ANATOMY
The oral cavity represents one of the most anatomically diverse regions within the head and neck. Within this relatively confined space operate highly specialized and important physiologic activities, beginning with the ability to communicate through facial animation and articulation. Deglutition originates with the maintenance of oral competence at the anterior oral cavity and then relies on subsequent lubrication, mastication, bolus formation, and then bolus transfer to the oropharynx. Similarly, within the oral cavity, sensory innervation for touch, pressure, temperature, and taste function is notable for its high degree of cortical representation. In order to achieve these critical functions, numerous tissue types are represented within the oral cavity including the mucosal lining, muscle, bone as well as unique tissues including teeth, salivary glands, and epithelium for taste. These tissues are supported by the region’s vast and redundant blood supply and innervation (
Figs. 119.1,
119.2 and
119.3).
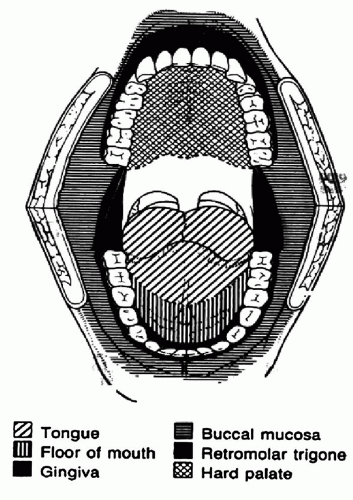
The oral cavity itself has a strict anatomic demarcation as a head and neck subsite. The boundaries of the oral cavity begin with the lips anteriorly and extend posteriorly to the vertical plane beginning at the junction of the hard and soft palate, continuing inferiorly through the circumvallate papillae at the sulcus terminalis and ending at the hyoid bone. The oral cavity is further divided into subareas including the “
lips, alveolar ridge, floor of mouth, oral tongue, hard palate, retromolar trigone, and the lateral buccal mucosa” (
Fig. 119.4). Each of these subareas has distinct anatomic features and lymphatic drainage pathways, the knowledge of which is imperative in the management of neoplasms of the oral cavity (
Table 119.1).
At the most anterior boundary of the oral cavity are the specialized myomucosal element of the “lips.” Anatomically the region of the lip begins anteriorly with the vermilion border and extends posteriorly along the red lip into the mucosal elements at the labial sulcus. The lateral demarcations of the lip are the bilateral commissures. The philtrum, or Cupid’s bow, occupies the midline superiorly and directly opposes the labial tubercle in the midline of the lower lip. The lip has a dense sensory innervation provided by the third branch of the trigeminal nerve and consists of its epithelial lining, muscle, and minor salivary glands. The blood supply is provided bilaterally by the labial arteries, which originate from the facial arterial system. The lymphatic drainage of the lower lip is primarily to the submental and submandibular triangles with subsequent drainage to the upper jugular chain. The lower lip demonstrates propensity for contralateral drainage and therefore bilateral metastasis. The upper lip does not demonstrate significant lymphatic crossover, and lesions of this region will usually metastasize to ipsilateral lymph nodes in level IB, periparotid lymph nodes, and level II jugulodigastric lymph nodes.
The “lower alveolar ridge” begins immediately behind the lower lip and its labial sulcus, and is composed of the specialized tooth-bearing bone of the mandible. The oral cavity components of the mandible include the symphysis, perisymphysis, and the bilateral mandibular bodies extending toward the ramus. The visible component of the alveolar ridge has a tightly adherent mucosal and gingival coverage that surrounds dentition where it is present. The bone of the alveolus, or the tooth-bearing component of the mandible, is characterized by thin bone surrounding the tooth with the tooth roots extending deeply into the cortical bone of the mandible. This becomes important as this thin bone can be susceptible to bone erosion by tumors, while the tooth root allows access to the deeper components of the mandible including the marrow space. The inferior aspect of the mandible is composed of dense cortical bone, which provides sufficient support, and this is the supporting bone that remains when teeth are lost and there is regression of the alveolus.
