Purpose
To determine whether the efficacy and safety achieved with 2.0 mg intravitreal aflibercept injections (IAIs) for diabetic macular edema (DME) during the phase III VISTA DME trial were maintained with individualized, as-needed treatment.
Design
Phase IV, multicenter, open-label extension study.
Methods
Sixty patients completing VISTA DME elected to enter the ENDURANCE extension study. All patients received IAIs in the presence of clinically relevant DME. Patients were observed at 4-, 8-, or 12-week intervals depending on the need for treatment. Main outcome measures were mean IAIs given through month 12 (M12), the proportion of patients receiving no IAIs, and the role of macular laser in decreasing treatment burden among patients requiring ongoing IAIs.
Results
A mean of 4.5 IAIs were administered through M12. Eighteen (30%) patients required no IAIs, and among those who met IAI retreatment criteria, a mean of 6.0 IAIs were administered through M12. Best-corrected visual acuity gains achieved during VISTA DME were maintained and stable with individualized dosing during ENDURANCE, fluctuating by <1.5 mean letters from the baseline at all time points. Likewise, mean central retinal thickness remained relatively stable during ENDURANCE. Thirty-seven (62%) patients met macular laser criteria at a mean of 19.5 weeks with no significant difference in the frequency of IAIs before or after macular laser.
Conclusion
Vision gains achieved during the 3-year VISTA DME trial were maintained through M12 of the ENDURANCE extension study with a reduced treatment frequency, with 30% of patients receiving no IAIs. No significant reduction in IAI frequency was observed after macular laser application.
Diabetic macular edema (DME) is a major cause of vision loss, with an increasing prevalence attributable to the worldwide diabetes mellitus epidemic. Currently, 4 intravitreally administered pharmacologic agents are approved by the U.S. Food and Drug Administration for the treatment of DME. Ranibizumab (Lucentis; Genentech, South San Francisco, CA) and aflibercept (Eylea; Regeneron Pharmaceuticals, Inc, Tarrytown, NY) are both protein-based medications that block the activity of vascular endothelial growth factor A (VEGF), a key cytokine in the pathophysiology of diabetic retinopathy (DR) and DME that increases vascular permeability and stimulates angiogenesis.
VEGF blockade has proven remarkably effective in several phase III study programs at reducing DME and improving visual acuity (VA) in eyes with center-involved DME causing VA loss. In the VISTA DME (Study of Intravitreal Aflibercept Injection in Patients With Diabetic Macular Edema) and VIVID DME (Intravitreal Aflibercept Injection in Vision Impairment Due to DME) phase III trials, patients were randomized to either macular laser or 2.0 mg intravitreal aflibercept injections (IAIs) given either monthly or every other month after 5 monthly doses. IAIs led to rapid and sustained VA and anatomic improvement compared to macular laser.
After an initial period of intensive anti-VEGF therapy, several analyses of patients with DME have suggested that less frequent anti-VEGF dosing may be effective at maintaining visual and anatomic gains in most patients. For example, the RESTORE (12 Month Core Study to Assess the Efficacy and Safety of Ranibizumab (Intravitreal Injections) in Patients With Visual Impairment Due to Diabetic Macular Edema and a 24 Month Open-label Extension Study), Diabetic Retinopathy Clinical Research Network (DRCR.Net) Protocol I, and OLE (open-label extension) studies have reported that mean VA gains obtained with initial monthly ranibizumab dosing were maintained with a substantial reduction in treatment burden using protocol-specified retreatment algorithms. All such analyses have used ranibizumab treatments, and no prospective data have been reported on the use of IAIs for DME management beyond the 3-year VISTA DME phase III trial endpoint.
