer (rather than benign conditions), and a family history of thyroid cancer.13 The increased use of medical diagnostic radiation, particularly computed tomography, is a potential candidate for the increased incidence of thyroid cancer14 (see Chapter 28, Chernobyl and Radiation-Induced Thyroid Cancer).
Polybrominated Diphenyl Ethers
Some have proposed a potential role of polybrominated diphenyl ethers (PBDEs) in the development of thyroid cancer.15,16 These ubiquitous flame retardants may be found in plastics, electrical appliances, televisions, computers, building supplies, foams, carpets, and upholstery.15 PBDEs and their metabolites may accumulate in human tissues and bear a striking structural similarity to thyroxine.17 These compounds have been shown to be endocrine disrupters, with thyroid and estrogen effects being the most common.15 Although there has been no direct link between an increased risk of thyroid cancer and PBDEs, their increased oncogenic potential in other tissues has made them an attractive candidate for further study in thyroid cancer.18,19
Obesity
Obesity is mentioned frequently in the discussion of etiologies for the rise in thyroid cancer incidence. Indeed, obesity and overweight have been linked to nearly one third of all U.S. cancer deaths in 2009 and is associated with an increased risk of developing numerous malignancies, including colorectal, breast, kidney, and endometrium.7 Several studies have identified a higher rate of thyroid cancer in overweight patients.20–22 A recent study of older U.S. adults found that obese adults had a nearly 40% increased risk for developing thyroid cancer when compared with normal-weight adults.23 Further studies are necessary to delineate the role of obesity in the development of thyroid cancer, particularly as the incidence of obesity continues to climb throughout Europe, North America, and Asia.24–26
Although the explicit cause for the increased rate of thyroid cancer remains elusive, increased diagnostic scrutiny does not appear to account for all of the new cases. There is little dispute that the majority of patients with PTC have favorable outcomes. It is important to bear in mind, however, that “low risk” does not mean “no risk.” Accordingly, patients deserve for their physicians to pay careful attention to the details of their disease to better counsel them about their individual risk for recurrent disease and mortality. Not all tumors are inconsequential in their impact on mortality. Albeit a small percentage of the overall population of thyroid cancer patients, 1630 people died from thyroid cancer in 2009.7 Although it may be true that a subset of patients have indolent tumors that have not progressed, some seemingly low-risk tumors have more aggressive tendencies that can result in death. It is not possible to consistently differentiate tumors that will have an indolent course from those that will ultimately lead to the patient’s demise. There are numerous variables and some tests modalities, especially computed tomography (CT)/positron emission tomography (PET) studies, that can help predict outcome of individual patients.
Causes of Papillary Thyroid Cancer
Radiation-Induced Thyroid Carcinoma
Exposure to ionizing radiation during childhood is a well-described cause of PTC, although the number of patients in whom such a history is elicited is approximately 5% to 10%.27 In the 1940s and 1950s, low doses of radiation were frequently used to treat benign conditions of the head and neck, such as tonsillitis, acne, and sinusitis. Though such treatment is no longer employed for benign disorders, the carcinogenic effect lingers as patients are at risk of developing malignancies up to 50 years after their exposure.28 Likewise, survivors of childhood malignancy treated with external radiation to the head and neck are at an increased risk of developing thyroid nodules and thyroid cancer.28
Hereditary Syndromes
The majority of thyroid cancers are sporadic; however, approximately 5% of nonmedullary thyroid cancers are hereditary.29 These hereditary cases have been divided into two groups: those tumors associated with a familial cancer syndrome, such as familial adenomatous polyposis, Gardner’s syndrome, Cowden’s disease, Carney’s complex type 1, and Werner’s syndrome, and those with thyroid tumors as the primary feature such as familial nonmedullary thyroid cancer (Chapter 29, Familial Nonmedullary Thyroid Cancer).
Familial Nonmedullary Thyroid Cancer (FNMTC)
FNMTC is most accurately defined by the presence of three or more first-degree relatives with well-differentiated thyroid cancer. In families with only two affected members, 38% actually have FNMTC; three or more affected persons in a family translates into a 96% likelihood of having a hereditary form of thyroid cancer.30 The pattern of inheritance appears to be autosomal dominant with incomplete penetrance.29 Patients with FNMTC may have a more aggressive tumor than their sporadic counterparts, as they had a shorter disease-free survival in a recent multicenter retrospective study.31 However, no studies have shown a difference in mortality from FNMTC cancer (see Chapter 29, Familial Nonmedullary Thyroid Cancer).