Medially, the mucosal surface of the alveolar ridge extends down the mandibular surface to the mylohyoid muscle and becomes one with the floor of mouth. Laterally, this region will extend to the gingival buccal sulcus and the insertion of the buccinator muscle. The notable tissues within the alveolar ridge include the mucosal and gingival lining as well as bone, muscle, marrow, minor salivary glands, and the teeth. The blood supply to the lower alveolar ridge is comprised of both an endosteal and a periosteal blood supply. Primary innervation is via the inferior alveolar nerve from V3, which enters the mandible at the mandibular canal within the medial ramus and exits at the mental foramen, as the mental nerve. The drainage of the alveolar ridge is dependent upon its location. Anterior lesions drain to level I and have bilateral drainage to level IB. Lateral lesions will drain to level IB as well as level II.
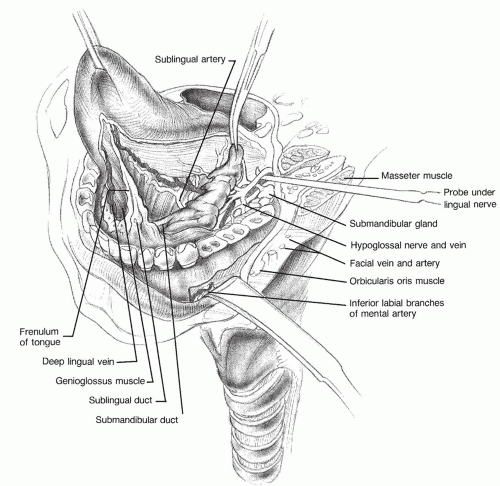
Directly within the lower alveolar ridge lies the crescentshaped region known as the “
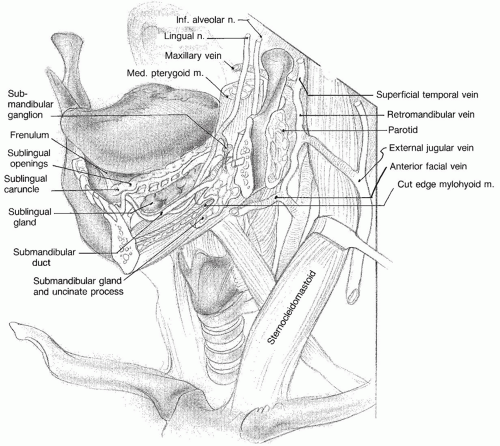
floor of mouth” (
Fig. 119.5). The floor of mouth extends from the attached gingiva of the medial alveolar ridge to the muscular tongue. Anteriorly, the region begins just posterior to the incisors and extends bilaterally to the anterior tonsillar pillars. The mucosal covering here is a very thin layer with a high degree of mobility. Within the floor of mouth are numerous salivary glands including minor salivary glands, the sublingual glands, as well as the ductal apparatus of the paired submandibular glands. All of these salivary glands have a drainage pathway within the floor of mouth, with the submandibular glands draining through Wharton duct and emptying anteriorly into the oral cavity on either side of the midline frenulum. The primary vascular supply to the floor of mouth is from the lingual artery and elements of the facial system with the primary neural supply being from the third branch of the trigeminal nerve via the lingual nerve. Anatomic injection studies demonstrate both a superficial and deep lymphatic drainage system for the oral cavity (
1). This superficial system can demonstrate bilateral drainage, whereas the deep system will drain primarily to the ipsilateral level I and upper jugular chain. Anterior floor of mouth lesions will have propensity to drain to the levels of IA and IB.