While anti-VEGF therapy leads to robust mean VA and anatomic improvements in DME management, many eyes demonstrate persistent DME despite aggressive therapy. For example, in the DRCR.Net Protocol T study, between 41–64% of patients met prespecified criteria for macular laser attributable to persistent DME causing VA loss. In this context, macular laser may be valuable in optimizing outcomes and reducing ongoing treatment burden. For example, through 5 years of follow-up in the DRCR.Net Protocol I study, application of macular laser at the initiation of intravitreal ranibizumab treatment appeared to lead to a reduction in retreatment frequency.
The ENDURANCE (Long-Term Efficacy and Safety of Intravitreal Aflibercept for the Treatment of DME in Subjects Who Completed the VISTA DME Trial) extension study was designed to evaluate both the ability of an individualized IAI retreatment approach to maintain the benefits achieved with IAIs during the VISTA DME trial and the ability of macular laser to decrease treatment burden among patients requiring ongoing IAIs.
Methods
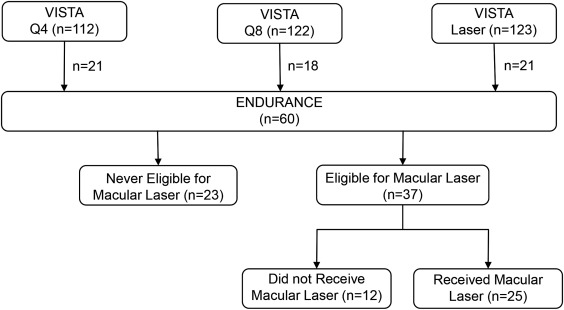
The ENDURANCE extension study is a phase IV, open-label study ( clinicaltrials.gov identifier NCT02299336 ) that assesses the need for ongoing IAIs following the 3-year, randomized, phase III VISTA DME trial. Of the 357 patients who completed VISTA DME, all of the patients at 4 major clinical sites (n = 89) were offered the opportunity to enroll in ENDURANCE. Data were collected at Retina Consultants of Houston (n = 26; Houston, Katy, and Woodlands, TX), Palmetto Retina Center (n = 16; West Columbia, SC), Northern California Retina Vitreous Associates (n = 9; Mountain View, CA), and Retina Vitreous Associates Medical Group (n = 9; Beverly Hills, CA). Prospective institutional review board (Sterling, Atlanta, GA) approval was obtained for this Health Insurance Portability and Accountability Act–compliant trial adhering to the tenets of the Declaration of Helsinki, and participants provided written informed consent.
At all visits, patients underwent Early Treatment Diabetic Retinopathy Study (ETDRS) best-corrected visual acuity (BCVA) testing at 4 m, slit-lamp and dilated ophthalmic examination, and spectral-domain optical coherence tomography (SD-OCT; Spectralis HRA.OCT, Heidelberg Engineering, Heidelberg, Germany). The SD-OCT acquisition protocol featured a volume scan (20 × 20, 49 lines, 768 A-scans per line) with 9-times image averaging.
During ENDURANCE, patients were treated with 2.0 mg IAIs as-needed in the presence of clinically relevant DME (CR-DME) determined by the investigator as DME limiting optimal visual function in the context of the current visual acuity, SD-OCT, dilated ophthalmoscopic examination findings, and fluorescein angiography (when performed).
The interval between patient visits was individualized based on disease activity. Initially, all patients were examined every 4 weeks (28 ± 7 days). If a patient received no IAIs at 3 consecutive 4-week visits, the interval between visits was lengthened to 8 weeks. At this point, if the patient received no additional IAIs at 3 consecutive 8-week visits, the interval between visits was lengthened to 12 weeks. If the patient developed recurrent CR-DME at any time, IAI was administered and the interval between visits was reduced to 4 weeks; at this time, the patient restarted the extension protocol, again being required to be evaluated without IAIs at 3 consecutive 4-week visits before extending to an 8-week visit interval.
