Purpose
To correlate intraoperative interface fluid dynamics during Descemet stripping automated endothelial keratoplasty (DSAEK) using intraoperative optical coherence tomography (iOCT) in the Prospective Intraoperative and Perioperative Ophthalmic Imaging with Optical Coherence Tomography (PIONEER) study with postoperative outcomes.
Design
Prospective consecutive, interventional, comparative case series.
Participants
One hundred seventy-eight eyes of 173 patients undergoing DSAEK from the Cole Eye Institute, Cleveland, Ohio.
Methods
Eyes that underwent DSAEK between October 2011 and March 2014 from the PIONEER intraoperative and perioperative OCT study were included. An automated interface fluid segmentation algorithm evaluated intraoperative dynamics of interface fluid before and after surgical manipulations. iOCT images were also captured at multiple intraoperative time points for 2 different DSAEK techniques, 1 that used an active air infusion system and 1 that did not.
Main Outcome Measures
Interface fluid metrics, graft nonadherence.
Results
iOCT measurements of interface fluid after final surgical manipulations and immediately before leaving the operating room identified that total fluid volume ( P = .002), largest fluid volume pocket ( P = .002), max fluid area ( P = .006), mean fluid thickness ( P = .03), and max fluid thickness ( P = .01) significantly correlated with graft nonadherence rates within the first postoperative week. After placement and optimization of intraoperative lenticle adherence, iOCT revealed a significant difference between the area, volume, and thickness of maximum fluid pockets between the 2 surgical techniques, but both techniques resulted in significant reduction of interface fluid during the procedure.
Conclusions
Larger residual interface fluid volume, area, and thickness at the end of surgery detected with iOCT are associated with early graft nonadherence and can be quantified with an automated algorithm. iOCT imaging can successfully capture technique-dependent differences in fluid dynamics during DSAEK.
Endothelial keratoplasty, including Descemet membrane endothelial keratoplasty (DMEK) and Descemet stripping automated endothelial keratoplasty (DSAEK), has become the choice corneal transplant procedure for the management of corneal endothelial dysfunction. It boasts greater tectonic strength, more predictable refractive outcomes, and lower rejection rates compared to traditional penetrating keratoplasty (PK). However, these surgeries still carry postoperative complications, among the most concerning of which are graft dislocation and graft failure. Many factors may influence complication rates, including residual graft–host interface fluid. In response, techniques such as sweeping the recipient stromal bed and pressurizing the anterior chamber were developed to promote graft adherence. Limited studies have quantified the efficacy of these maneuvers intraoperatively or correlated perioperative interface fluid with postsurgical outcomes.
Intraoperative optical coherence tomography (iOCT) is an emerging field that can inform surgical decision-making. It presents a method with which to qualitatively and quantitatively evaluate the effects of surgical manipulations on tissues. A number of studies have described the use of iOCT for a variety of conditions and procedures in both the anterior and posterior segment. iOCT in DSAEK has shown the feasibility to identify and quantify interface fluid that is not readily apparent through the surgical microscope, and it has been used to confirm graft adherence at the end of the operation. This is the first and largest study to correlate iOCT measurements of intraoperative fluid dynamics with early graft nonadherence using automated, quantitative values. We also comment on differences in iOCT findings between 2 surgical techniques and present postoperative findings with 1 year of follow-up for eyes undergoing DSAEK in the Prospective Intraoperative and Perioperative Ophthalmic Imaging with Optical Coherence Tomography (PIONEER) study.
Methods
The PIONEER study is a prospective intraoperative and perioperative OCT study initiated at the Cleveland Clinic in October 2011 and is registered under US National Institutes of Health/ ClinicalTrials.gov ( NCT02423161 ). Briefly, it is a single-site, multisurgeon study whose focus is to examine the feasibility, utility, and safety of iOCT during anterior and posterior segment surgeries using a microscope-mounted spectral-domain OCT system. The prospective study was approved by the Institutional Review Board at the Cleveland Clinic and is adherent to the principles that were established in the Declaration of Helsinki. All patients provided informed consent. In this investigation, we evaluated 178 consecutive eyes of 173 consecutive patients from the PIONEER study that underwent DSAEK between October 2011 and March 2014.