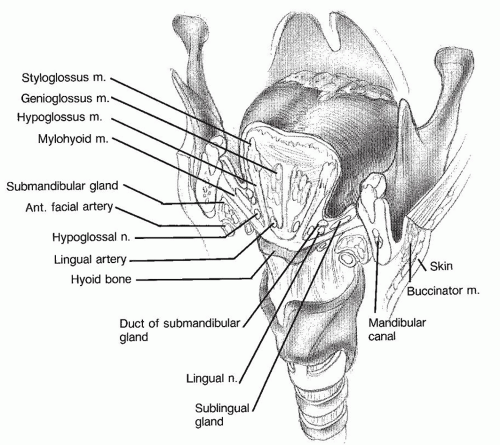
The “
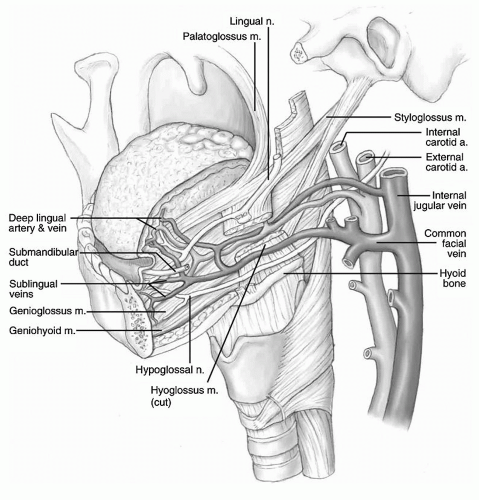
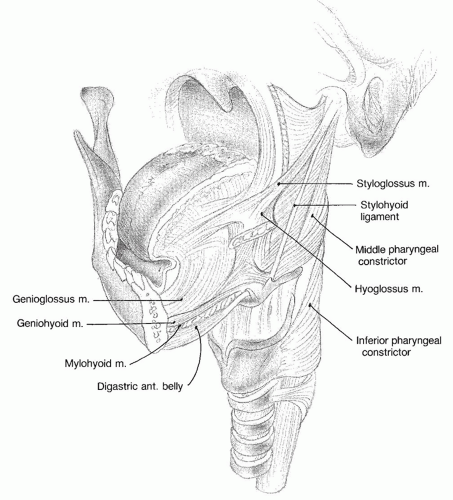
oral tongue” consists of that portion of the tongue that extends from the tip to the region of the circumvallate papillae and constitutes two-thirds of the entire tongue. The oral tongue has four distinct anatomic regions: the tip, the lateral borders, the dorsum, and the ventral surface. The oral tongue is a highly specialized muscular unit with a similarly specialized mucosal lining having a dense innervation for touch and specialized epithelium for taste. The muscular component of the oral tongue is composed of six paired muscles (
Fig. 119.6). The three extrinsic muscle groups include the paired genioglossus, hyoglossus, and styloglossus muscles that achieve the gross movements of the body of the tongue. The intrinsic musculature of the tongue includes the lingual muscles and the vertical and transverse muscular units that provide most of the tongue’s bulk. These muscles have intricate mobility and interaction critical for the functions of speech and deglutition. The motor innervation of the tongue is provided by the twelfth cranial nerve with sensory innervation from the lingual nerve via V3. Taste sensation is delivered from thousands of gustatory receptors located within the taste buds of the oral cavity through the chorda tympani and the glossopharyngeal nerve. Lesions on the tip of the tongue and midline dorsum can have a propensity for bilateral lymphatic spread as well as spread to level IA. Lateral tongue lesions will primarily spread to the ipsilateral level IB and level II. Additionally, there are anatomic and clinical series alerting practitioners to the potential of spread to levels III and IV of the neck from tongue carcinomas even in the absence of spreads to level I or level II.