Beginning at week 12, patients were eligible for macular laser if ≥2 IAIs were given in ≤24 weeks. After the initial macular laser treatment, patients were eligible for macular laser retreatment after 90 days if they again received ≥2 IAIs within the previous 90 days. When eligible for macular laser, patients underwent fluorescein angiography. To qualify for macular laser, a patient was required to show either microaneurysms or diffuse leakage that the investigator believed would benefit from treatment with macular laser. Patients discontinued treatment with macular laser if any of the following were present: significant macular ischemia involving the foveal avascular zone (FAZ), if treatment would be too close to the FAZ, or if the macular edema was not related to DME. Targeted laser treatment was applied to all leaking microaneurysms outside of the FAZ, and grid laser treatment was applied to areas of diffuse leakage and areas of retinal ischemia outside of the FAZ.
Prophylactic peri-IAI topical ophthalmic antibiotics were not used. The use of subconjunctival anesthesia was optional at the investigator’s discretion (ie, 2% lidocaine without epinephrine at the injection site). After topical anesthesia, the periocular skin, eyelids, and eyelashes were disinfected with 10% povidone iodine swabs, and 5% povidone iodine ophthalmic solution was applied to the ocular surface. After IAI, finger-counting testing was performed. All patients were monitored with safety assessments consisting of ocular adverse events (AEs) and serious adverse events (SAEs). The population used for safety analyses included all patients enrolled.
The accuracy of the SD-OCT automated segmentation (ie, internal limiting membrane and Bruch’s membrane) was evaluated, and segmentation errors were corrected manually before computation of change maps and change in central retinal thickness (CRT). Of the 6 patients (10%) who did not complete year 1, corresponding data are included until the date of study withdrawal. Statistical comparisons were performed with paired Student t tests or analysis of variance (ANOVA) using SAS software (version 9.1.3; SAS Inc, Cary, NC) where appropriate.
Specific efficacy analyses were evaluated by prior treatment group from the VISTA DME trial: 2 mg IAI every 4 weeks (Q4), 2 mg IAI every 8 weeks after 5 initial monthly doses (Q8), or macular laser treatment (laser). The number of patients enrolled in ENDURANCE from the original VISTA DME arms was 21, 21, and 18 for the Q4, Q8, and laser arms, respectively. Through 3 years of fixed IAI dosing, the Q4 and Q8 arms achieved similar visual and anatomic gains; therefore, outcomes during the core VISTA DME trial among these arms were pooled. Beginning at week 24 of VISTA DME, patients randomized to laser were eligible for criteria-based rescue treatment with IAIs if substantial VA was lost; data including the VA gains achieved with subsequent IAI rescue are shown for this population. Beginning at week 100 of VISTA DME, all patients randomized to laser were eligible for criteria-based IAIs.
Results
Sixty patients enrolled in the ENDURANCE extension study between November 2014 and March 2015 ( Figure 1 ). Demographics were similar to the overall VISTA DME population, with ENDURANCE patients demonstrating a BCVA gain of 10 ETDRS letters and 190-μm reduction in CRT compared to VISTA DME baseline because of the visual and anatomic benefits achieved during VISTA DME ( Table 1 ). At ENDURANCE baseline, the mean age was 65.3 years (range 43–81 years), the mean glycated hemoglobin was 7.5% (range 5.0–11.6%), the mean BCVA was 69.6 ETDRS letters (approximate Snellen equivalent 20/40), and the mean CRT was 290 μm (range 182–591 μm). Fifty-four (90%) patients completed month 12 (M12); 3 patients withdrew consent at baseline, week 8, and week 12, because of a motor vehicle accident, patient preference, and moving out of state, respectively, while 3 were lost to follow-up. Of 666 possible visits among all patients through M12, 29 (4%) were missed.