Surgical Techniques
Two surgical techniques were used, with the primary difference between them being the use of an active air infusion system (technique B), while technique A manually introduced air into the anterior chamber for graft positioning. All technique A cases were performed by a single surgeon (W.J.D.); technique B surgeries were also performed by a single surgeon (J.M.G.). Neither patient age, sex distribution, indication for surgery, previous ocular surgery, nor ocular comorbidities significantly differed between groups. Both techniques used manual corneal sweeping to help eliminate graft–host interface fluid and encourage adherence of the donor lenticle. The differences between techniques A and B are highlighted in Table 1 .
| Technique A | Technique B | |
|---|---|---|
| Incision | Scleral tunnel | Clear cornea |
| Incision location | Temporal | Temporal |
| Tissue preparation | Precut | Surgeon cut |
| Viscoelastic use | Yes | Yes |
| Peripheral roughening | Yes | No |
| Manual air pressurization | Yes | No |
| Active air infusion system | None | Accurus a or VersaVIT b |
| Manual sweep after air infusion | Yes | Yes |
a Alcon Surgical, Fort Worth, TX.
Surgeries were performed under monitored anesthesia care using a retrobulbar block. In technique A, all donor tissue was precut by the local eye bank (Cleveland Eye Bank/Eversight, Cleveland, OH). A temporal, 5-mm scleral tunnel incision was used, and the anterior chamber was maintained with sodium hyaluronate (Healon; Abbott Medical Optics, Abbott Park, IL) during Descemet membrane stripping. The peripheral host stroma was roughened with a Terry scraper (Bauch and Lomb Storz, Bridgewater, NJ). The viscoelastic was removed with single port irrigation and aspiration. A thin bead of sodium hyaluronate was injected onto the endothelial surface of all donor tissue before folding, and noncoapting forceps were used for graft insertion. The anterior chamber was filled with filtered air, and the lenticle was unfolded and centered. The eye was brought to a high intraocular pressure with additional filtered air and left in place for approximately 10 minutes. The recipient stromal bed was manually swept using the side of a cannula to eliminate graft–host interface fluid and encourage graft adherence. Excess air was then removed from the anterior chamber until a physiological intraocular pressure was achieved. Corneal vent incisions were not created.
In technique B, the surgeon cut his own tissue in the operating room. The donor corneoscleral rims were mounted on an artificial anterior chamber (Moria S.A., Doylestown, PA) and a Moria ATLK microkeratome with a 300-μm head was used to remove an anterior corneal lenticle. The host corneal epithelium was marked with a Weck trephine (Solan Medtronic, Jacksonville, FL) and gentian violet dye, and a clear cornea temporal incision was made with a 2.75-keratome blade. The anterior chamber was filled with a cohesive viscoelastic, and a reverse-bent Price–Sinsky hook scored the host Descemet membrane, which was then stripped with a Melles stripper. The viscoelastic was removed with irrigation/aspiration. A Barron–Hessburg trephine (Katena, Denville, JN) was used to punch a posterior corneal lenticle that was folded 60/40 and inserted into the anterior chamber using Utrata forceps through an enlarged 5.2-mm temporal incision. Filtered air coupled with a bent 30-g needle on a 3-cc syringe was used to unfold and center the lenticle. An air infusion system (Accurus Surgical System [Alcon Surgical, Fort Worth, TX] or VersaVIT [Synergetics, O’Fallon, MO]) was then used to pressurize the anterior chamber (40–60 mm Hg) and promote graft adherence to the host stroma. Centripetal corneal sweeps with an irrigating cannula were performed for 1 minute under peak pressure. An air-fluid exchange was performed with the infusion system to leave the desired air fill at the end of the case. Corneal vent incisions were not created.
Intraoperative OCT Imaging
The iOCT image acquisition protocol has been previously described. In brief, a portable spectral-domain OCT system (Envisu C2200; Bioptigen, Research Triangle Park, NC) with customized microscope mount was used. iOCT images were obtained according to a standard imaging protocol and a 12- × 12-mm cube that maximally covered the graft–host interface, which was visually confirmed by the surgeon. Surgery was paused for a median time of 1.9 minutes per scan session. In technique A, the first image was taken after the graft appeared to be apposed with a full anterior chamber air fill and after initial sweeps had already been performed. In technique B, the first image was obtained after the graft was unfolded and supported with a manual injection of air but before active infusion and corneal sweeps. Additional images for both techniques were obtained after surgical manipulations based on iOCT appearance or surgeon discretion, but only the first and final image after the last surgical manipulation and before leaving the operating room were used for analysis.