Proceeding posteriorly from the upper lip, begins the “upper alveolar ridge.” In parallel to the lower alveolar ridge, this region is characterized by dense tooth-bearing bone and has similarly specialized tissues. Most anteriorly in the upper alveolar ridge is the triangular-shaped premaxilla. This portion of the maxillary alveolar ridge
contains the incisors and has its apex at the incisive foramen. Posteriorly, the upper alveolar ridge extends to the maxillary tuberosities bilaterally. Similar to the lower alveolar ridge, the mucosa and gingiva are firmly adherent to the underlying bone, conferring the same propensity for cortical erosion, and spread down tooth roots, which exit in the lower alveolar ridge. In contrast to the lower alveolar ridge, the deep bone of the upper alveolar ridge lacks the same dense cortical characteristics of the mandible and instead thins as the maxillary sinuses begin. The thin bone at this interface is less resistant and readily allows tumor spread into the sinuses. The upper alveolar ridge is innervated by the second branch of the trigeminal nerve and has a considerable blood supply from the facial artery as well as branches of the internal maxillary artery including the sphenopalatine artery. Lymphatic drainage is to level IB, periparotid lymph nodes, and level II.
Within the bony borders of the upper alveolar ridge rests the anatomic unit of the “hard palate.” The hard palate is primarily thin bone with a very tight mucosal layer overlying it on both the oral and nasal surfaces. The paired palatine processes of the maxilla extend to the level of the second posterior molar and form the bulk of the hard palate. At the most posterior edges are the paired horizontal processes of the palatine bone through which the greater palatine nerve and vessels exit via the greater palatine foramen. Although the periosteum overlying the hard palate is very dense and resistant to tumor invasion, there are important areas of potential tumor spread related to the foramen of the hard palate including the greater palatine, the lesser palatine, and the incisive foramen. In addition to its mucosal covering, there are numerous minor salivary glands throughout the hard palate region. Primary lymphatic drainage is to levels I and II.
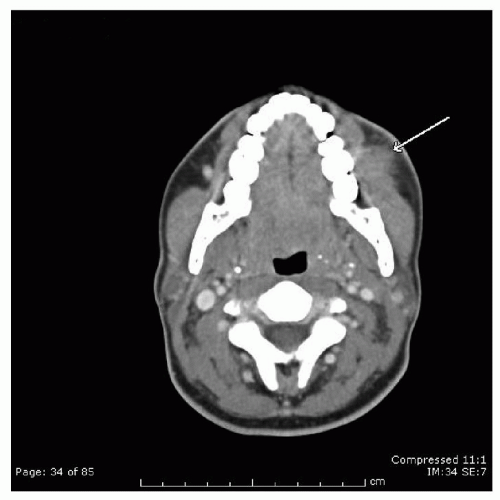
At the most posterior margin of the oral cavity is the “retromolar trigone or RMT.” This region consists of a mucosally covered triangular area that overlies the ascending ramus of the mandible. The base of this triangle rests at the last lower molar of the lower alveolar ridge and the posterior body of the mandible. The apex of the RMT is the maxillary tuberosity. The medial border is the anterior tonsillar pillar, and the lateral border is the buccal mucosa. The mucosa of the RMT is very tightly adherent to the underlying mandible and is an area prone to mandibular invasion even with small lesions. The lymphatic drainage of this area is to levels I and II, but because of its proximity to the oropharynx, a special consideration must be given to both levels IIA and IIB with RMT lesions.
The last and most lateral component of the oral cavity is represented by the “buccal mucosa.” This region extends from the RMT and the pterygomandibular raphae posteriorly to the oral commissure and lips bilaterally. The region has a mucosal lining surface that extends from the upper alveolar ridge superiorly to the inferior alveolar ridge inferiorly. Deep to the mucosal lining is the pharyngobuccal fascia and the buccinator muscle. The parotid duct pierces the buccinator and the pharyngobuccal fascia to exit at the Stensen duct lateral to the upper second molar. Tumors penetrating these anatomic layers can readily involve the laterally placed buccal fat pad and the subcutaneous as well as cutaneous structures of the cheek. Sensation to this region is provided by nerves from the second and third branch of the trigeminal system, whereas the buccinator muscle is innervated by the seventh cranial nerve. The lymphatic drainage to the buccal mucosa is quite dense and primarily drains to levels I and II but may extend to the periparotid lymph nodes as well.