Familial Adenomatous Polyposis (FAP)
FAP is an autosomal dominant disorder that results from a mutation in the APC tumor suppressor gene on chromosome 5q21.32 The syndrome is characterized by the development of multiple adenomatous polyps with a high malignant potential throughout the gastrointestinal tract during early adulthood. PTC occurs in approximately 2% of patients with FAP. Young women, however, are at highest risk of developing thyroid cancer; their chance of being affected is 160 times higher than it is for women without FAP.33 These tumors are frequently associated with a cribriform growth pattern on histologic examination.34
Gardner’s Syndrome
This is a variant of FAP that is associated with numerous extracolonic manifestations including supernumerary teeth, desmoids tumors, hamartomas of the upper gastrointestinal tract, and thyroid tumors. Again, only 2% of patients will develop thyroid cancers with young women being at highest risk.29
Cowden’s Disease
This is an autosomal dominant disorder caused by a mutation in the PTEN tumor suppressor gene on chromosome 10.35 These patients are at elevated risk for tumors of the breast, colon, endometrium, and thyroid.35 Follicular adenomas and carcinomas are frequently seen in this syndrome, but PTC has also been described in this syndrome. Based on its rarity, however, it is difficult to rule out the possibility of chance occurrence of PTC in this syndrome.35
Werner’s Syndrome
Also called adult progeria, Werner’s syndrome is associated with premature aging, cataracts, muscular dystrophy, scleroderma-like skin changes, diabetes mellitus, and a high incidence of tumors. It is inherited in an autosomal recessive pattern and has been linked to mutations in the WRN gene, which appears to be involved in the response to DNA damage during replication and transcription.36 Thyroid cancers may occur at an early age. Follicular carcinoma is more commonly seen, but papillary and anaplastic cancers have been seen at high frequencies as well.29
Carney’s Complex
This is an autosomal dominant condition associated with a mutation in the PRKAR1 gene on chromosome 17.37 Patients with this syndrome have characteristic blue nevi and lentigines, schwannomas, myxomas, and endocrine tumors involving the adrenals, pituitary, testicles, and thyroid gland. Up to 75% of patients have thyroid nodules on screening ultrasonography; the majority are follicular adenomas, but these patients are also at higher risk of developing PTC or FTC.38
Factors Influencing Prognosis
Effect of Tumor Variables
Histology
Classic PTC
The diagnosis of classic PTC requires nuclear features including intranuclear inclusions and nuclear grooves. The nuclei are larger than usual and often overlap; some may appear empty and are often called “Orphan Annie eyes.” The nuclear features of PTC distinguish it from other tumors, enabling it to be diagnosed on fine-needle aspiration. In addition, the tumor architecture is characterized by branching papillae with a central fibrovascular stalk. Psammoma bodies, or “ghosts” of infarcted papillae, are virtually pathognomonic of PTC. These concentric, lamellated, calcified spheres may be seen in approximately 50% of cases. Classical PTC is typically unencapsulated with invasive and ill-defined margins. Nevertheless, this common histologic type is generally associated with an excellent prognosis (see Chapter 44, Surgical Pathology of the Thyroid Gland).