| VISTA DME (n = 459) | ENDURANCE (n = 60) | |
|---|---|---|
| Mean age, y | 62.3 | 65.3 |
| Female, n (%) | 209 (45.5) | 29 (48.3) |
| Race, n (%) | ||
| White | 384 (83.7) | 46 (76.7) |
| Black or African American | 51 (11.1) | 9 (15.0) |
| Asian | 10 (2.2) | 5 (8.3) |
| Mean HbA1c, % | 7.8 | 7.5 |
| Mean duration of diabetes, y | 17.1 | 18.6 |
| Mean BCVA, letters | 59.3 | 69.6 |
| Snellen equivalent | 20/63 | 20/40 |
| Mean CRT thickness, μm | 482 | 290 |
Visual Acuity and Anatomic Outcomes
The mean change in BCVA from the VISTA DME baseline through M12 of the ENDURANCE extension study is presented in Figure 2 . Overall, BCVA outcomes were stable during ENDURANCE, fluctuating by <1.5 mean letters at all ENDURANCE time points. Patients who required no IAIs during ENDURANCE experienced a BCVA change of +0.8 mean letters through M12. Patients who received ≥1 IAI experienced a BCVA change of +0.6 mean letters over the same time period. BCVA was similar at M12 of ENDURANCE for each of the original VISTA DME randomized arms ( P = .73). After transitioning from fixed dosing for the Q4 and Q8 arms to individualized dosing during ENDURANCE, BCVA gains achieved at the end of VISTA DME were maintained through M12 of the ENDURANCE extension study. Similarly, BCVA gains achieved in the laser arm with as-needed IAIs during year 3 of VISTA DME were maintained with individualized dosing during ENDURANCE.
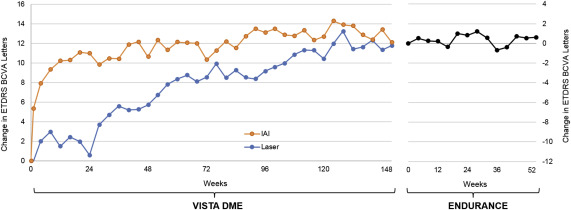
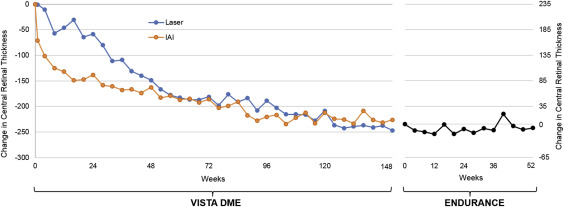
Similar to BCVA, mean CRT remained relatively stable during ENDURANCE with individualized IAI retreatment ( Figure 3 ). Gross diabetic retinopathy severity levels were similar at ENDURANCE baseline compared to M12 ( Table 2 ); however, 2 patients developed new neovascularization, progressing from nonproliferative to proliferative DR: 1 patient at week 24 after receiving no IAIs during ENDURANCE and 1 patient at week 52, 24 weeks after their last IAI.
| Diabetic Retinopathy Severity | Baseline | Month 12 |
|---|---|---|
| Mild to moderate NPDR, n (%) | 43 (72) | 37 (69) |
| Severe NPDR, n (%) | 3 (5) | 3 (6) |
| PDR, n (%) | 14 (23) | 14 (26) |
Retreatment Frequency
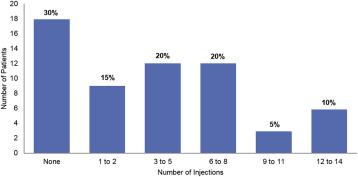
During ENDURANCE, 18 (30%) patients did not have CR-DME and therefore did not require additional IAIs to maintain the gains achieved during VISTA DME. The remaining 42 (70%) patients received ≥1 IAI during ENDURANCE, with 21 (35%) patients receiving <6 IAIs, and 6 (10%) patients receiving between 12–14 IAIs ( Figure 4 ). Overall, the weighted mean number of IAIs through M12 was 4.5 and the mean number of IAIs during ENDURANCE was similar between each of the original VISTA DME randomized arms, with no significant differences in injection frequency during ENDURANCE between patients who were originally randomized to Q4, Q8, or laser in VISTA DME ( P = .19). Among patients who met IAI retreatment criteria during ENDURANCE, the mean number of IAIs through M12 was 6.0.