EPIDEMIOLOGY
Assessment of statistics related to oral cavity cancer must done with care as often cancers of the oropharynx, a distinct subsite of the head and neck with a unique epidemiology, are included under the term “oral cancers.” True oral cavity malignancies are the 10th most common cancer worldwide in men with over 170,000 new cases per year and a mortality rate approaching 50% and the 15th most common in women with over 83,000 cases and a similar mortality rate (
2). Globally, the incidence of oral cavity cancer is 1.5 times higher in men than women (
3), whereas in the US the male:female ratio is 2:1 (
4). Certain geographic regions have a very high incidence such as Micronesia with 24 cases per 100,000 in men compared to 6.7 per 100,000 men in Western Europe (
5).
In the US, head and neck cancer—inclusive of all subsites —is the sixth most commonly occurring cancer. Review of the Surveillance, Epidemiology and End Results (SEER) data base indicates that 38% of these occur in the oral cavity, making it the most common subsite. Thirty-two percent will occur in the oral tongue and 20% in the floor of mouth, making these the most commonly affected sub areas within the oral cavity (
6). In 2011, the American Cancer Society estimated that 25,820 new cases of oral cavity cancer would occur in the US of which nearly 12,000 would
originate in the tongue (
7). Five to six thousand oral cavity cancer-related deaths occur per year (
7). Yet, the incidence and mortality rates of oral cavity cancer have been declining, albeit in small increments, over the past few decades. From 1975 to 2008, the incidence of oral cavity in the United States decreased by 1%, and the mortality declined by 1.3% (
8). This likely reflects decreases in tobacco consumption within the population. Racial disparities also exist. Although the incidence of oral cavity and oropharynx cancers is essentially equal among African American and White US males, death rate of African American males is nearly twice that of Whites (
9). Although the average age of occurrence of an oral cavity cancer is approximately 60 years and nearly 95% of patients will be over 40 years old at the time of presentation, there is concern that the incidence of tongue cancer in younger patients has been steadily increasing over the same time period (
10,
11).
The overwhelming majority, nearly 95%, of oral cavity cancers will be squamous cell carcinomas (SCCA) arising from the squamous epithelium lining the oral cavity. The next most common histology is tumors of salivary gland origin. Because of the diverse tissues within the oral cavity, a wide variety of tumors may occur in this region. Sarcomas of bone (osteosarcoma), muscle (rhabdomyosarcomas), and other soft tissues (fibrosarcomas and liposarcomas) may occur. Similarly, lymphomas, mucosal melanomas, and nerve sheath tumors may also present in the oral cavity. Tumors of dental origin, both benign and malignant, complete the differential of primary lesions within the oral cavity. Metastatic lesions, albeit rare, can also occur (
Table 119.2).
ETIOLOGY
Use of tobacco and related products, as well as alcohol consumption, are the greatest risk factors for developing the most common carcinoma of the oral cavity—SCCA. Approximately 75% of oral cavity carcinomas will be related to tobacco and alcohol use (
12). The risk of developing oral cavity cancer is correlated with tobacco use in a dose-dependent fashion. Oral cavity cancer risk increases 1.5-fold in light smokers (less than 10 cigarettes per day) and increases to a fourfold greater risk in smokers of greater than 30 cigarettes per day (
13). The risk markedly increases with a smoking history of longer than 20 years and greater than 20 cigarettes daily (
13). In Europe and South America, non-alcohol users who smoked had a threefold greater incidence of oral cavity SCCA than nonsmoking nondrinkers. In former smokers, the risk of developing cancer declines with time. After 9 years of cessation, patients had a 50% reduction of risk of cancer (
14), and patients have only a 1.5-fold increase in cancer rates after 20 years of cessation in comparison to nonsmokers (
14). In patients cured of a primary oral cancer malignancy, 40% recurred or developed a secondary head and neck primary, whereas only 6% of patients recurred after smoking cessation (
15).