Follicular variant of PTC (FVPTC)
Though recognized as a distinct entity for many years,39 this common histologic subtype did not garner widespread acceptance into the cytopathologic nomenclature until it was described in a case series by Chem and Rosai in 1977.12 Characterized by its follicular architectural pattern, the nuclear changes are variably present throughout the tumor. Some have suggested that it is overdiagnosed,40 which may, in part, be a consequence of the lack of uniform diagnostic criteria.41 Fortunately, this distinction is an academic rather than clinical issue. The tumor’s behavior is very similar to that of classic PTC.42–44
Tall cell variant (TCV)
This variant is characterized by elongated tumor cells (height twice the width) lining the papillary structures, involving at least 50% of the tumor.45 Proper identification of this variant is critical, as it is associated with a more aggressive phenotype. TCV is more likely to present with larger tumors, lymph node and distant metastases, and extracapsular extension.46 Mortality is significantly higher than for classic PTC.46 Although it is not known why these tumors are more aggressive, it could be related to their molecular profile; as many as 80% may express BRAF (see the section on oncogenes presented later).47–49 In addition, these tumors are frequently PET positive and radioiodine refractory.50
Columnar cell variant
This very rare subtype accounts for only 0.15% to 0.2% of all PTCs.51 It may be confused with TCV as its cells have a height twice the width, but the elongated nuclei and prominent nuclear stratification distinguish it from other histologic variants.52 The encapsulated version of this tumor has a very good prognosis; however, the diffusely invasive form is very aggressive.51 Unencapsulated tumors are larger at presentation, have a high rate of extracapsular spread, have distant metastases in up to 87%, and mortality is significantly higher than it is for classic PTC.53
Diffuse sclerosing variant
This variant represents approximately 5% of all PTCs and is seen more frequently in children exposed to radiation at Chernobyl.54,55 A diffusely infiltrative tumor, it is typically bilateral with a rate of lymph node metastases approaching 70%.56 The histologic appearance of the tumor may be confused with chronic thyroiditis; it is characterized by squamous metaplasia, sclerosis, psammoma bodies, and abundant lymphatic invasion.54 Rates of distant metastases are variably reported but appear to be higher than that of classic PTC.52 In spite of this seemingly aggressive behavior, there is debate about whether long-term prognosis is worse than for classic PTC.52
Solid or trabecular variant
This tumor may be seen in adults but is more commonly associated with children after radiation exposure, particularly after the Chernobyl accident.55 Nuclear features are similar to classic PTC; the architectural pattern is predominantly or exclusively solid.54 The majority of tumors present with extrathyroidal invasion,55 and cervical node metastases are common.52 In children, this tumor has a similar survival rate as that of classic PTC.55 The prognosis is less clear for adults, as conflicting mortality rates with this tumor have been described.57,58
Insular variant
Characterized by high cellularity with scant colloid, this tumor forms well-defined nests (insulae) of monotonous round cells. Some tumors have papillary differentiation, whereas others are categorized as a variant of FTC. The mean tumor diameter at presentation is greater than 5 cm,59 cervical nodal metastases are seen in more than half of patients, and up to 70% have distant metastases.60 Viewed as an intermediate tumor between that of classic PTC and anaplastic thyroid carcinoma, it is associated with 30-year cancer-specific mortality rate of 25% (versus 8% for classic PTC).61
PTC in a Thyroglossal Duct
Thyroglossal duct remnants are commonly seen, comprising more than 75% of midline neck tumors in children and 7% in adults, yet finding a cancer within one of these cysts is rare.62 One large study reported a prevalence of cancer in 1.3% of remnants.63 PTC is the most common tumor (90%), most are less than 1 cm, and they are typically confined to the cyst.64 Because so few cases have been reported, no consensus exists about the optimal management. Most agree that a Sistrunk procedure is the best way to excise a thyroglossal duct cancer.63,64 The necessity for total thyroidectomy is controversial, however, as only 25% to 56% of patients have concomitant malignancy in the thyroid.64–66 A retrospective analysis of 18 patients treated at one institution found that 75% had lateral with or without central lymph node metastases and 56% were found to have tumor foci in the thyroid.66 Twelve patients were given radioiodine. With a median follow-up of 12 years (range 1 to 22 years), 11 patients had stimulated thyroglobulin levels available; 10 were undetectable, the other was 2 ng/mL, and all had a negative neck ultrasound.66 Other studies have reported similarly encouraging results with follow-up, regardless of the treatment64,65 (Chapter 6, Thyroglossal Duct Cysts and Ectopic Thyroid Tissue).
Tumor Size
Primary tumor size is closely associated with the outcome of PTC. A retrospective study67 of 52,173 patients with PTC found that 10-year cumulative recurrence rates increased incrementally from 5% for tumors <1 cm to 25% for tumors >8 cm (Figure 18-1) and that cancer-specific mortality rates increased incrementally from 2% for tumors <1 cm to 19% for tumors >8 cm (Figure 18-2) demonstrating that primary tumor size is closely associated with disease outcome, including both 10-year tumor recurrence and cancer-specific mortality rates68,69 and with higher rates of locoregional and distant metastases68 (see Chapter 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer).