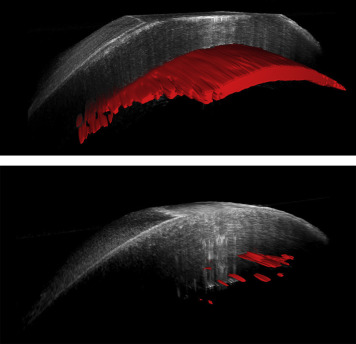
Intraoperative interface fluid was quantified postoperatively using manually validated automated analysis software, as previously described. The software calculates multiple parameters from the iOCT images related to interface fluid segmentation, including total interface fluid volume, largest isolated fluid pocket volume, largest fluid area, mean fluid thickness (ie, lenticle to host gap), and largest fluid thickness. Figure 1 provides a 3-dimensional reconstruction example of the volumetric and interface segmentation. Parameters were quantified and compared between techniques using a 2-tailed Student’s t test. Univariate analysis was used to correlate iOCT data with early graft nonadherence for all cases.
Clinical Outcomes
Clinical outcome data were collected at postoperative day 1, week 1, month 3, month 6, and up to year 1. All patients had data available for ≥1 early postoperative visit within the first week, and all but 4 had ≥1 late follow-up visit at postoperative month 6 or year 1. Outcome measures included graft nonadherence identified on clinical examination and the need for further postoperative intervention (eg, rebubble or keratoplasty). The correlation of outcome measures with patient characteristics, such as primary diagnosis, ocular comorbidities, and previous surgery, were assessed by univariate analysis using a 1-tailed Fisher’s exact test for presence of the condition.
Statistical software (JMP Pro version 10.0.2; SAS Institute Inc, Cary, NC) was used for data analysis. Significance of P values was set to < .05 with adjustments made via Bonferroni corrections for the large number of comparisons made when correlating outcomes with patient characteristics. After Bonferroni corrections, statistical significance was defined as P < .006, < .005, and < .003 when stratifying outcome correlations by primary diagnosis, previous intraocular surgery, and ocular comorbidities, respectively.
Results
Patient Demographics and Clinical Characteristics
Patient demographics are shown in Table 2 . The most common indication for surgery was Fuchs endothelial corneal dystrophy (66.9%). The most frequent ocular comorbidity was glaucoma (19.7%). The most common previous surgery, excluding cataract or lens extraction, was previous DSAEK (9.0%), vitrectomy (8.4%), or tube shunt (6.2%). Other primary diagnoses, ocular comorbidities, and previous ocular surgeries occurring with less frequency are listed in Table 2 .
| N | % | |
|---|---|---|
| Eyes | 178 | |
| Age (y) | 71.6 ± 12.5 | |
| Sex | ||
| Male | 62 | 34.8 |
| Female | 116 | 65.2 |
| Surgical method | ||
| Technique A | 105 | 59.0 |
| Technique B | 73 | 41.0 |
| Preoperative diagnosis | ||
| Fuchs dystrophy | 119 | 66.9 |
| Bullous keratopathy | 34 | 19.1 |
| Failed DSAEK | 14 | 7.9 |
| Failed PK | 6 | 3.4 |
| Other | 5 | 2.7 |
| Ocular history/comorbidity | ||
| Glaucoma | 35 | 19.7 |
| Diabetic retinopathy | 6 | 3.4 |
| Cystoid macular edema | 5 | 2.8 |
| Previous cornea ulcer | 4 | 2.2 |
| All others | <3 | |
| Previous ocular surgery | ||
| DSAEK | 16 | 9.0 |
| PPV | 15 | 8.4 |
| Tube shunt | 11 | 6.2 |
| PK | 11 | 6.2 |
| Trabeculectomy | 6 | 3.4 |
| Lens status at time of surgery | ||
| Pseudophakic | 119 | 66.9 |
| Phakic | 56 | 31.5 |
| Aphakic | 2 | 1.1 |
| Pseudophakic sutured IOL | 1 | 0.5 |
Intraoperative OCT Findings and Early Graft Nonadherence
iOCT yielded excellent visualization of the graft–host interface, and evaluation of the images showed the dynamics of interface fluid reduction over time ( Figures 1 and 2 ). iOCT showed significant reduction in fluid after surgical manipulations with both techniques ( P < .01). Final interface fluid measurements at case completion were measured for all cases. Comparative assessment of iOCT parameters between the techniques revealed that there was a trend toward decreased overall fluid volume with technique B ( P = .06). Technique B also resulted in decreased fluid parameters for largest fluid pocket volume, maximum interface fluid area, mean fluid thickness, and maximum fluid thickness at the conclusion of the case compared to technique A ( Table 3 ).