Tobacco use in any form has been associated with oral cancer. Although not as strongly and directly correlated with oral cavity SCCA, smokeless tobacco (snuff) may confer as great as a fourfold increase in oral cavity carcinoma (
16). A cohort study of women in India revealed a greater than ninefold increase in the incidence of oral cavity cancer in women who used chewing tobacco greater than 10 times per day and was also related to duration of use (
17). Pipe smoking has been associated with cancers of the oral cavity, pharynx, and esophagus, with heavy drinking increasing the risk multiplicatively (
18). Pipe smoking associated with cancer of the lip is due to both the temperature and permeability of the pipe stem (
19). The lateral tongue and floor of mouth also warrant close consideration.
Similar to tobacco for the stimulant and carcinogenic effects, the areca nut is the seed of the Areca palm and when wrapped in the betel leaf is referred to as “betel nut.” In India and Southeast Asia, the betel nut is chewed with a mixture of other flavorings, cured tobacco, and lime and called “quid.” Betel nut quid has been shown to be highly carcinogenic (
20). Buccal mucosa cancers are nearly eight times more prevalent in betel nut users and in high-use regions such as India, Southeast Asia and northeast Brazil, where oral cavity cancers can account for between 25% and 50% of all cancers (
21). In a cohort study of over 10,000 participants in Taiwan, users of tobacco, alcohol, and betel nut quid had a greater than 46-fold increase in oral cavity cancers (
22). Other alternative uses of tobacco and like products also increase the risk of oral cancers. The practice of reverse smoking—placing the lit end of the cigarette in the mouth during inhalation—is associated with a 47-fold increase in rates of hard palate carcinoma among women in India (
23).
The synergistic effect of tobacco and alcohol consumption has long been recognized in the development of head and neck cancer. An estimated 75% of all head and neck cancers are caused by tobacco and alcohol combined, and those who smoke two packs per day and drink four units of alcohol per day are 35 times more likely to develop cancer compared with healthy controls (
12). Significant overlap in the consumption of tobacco and alcohol can confound epidemiologic attempts at risk stratification. Yet, studies indicate an independent role for alcohol in inducing malignancy of the oral cavity, in a dose-dependent fashion (
24,
25). Dose-response curves demonstrate a doubling in relative risk for light users of alcohol (30 g/day) compared to a nearly 10-fold increase in relative risk for heavy users of alcohol (130 g/day) (
26). The effect is consistent across types of alcoholic beverages including beer, wine, and hard alcohol (
27). No clear trend is identifiable for duration of alcohol use. Decreased risk of head and neck cancer can be achieved with alcohol cessation but requires years of abstinence, and the return to the risk level of nonalcohol users does not standardly occur until 20 years after cessation (
27).
The human papilloma virus (HPV) infects epithelial cells and has been implicated as an early carcinogenic event in head and neck cancer with the HPV-16 subtype being the most common. There has been a significant increase in HPV+ oropharyngeal cancers, which demonstrate an overall favorable prognosis when compared to traditional oropharyngeal squamous cell cancers (
28). However, HPV+ oropharyngeal SCCA is a distinct clinical entity highly selective to the oropharyngeal subsites. HPV DNA has been variably isolated from a wide range of oral cavity cancer specimens (
29). Review often indicates significant overlaps with the oropharyngeal subsites and not true oral cavity origin. The same increase in cure rates of oral cavity cancers has not yet been observed, and therefore, current treatment methods for oral cavity squamous cell cancers should not be altered. Efforts to test oral cavity cancers for HPV and related markers may be helpful in future treatment algorithms, but a clear trend needs to be established prior to altering management.
Nonsmokers and nondrinkers may also present with oral cavity cancers, and fortunately, many of these present with early-stage disease affording a favorable prognosis. A greater proportion of these patients are female, and the common sites include the lateral tongue, maxillary alveolar ridge, and buccal regions. There is also an observed trend for an increased incidence in patients under 40 years of age (
6).