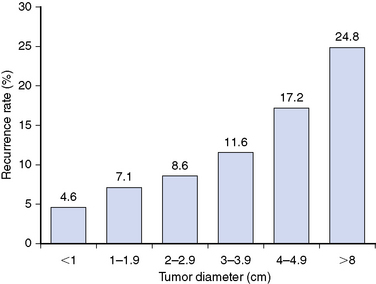
Figure 18-1 This figure is derived from data in a retrospective study by Bilimoria et al. of 52,173 patients with PTC that found 10-year cumulative recurrence rates increased incrementally with primary tumor size, going from a 5% recurrence rate among tumors < 1 cm to 25% in tumors > 8 cm (p < 0.001).67
(Data from Bilimoria KY, Bentrem DJ, Ko CY, et al: Extent of surgery affects survival for papillary thyroid cancer, Ann Surg 246:375-381, 2007.)
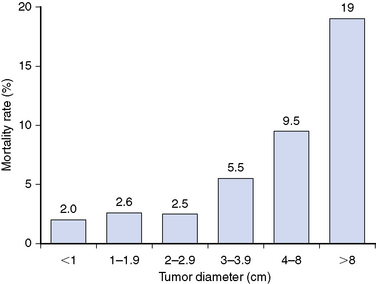
Figure 18-2 This figure, which is derived from data by Bilimoria et al.,67 demonstrates that 10-year cancer-specific mortality rates increased stepwise from 2% among PTCs < 1 cm to 19% among tumors > 8 cm (p < 0.001) for each paired comparison of tumor size.
(Data from Bilimoria KY, Bentrem DJ, Ko CY, et al: Extent of surgery affects survival for papillary thyroid cancer, Ann Surg 246:375-381, 2007.)
Tumor Multifocality
Tumor multifocality may affect prognosis. Patients with PTC in one thyroid lobe have nearly a 45% chance of cancer in the contralateral lobe.70 This is one of the reasons why recurrence rates are often higher in patients treated with hemithyroidectomy.67,71,72 One study found that tumor multifocality was associated with a higher risk of persistent or recurrent disease, even in patients treated with total thyroidectomy.73 Tumor multifocality is also found in papillary thyroid microcarcinomas (PTMC). One study of PTMC found that the only factors significantly influencing recurrence rates were the number of histologic foci (p < 0.002) and the extent of initial thyroid surgery (p < 0.01).74 Another study of PTMC found that recurrent locoregional disease was more likely in patients with cervical lymph node metastases at the time of presentation, multifocal disease, and in those not treated with remnant ablation.75
Extrathyroidal Invasion
Microscopic extension of tumor outside the thyroid bed identified by central lymph node compartment (level VI) dissection is found in as many as 30% of patients.7,77 This is associated with a higher risk of persistent or recurrent disease,73 an increased likelihood of cervical lymph node metastases,78 and reduced survival,68 as compared to patients without extracapsular spread. Macroscopic extrathyroidal PTC visualized at surgery is found in up to 9% of patients.79,80 Invasion into the surrounding musculature, esophagus, or trachea has been associated with higher recurrence rates79,80 and reduced survival,79,80 and it may benefit from external beam radiotherapy81 after aggressive surgery82,83 (see Chapters 34, Surgery for Locally Advanced Thyroid Cancer: Larynx, and 35, Surgery for Locally Advanced Thyroid Cancer: Trachea, 50, Postoperative Management of Differentiated Thyroid Cancer, 51, Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer, and 52, External Beam Radiotherapy for Thyroid Malignancy).