Lichen planus is a chronic, inflammatory process affecting the oral cavity with a predilection for the buccal regions. Reports describe squamous cell cancers arising within areas of lichen planus, but the true malignant transformation rate is demonstrated to be less than 2% (
30). Lichen planus has an unclear etiology and often responses to topical steroids. Patients with lichen planus should be observed closely for the formation of dominant and rapidly progressive lesions more consistent with carcinoma. Such observation and the use of judicious biopsy will prevent concerning lesions from proliferating under the supposition of a benign process.
Another small but important subgroup of patients warranting close observation are those who have undergone allogenic bone marrow transplantation. A chronic inflammatory process accompanies graft versus host disease (GVHD) in these patients, and although the most common second cancers noted after transplantation are hematologic, squamous cell cancer is the most common solid tumor seen in GVHD patients (
31). All regions of the oral cavity are susceptible to GVHD and therefore warrant close observation.
PRESENTATION
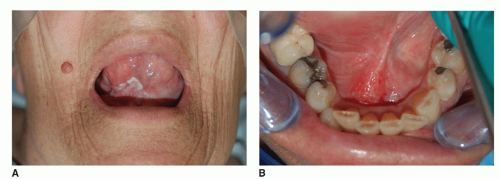
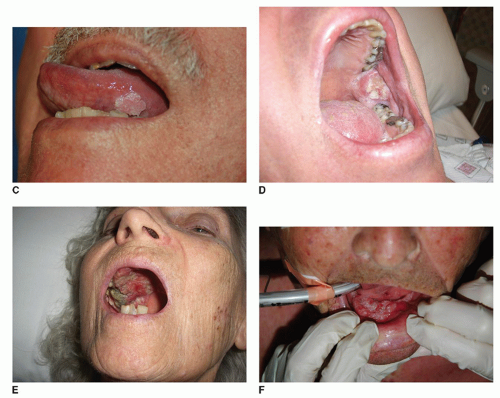
Oral cavity neoplasms present in a variety of forms (
Fig. 119.7A-F). Growths will usually present as an atypical area affecting the lining of one of the specific subsites of the oral cavity. This may be as simple as a small rough
area or may be quite extensive involving multiple subsites. Leukoplakia is a commonly used term to describe a discrete persistent white patch in the oral cavity. These areas cannot be wiped off and are usually the result of chronic irritation. The malignant potential of leukoplakia is rather small, but it is generally felt that between 5% and 15% of these lesions can harbor malignancy (
32). A baseline biopsy is not an unreasonable initial approach to the management of the leukoplakic lesion. Erythroplakia, a red plaque, is a more concerning entity. Erythroplakic lesions are felt to have a much greater potential for malignant transformation that can be five to seven times that of leukoplakia. All erythroplakic lesions warrant a biopsy for full evaluation and close follow-up if found to be nonmalignant.
Oral cavity lesions may also present as an ulcerative lesion within one of the subsites. This can be a shallow area without deep infiltration but also can manifest a significant infiltrative pattern into involved regions such as the tongue, floor of mouth, or alveolar ridges. Often, these deeply infiltrative ulcerative lesions are quite painful. At the other extreme, some lesions may be highly exophytic and present with a small base of true invasion. Likewise, an exophytic lesion can have a broad base with deep infiltration. These lesions also can present with pain.
Other manifestations of oral cavity neoplasms include bleeding without any predisposing trauma. As noted, persistent pain of the oral cavity lasting longer than 3 weeks is concerning for a neoplasm. Patients may also complain of new loose teeth in either the upper or lower alveolar ridges without any significant predisposing trauma, dental issues, or lack of hygiene. Another less common presentation is that of bad breath secondary to necrotic tumor. Oral cavity neoplasms can also present as a painless submucosal masses that can be quite extensive. Neoplasms such as this should raise concern for an etiology other than that from the squamous mucosal lining of the oral cavity. Salivary gland neoplasms would be the most common such lesions, especially in the floor
of mouth or a buccal region. Tumors originating from the mandible or maxilla, as well as from the teeth, also warrant consideration.