Lymph Node Metastases
Cervical lymph node metastases may be found at the time of initial surgery in as many as 53% of patients with PTC76; however, the incidence rates vary widely, depending on the mode of nodal detection. Prophylactic central lymph node dissections yield high rates of lymph node micrometastases (53% to 65%) (Figure 18-3),76,84 whereas macrometastases (i.e., when lymph node metastases are detected by preoperative ultrasound or during surgery) occur in a smaller but still substantial percentage, typically between 30% to 40%, of patients.80 The importance of neck ultrasonography on the management of PTC was highlighted by a study in which patients with preoperative positive lateral neck lymph nodes on ultrasound had significantly worse lymph node recurrence-free survival as compared to patients without preoperatively detectable lateral lymph node metastases.85 Patients who did not have preoperatively detected lymph node metastases on cervical ultrasonography received no benefit in terms of recurrence-free survival when prophylactic neck dissection was performed.85 Other studies have confirmed that lymph node macrometastases detected by ultrasonography are associated with lower recurrence-free survival rates for PTMC.86 The number of grossly involved lymph node metastases is inversely related to recurrence-free survival,86 whereas the impact of lymph node metastases on cancer-specific survival is less clear. Several studies have not been able to demonstrate an increase in mortality rates for patients with lymph node metastases,87–89 whereas other studies have shown reduced survival.90,91 The disparity among studies regarding the effect of lymph node metastases on mortality may be explained through an analysis of the SEER database91 in which patient age at the time of lymph node surgery was investigated; those over age 45 years with lymph node involvement had a 46% increased risk of death as compared with similarly aged patients without lymph node metastases. In contrast, there was no effect on survival in patients less than age 45 years with lymph node metastases (p < 0.001).91
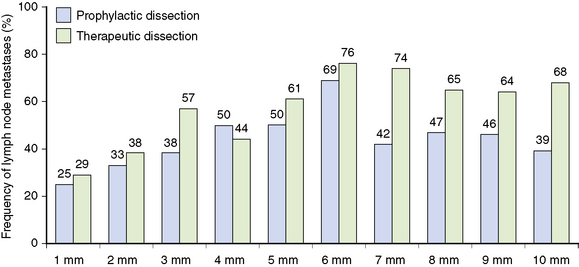
Figure 18-3 This figure is derived from data in a study by Wada et al. of patients with PTMC,84 which found that lymph node metastases were more frequent in the therapeutic dissection group as compared with the prophylactic lymph node dissection group (95.8% versus 60.9%) for the central compartment. Lymph node recurrences were thus more common in the therapeutic group than in the prophylactic group (16.7% versus 0.43%) but did not differ in the prophylactic group versus the no-dissection group (0.43% versus 0.65%).
(Data from Wada N, Duh QY, Sugino K, et al: Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection, Ann Surg 237:399-407, 2003.)
Distant Metastases
Although distant metastases are uncommon in PTC, they are present in approximately 5% of patients at the time of initial presentation, and another 2.5% to 5% will develop distant metastases after initial therapy.92 The most common sites of involvement are lung (50%) and bone (25%), followed by both lung and bone (20%) and other tumor sites (5%).92,93 The presence of distant metastases portends a poor prognosis. One study found a 50% survival rate of 3.5 years.94 However, subsets of patients have better survival rates, especially postpubertal children, those with microscopic metastases, and patients with iodine-avid tumors.93,94 In addition, the ability to achieve a negative posttreatment diagnostic whole-body radioiodine scan (RxWBS) after radioiodine therapy was associated with a 92% overall 10-year survival rate, as compared with a 19% rate for patients who did not have a negative RxWBS.93 Additional prognostic information about distant metastases may be gained by performing18 FDG-PET/CT scanning. One study found an inverse relationship between survival and degree of FDG-PET avidity of the most active lesion as well as the number of FDG-PET avid lesions.95 Patients with a positive18 FDG-PET scan had a 7.28-fold increased risk of dying from thyroid cancer as compared with patients who had a negative scan95 (see Chapters 50, Postoperative Management of Differentiated Thyroid Cancer, 51, Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer, 52, External Beam Radiotherapy for Thyroid Malignancy, and 55, Medical Treatment for Metastatic Thyroid Cancer).
Oncogenes
Oncogenes comprise an important new field of study that has been rapidly transformed into clinical studies. Analysis of these genetic aberrations has yielded information about the pathogenesis of thyroid cancer as well as providing new avenues for treating radioiodine-refractory PTC and FTC. Clinicians soon may be able to routinely utilize these molecular markers as an additional tool for diagnosing malignancy in a thyroid nodule on FNA.96
The most common oncogene in sporadic PTC is BRAF.97 Its prevalence varies with the geographic locale being sampled, but pooled analyses have found that approximately 39% of PTCs have this mutation.98 Although the clinical significance of BRAF has been debated, most studies have found that this tumor mutation is associated with adverse clinicopathologic characteristics of PTC, including rapid tumor progression and tumor recurrence in older age patients, lymph node metastases, extrathyroidal invasion, and advanced tumor stages; it also has been associated with treatment failure.99,100 The BRAF mutation has even been found with PTC recurrence in patients with what would have been otherwise regarded as low-risk tumors.97,98,101 Another study found that patients with BRAF-positive tumors have a higher overall mortality rate.101 However, several studies have not confirmed a correlation between the BRAF mutation and a worse clinical outcome.98,102 Indeed, with such a high rate of BRAF positivity (up to nearly 40%) and an overall excellent outcome for the majority of patients with PTC, not all patients with BRAF do poorly. Moreover, not all patients with aggressive tumors have the BRAF mutation, suggesting other factors play a role in determining tumor phenotype. Further study is needed to identify patients at highest risk for poor outcomes among those with a BRAF mutation.
Other mutations that have been associated with papillary thyroid carcinomas include the RET/PTC and PAX8/PPARγ rearrangements and RAS point mutations. Numerous rearrangements of the RET receptor tyrosine kinase gene have been associated with papillary thyroid carcinomas, the most common being RET/PTC1 and RET/PTC3. The prevalence of this genetic alteration is variable, depending on the study, the sensitivity of the detection methods, and geographic variability; estimates range from 20% to 50%.103,104 RET/PTC3 was the most prevalent mutation among children exposed to radiation after the Chernobyl accident.105 The clinical implications of a RET/PTC rearrangement in a tumor are unclear. There is evidence that a favorable prognosis may be found in the presence of this mutation in some cases,106,107 whereas others have found no association with patient outcomes.108,109 The RAS mutation may be found in PTC, FTCs, and follicular adenomas (FAs) with unclear prognostic implications.107 The PAX8/PPARγ fusion oncogene is found in about 36% of FTC, 11% of FAs, 13% of FVPTC, and 2% of Hürthle cell carcinomas.110 It has not been described in classic PTC. The presence of this molecular marker in follicular adenoma, a benign tumor, raises the question the role of PAX8/PPARγ in tumor develpment.111 The usefulness of this mutation as a predictor of clinical outcomes is also disputed. One study found that tumors with PAX8/PPARγ rearrangement are more likely to have multifocal capsular and vascular invasion,112 whereas others have not been able to reproduce these findings113,114 (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia).
Effect of Patient Variables on Prognosis
Age
Age at the time of tumor diagnosis is one of the more important contributing factors to prognosis; after the age of 40, recurrence and mortality rates increase significantly.68 The recurrence and mortality rates of PTC in patients beyond the age of 60 years become even more steep68 (Figure 18-4).
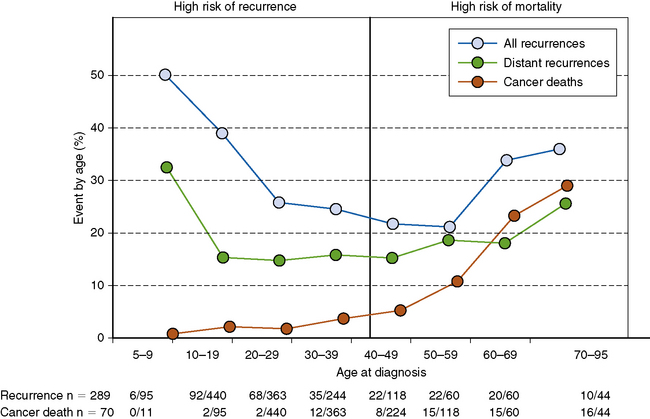
Figure 18-4 This figure shows that age at the time of tumor diagnosis is one of the more important contributing factors to prognosis. After age 40 years, recurrence and mortality rates increase significantly,68 and the recurrence and mortality rates of PTC in patients beyond the age of 60 years become greater.
(Data from Mazzaferri EL, Jhiang SM: Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer, Am J Med 97:418-428, 1994.)
Children and adolescents (<20 years of age) are more likely to have more advanced tumor stage at the time of diagnosis. One large study found that 64% of children had cervical lymph node metastases and 23% had distant metastases at the time of diagnosis.115 In contrast, studies of adults have found that up to 40% have lymph node metastases and only 5% present with distant metastases.68 Likewise, the pediatric recurrence rates over 20 to 30 years are nearly twice those of adults, 40% versus 20%, respectively.68 Despite the extent of disease at the time of diagnosis, children generally have excellent survival rates. One large study found a 2% cause-specific mortality rate after 40 years of follow-up.116
Most authorities suggest children with PTC should be treated with total thyroidectomy and radioiodine,117–119 although others prefer surgery alone.116 Differentiated thyroid cancer has a more aggressive presentation in prepubertal children, and rigorous initial surgical and 131I treatment followed by thyrotropin suppression has resulted in favorable outcomes120 (see Chapter 27, Pediatric Thyroid Cancer).
Autoimmune Thyroid Disease
Some patients have concomitant Hashimoto’s thyroiditis with well-differentiated thyroid cancer. Whether such lymphocytic involvement increases the risk of developing thyroid cancer remains uncertain. In a recent prospective study of patients with Hashimoto’s disease and a thyroid nodule, the risk of malignancy in that nodule was slightly reduced as compared to patients with thyroid nodules in the absence of thyroid autoantibodies.121
The prognostic impact of coexistent chronic lymphocytic thyroiditis and PTC is uncertain. A recent study of 214 patients with Hashimoto’s disease treated at one institution found these patients had smaller tumor sizes (p = 0.02) as compared to patients without the autoimmune component and were less likely to have recurrent disease, although this did not reach significance on multivariate analysis.122 A large study of patients in Japan found that recurrence-free survival was improved in the presence of autoimmune thyroid disease 35 years after the primary surgery (p < 0.0001).86 On the other hand, another recent study found no impact of autoimmune disease on the prognosis123 (see Chapter 4, Thyroiditis).
A recent study explored the relationship of high serum thyroid-stimulating hormone (TSH) levels and the development of thyroid cancer in thyroid nodules and found that the incidence of thyroid cancer increased with higher serum TSH concentrations, a relationship that remained even in the high-normal TSH ranges.124 Furthermore, the study discovered that a higher preoperative TSH in patients with thyroid cancer is associated with extrathyroidal extension of disease.125
The prognostic impact of coexistent Graves’ disease and PTC is also controversial. One study of PTC with Graves’ disease found that in addition to being larger, these tumors were more often multifocal, locally invasive, and metastatic to lymph nodes.126 Another study found that Graves’-associated PTC had a higher rate of distant metastases and greater risk for recurrent disease.127 A more recent analysis failed to support these findings.128
Delay in Therapy
A delay in the diagnosis and treatment of thyroid cancer can adversely affect outcome. One study found that a false negative FNA cytology result delayed surgery for 28 months.129 This group of patients had a higher risk of capsular and vascular invasion and was more than twice as likely to have persistent disease at the time of follow-up (hazard ratio 2.28). In another retrospective study,68 mortality rates nearly doubled (13% versus 6%, p < 0.0001) after a > 1-year delay in therapy.
Effect of Treatment Variables on Prognosis
The Historical Debate Concerning Initial Thyroid Surgery
Since the 1950s there has been considerable debate about the optimal surgery for the management of differentiated thyroid cancer, with some advocating total or near-total thyroidectomy,130–133 and others recommending lobectomy134–137 for the management for PTC; however, there now is a reasonable consensus among the current guidelines that recommend total or near-total thyroidectomy for the initial surgical management of PTC,138–141 which is largely based upon patient age, tumor stage, and histology. The optimal goal of surgery is to remove the primary tumor in its entirety and any associated locoregional disease, while avoiding damage to the parathyroid glands, the recurrent laryngeal nerve, and other tissues. The most recent debate has been focused on cervical lymph node dissection in the initial surgical management of PTC.
The Extent of Thyroid Surgery
Most authorities now recommend total or near-total thyroidectomy for adults and children with PTC ≥ 1 cm, unless there is a contraindication to the operation.67,138–140 The American Thyroid Association Management Guidelines for patients with differentiated thyroid cancer recommend total or near-total thyroidectomy for all patients with a preoperative diagnosis of thyroid cancer > 1 cm unless there are contraindications to this surgery.138 Still, lobectomy alone may be sufficient treatment for small (< 1 cm), low-risk, unifocal, intrathyroidal PTCs, without previous radiation exposure or clinically involved lymph node metastases67,138 (see Chapter 30, Principles in Thyroid Surgery).
Completion Thyroidectomy
Completion thyroidectomy is usually necessary when a diagnosis of malignancy is made after lobectomy for an indeterminate or nondiagnostic FNA. Patients who are found to have malignancy may require completion thyroidectomy to provide complete resection of multicentric disease142 and to allow radioiodine therapy when deemed necessary. Most,143,144 but not all,145 studies of PTC have found a higher rate of cancer in the opposite lobe when the tumor is multifocal, as opposed to unifocal disease in the ipsilateral lobe. The surgical risk of a two-stage total thyroidectomy is similar to that of a near-total or total thyroidectomy.146
The Extent of Lymph Node Surgery
The benefit of lymph node dissection for the management of PTC is uncertain. Some argue that the presence of lymph node metastases does not alter the prognosis and removing them has no clinical impact.147 However, a study of the SEER database of more than 9900 patients with PTC and FTC found on multivariate analysis that age > 45 years, presence of distant metastasis, large tumor size, and lymph node metastases significantly predicted a poor long-term outcome. All-cause survival at 14 years was 82% and 79% for patients with and without lymph node metastases, respectively (p < 0.05), demonstrating that all-cause mortality is adversely affected by lymph node metastases in patients ≥45 years of age.90
Although no prospective randomized trials have been performed comparing outcomes after lymph node dissection versus watchful waiting, a prospective study from Sweden with contemporaneous Norwegian controls found that mortality was significantly lower in the group that had a prophylactic central neck lymph node dissection (1.6% versus 8.4% to 11.1%).148 Moreover, several retrospective studies suggest a survival benefit from removal of metastatic lymph nodes.73,149 One study found that systematic compartment-oriented dissection of lymph node metastases significantly improved survival (p < 0.005, T1-T3) and recurrence rates (p < 0.00001, T1-T3), especially among younger patients and men with T1-T3 tumors.149 However, historical controls were used in this study. Another study found that selective unilateral central compartment dissection is associated with a higher rate of unmeasurable serum thyroglobulin levels 6 months after surgery and a greater likelihood of being disease-free as compared with historical controls.52 Still, further studies are needed to examine the full therapeutic impact of lymph node dissections.
Radioiodine Remnant Ablation (RRA)
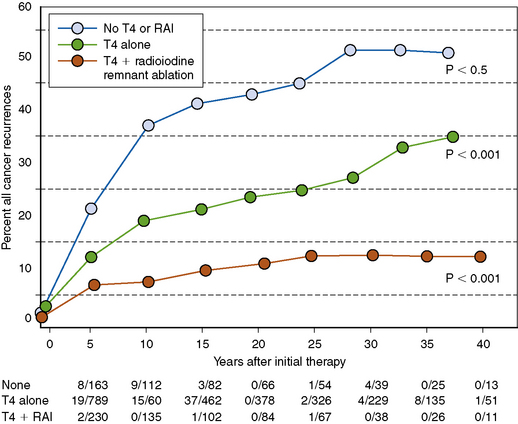
However, the impact of RRA on patient outcome has long been the subject of debate, which has mainly focused on the efficacy of RRA in reducing the rates of tumor recurrence and mortality in patients with low-risk disease. Some studies have found that RRA lowers the risk for recurrence68,150 and metastases,150 thus improving recurrence and mortality rates (Figure 18-5),68 whereas other studies have not found any advantage.151
RRA has, however, been consistently shown to benefit patients at high risk for recurrence and cancer-specific mortality, such as age ≥ 45 years with pT3 or pT4 tumors, and patients with gross extrathyroidal extension or distant metastases152 (see Chapters 50, Postoperative Management of Differentiated Thyroid Cancer, and 51, Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer).
Thyroid Hormone Suppression Therapy
Thyrotropin (TSH) is a stimulant of follicular cell growth, including that of all differentiated thyroid cancers and is intentionally suppressed with levothyroxine during follow-up. A meta-analysis found that this form of therapy reduced the likelihood of major adverse events, including disease progression, recurrence, and death.153 Still, the appropriate level and duration of TSH suppression remain unknown. A multi-institutional registry found that only patients with stages III and IV disease had improved survival after TSH suppression to less than 0.1 mIU/L, whereas patients with stage I disease derived no therapeutic advantage from TSH suppression therapy, and no effect on disease-free survival was found for any of the tumor stages.154 The recommended levels of TSH suppression are summarized in Table 18-3.152
Table 18-3 TSH Maintenance during Long-Term Follow-up
| Patients at high-risk or intermediate-risk | TSH <0.1 mIU/L |
| Low-risk patients | TSH 0.1-0.5 |
| Low-risk patients who have not had remnant ablation | TSH 0.1-0.5 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree