MS: Optic neuritis may be the initial presentation of this disorder
Age of onset: 20 to 50 years, mean age of 30 to 35 years
Gender: More common in women (Female : male ratio is 3:1)
Incidence: Between 1 and 5 per 100,000
Prevalence: Approximately 115 per 100,000
Unilateral pain or discomfort around the orbit or with eye movement is present in approximately 90% of the patients. It may precede or occur concurrently with the visual loss.
Decreased visual acuity: The degree of visual loss varies widely and is usually monocular, although a small subgroup, particularly children, often have both eyes affected simultaneously.
Decreased color vision
Other: Positive visual phenomena (photopsias), such as flashes of lights, showers of sparks, or flashing black squares, may
occur spontaneously or in response to loud noises.
Uhthoff phenomenon: Worsening of visual function with heat or exercise
Relative afferent pupillary defect (RAPD) if the process is unilateral or asymmetric.
Decreased visual acuity is the rule but need not be present.
Acquired dyschromatopsia with the color deficit often being greater than the degree of visual acuity loss.
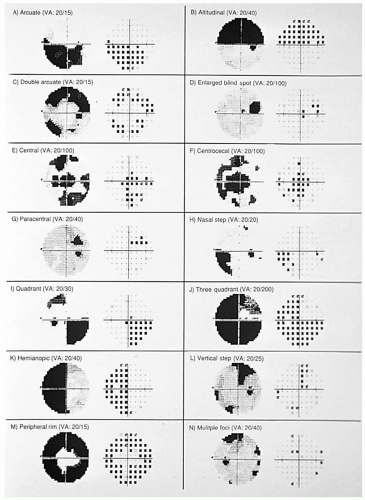
Virtually any type of optic nerve visual field defect can occur in optic neuritis (Fig. 5-1). Visual field defects are often found in the contralateral eye.
Contrast sensitivity impairment is found in virtually all patients with optic neuritis.
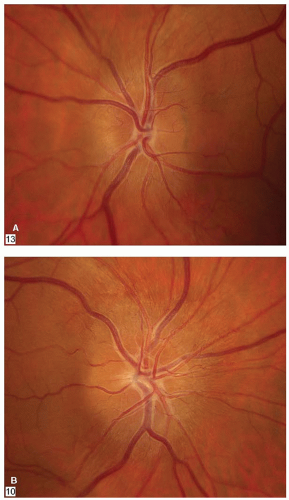
The optic disc appears normal (retrobulbar neuritis) in about two-thirds of the patients. Optic disc swelling (Fig. 5-2) will be present in 20% to 40% of the cases. The degree of swelling does not correlate with the severity of optic nerve dysfunction. Optic disc or peripapillary hemorrhages are uncommon.
Vitreous cells, particularly overlying the optic disc, may be seen but are usually minimal.
Peripheral venous sheathing occurs in some patients with demyelinating optic neuritis.
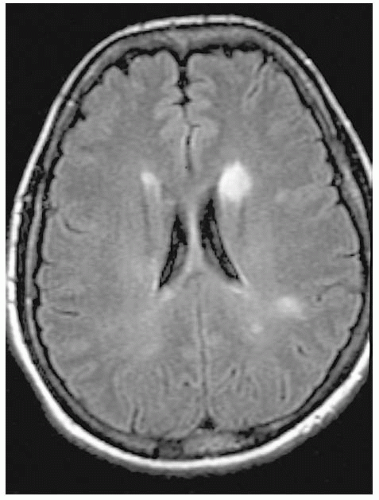
Neuroimaging: MRI is the modality of choice for investigating optic neuritis. If the patient has a typical presentation of optic neuritis, an MRI need not be performed to exclude a compressive lesion or to confirm the diagnosis (which is made on clinical grounds). An MRI can be performed to detect subclinical demyelinating plaques (Fig. 5-3) to
assist in determining the prognosis for developing MS,
evaluate patient’s potential benefit from intravenous methylprednisolone and disease-modifying therapy.
Cerebrospinal fluid (CSF) examination may be performed; however, this testing is not necessary and is being performed less frequently. The following abnormalities have been identified in patients with optic neuritis, although none is diagnostic of MS:
Pleocytosis and elevated protein
Oligoclonal bands
Myelin basic protein
Increased IgG index.
Visual-evoked responses (VER) are almost always abnormal, showing a prolonged latency on the side of the affected optic nerve.
No serologic tests or CSF studies need to be performed unless the patient’s course does not follow that of typical optic neuritis or the patient’s history or examination suggests an underlying systemic illness (Table 5-1).
TABLE 5-1. Results of Investigations of Patients in Optic Neuritis Treatment Trial | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
Fortunately, visual recovery without treatment is the rule following an episode with approximately 90% of the patients recovering to 20/40 or better vision within weeks. The Optic Neuritis Treatment Trial (ONTT) evaluated the benefits of treatment with corticosteroids. The study identified the following:
Oral prednisone
▶ showed no benefit to recovery of vision,
▶ resulted in doubling the risk of further attack of optic neuritis.
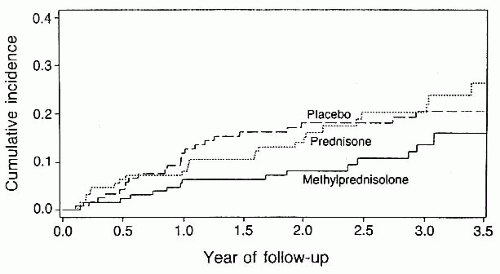
Intravenous methylprednisolone (250 mg, every 6 hours for 3 days) followed by oral prednisone (1 mg/kg/day) for 11 days, then a 3-day taper.
▶ Hastened visual recovery by 2 weeks
▶ Did not change the final extent of visual recovery
▶ Is helpful for the management of periocular pain
▶ Delayed neurologic symptoms and signs of MS for 2 years. The beneficial effect wore off after 2 years.
Initially, the visual loss may worsen over several days to 2 weeks.
Improvement initially is rapid and starts approximately 3 weeks after onset.
Recovery of vision is nearly complete by 5 weeks after onset.
Improvement may continue up to 1 year.
TABLE 5-2. Optic Neuritis Treatment Trial | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
TABLE 5-3. ONTT Baseline Data | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-4. Visual Recovery | |||||||||
|---|---|---|---|---|---|---|---|---|---|
|
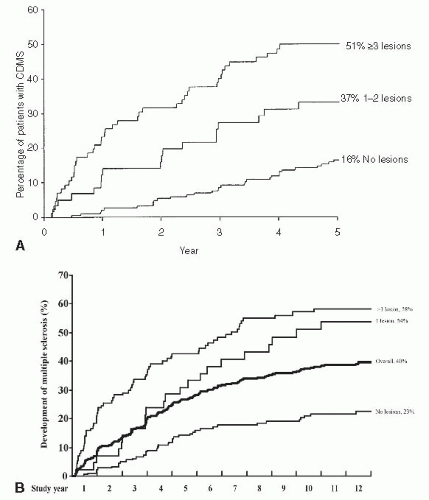
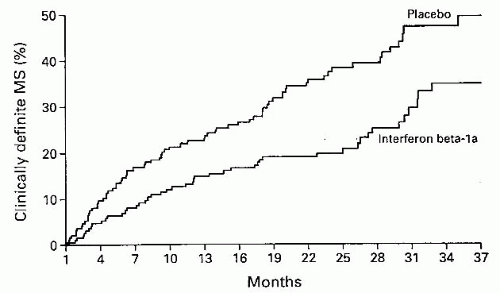
investigated the efficacy of interferon beta-1a versus placebo both on the development of CDMS and on the development or evolution of MRI lesions (Fig. 5-6). The results and conclusions of CHAMPS are shown in Table 5-7 also. Subsequent studies have shown that other disease modifying agents may be used in place of interferon beta-1a.
TABLE 5-5. Cumulative Risk of Development of Multiple Sclerosis | |||||
|---|---|---|---|---|---|
|
TABLE 5-6. Cumulative Probability of Clinically Definite Multiple Sclerosis by Treatment Group | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
TABLE 5-7. The Controlled High-Risk Subjects AVONEX Multiple Sclerosis Prevention Study (CHAMPS) | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||
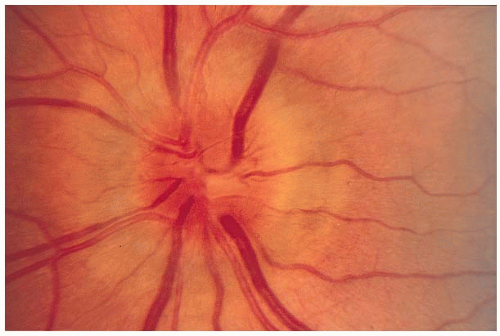 FIGURE 5-2. The optic disc is elevated and hyperemic with opacification of the retinal nerve fiber layer. The fellow optic disc was normal. |
It is thought that specific autoantibodies (NMO-IgGs) target aquaporin-4, a water channel on astrocytes within the central nervous system (CNS).
Although the exact series of pathological events is not known, the autoantibody undermines the integrity of the myelin surrounding the optic nerves and the spinal cord leading in most cases to significant visual impairment and neurological compromise.
NMO has a predilection for women and Asian individuals.
The clinical course of NMO is variable.
Clinical features of NMO include unilateral or bilateral optic neuritis, transverse myelitis, or both. The myelopathy may precede the neuropathy or follow the visual loss.
The visual impairment from optic neuritis in NMO is more often bilateral, although at times sequential, than MS on presentation,
Neuro-ophthalmic features include
pain on eye movement
central loss of vision
profound visual field defects
dyschromatopsia
retinal nerve fiber layer thinning on optical coherence tomography
Other neurological features include
decreased coordination
paraplegia or quadriplegia
sensory impairment
bladder and bowel impairment
fatal respiratory failure
The prognosis of NMO is generally poor. The cycle of remission and relapse in NMO tends generally to be more frequent and severe. Visual loss is profound and often permanent with associated paraplegia.
Treatment consists of high-dose intravenous steroids with a slow taper. Recent evidence suggests long-term immunosuppressive therapy is beneficial. Plasmapheresis may be used acutely in severe cases or with paraplegia.
If acute optic neuritis is bilateral, painless, unremitting or if the patient has a history of systemic autoimmune disease or is of Asian descent, the patient should be tested for NMO-IgG antibody titer (Table 5-8).
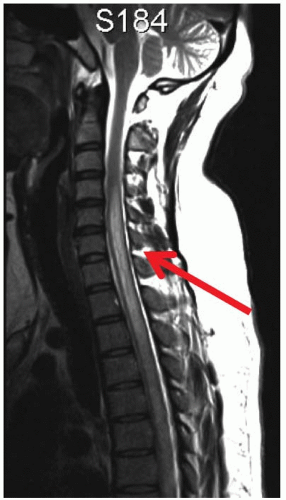
Neuroimaging: The diagnosis of NMO is usually made with an abnormal cervical spine
MRI (long T2-weighted hypersignal over more than three segments) and a brain MRI which is typically normal. The brain MRI may show T2-weighted hypersignals that are atypical for MS, for example, not involving the periventricular white matter (Fig. 5-7).
Serological tests: Patients usually have a positive NMO immunoglobulin (aquaporin 4 antibody) on serological testing.
CSF: CSF usually demonstrates a neutrophilpredominant white blood cell (WBC) count that exceeds 50 cells/mo and elevated protein. Furthermore, the CSF is free of oligoclonal bands in approximately 75% of the patients.
Serological tests: Seropositivity for NMOimmunoglobulin G (IgG) autoantibodies is highly specific for NMO.
TABLE 5-8. Diagnostic Criteria for Neuromyelitis Optica | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Acute attack: Treatment is with high-dose intravenous methylprednisolone (i.e., 1 g/day for 3-5 days) usually combined with other immunosuppressive agents (azathioprine). Refractory or aggressive disease can be treated with plasmapheresis.
Relapse prevention: Immunomodulation with long-term prednisone, azathioprine, or rituximab has been shown to be helpful.
Neuroretinitis is considered to be an infectious or immune-mediated process. Up to 50% of patients have an antecedent viral illness, usually affecting the respiratory tract, a few weeks before the onset of visual symptoms.
Neuroretinitis usually affects children and young adults although it may occur in persons of all ages.
The vast majority of patients with neuroretinitis have no cause identified or it is associated with cat-scratch disease (Bartonella henselae). Other various infections have been implicated and include syphilis, Lyme disease, and toxoplasmosis.
Many causes of optic disc swelling, e.g., papilledema and hypertensive optic neuropathy, may have retinal exudates in a stellate formation; however, the star formation is usually incomplete (Fig. 5-9; see Fig. 5-23). Therefore, the presence of disc edema and a macula star is not pathognomonic for neuroretinitis.
Painless loss of vision
Visual acuity: Ranges from 20/20 to light perception
RAPD if unilateral or asymmetric
Decreased color vision
Typical fundus picture:
Optic disc swelling: Mild to severe
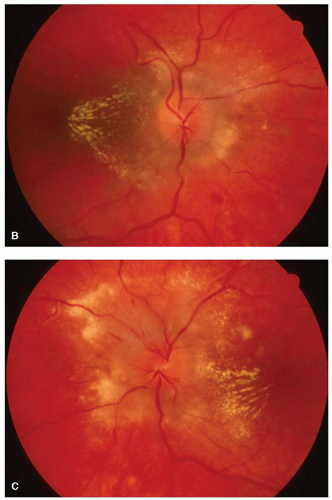
Macular star and exudates deposited within Henle’s layer, giving a typical star or hemistar appearance to the macula. This usually appears after the appearance of optic disc swelling.
Fluorescein angiography shows leakage of dye from vessels on the disc; whereas macular vasculature is usually entirely normal. This investigation is not necessary for the diagnosis.
Neuroretinitis is usually a self-limiting condition, with the optic disc swelling resolving in 6 to 8 weeks. The macular star can take several months and even up to 1 year to resolve. The majority of patients regain good vision.
If visual symptoms persist, the patient usually complains of metamorphopsia or blurred central vision.
History of being scratched by a kitten, which maximally exposes the patient to cat-scratch fever. In this circumstance, titers for Bartonella henselae should be drawn.
A swollen optic nerve and exudates, and a star or hemistar figure in the macula may be seen in other conditions, for example, hypertensive optic neuropathy or papilledema (see p. 62; Fig. 5-9). In these conditions the macula star often is located only nasal to the macula suggesting it is ‘spillover’ from optic nerve head swelling. Therefore, the patient’s blood pressure should be obtained.
It should be noted that this fundus picture is not recognized as the equivalent of optic neuritis. It does not increase the risk of developing MS.
Treatment depends on etiology.
Antibiotic therapy may be administered for cat-scratch fever.
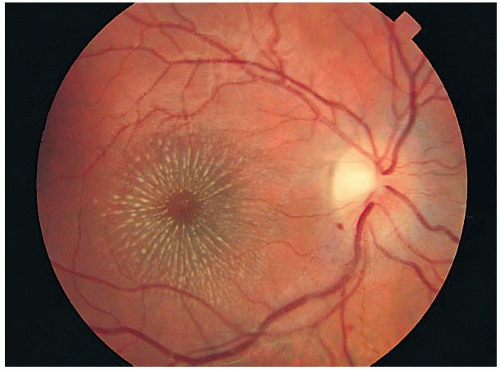 FIGURE 5-8. The optic disc is swollen with nerve fiber layer edema and a hemorrhage at the 7 o’clock position off the disc. The exudates in the macula form of a star. |
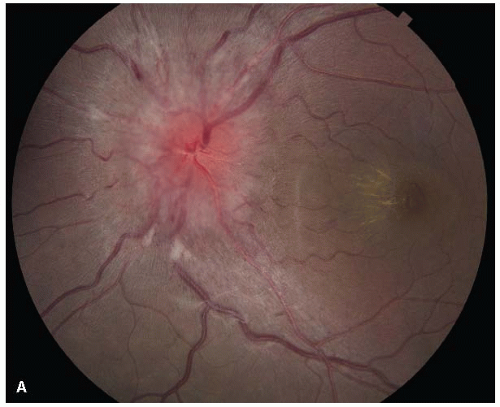 FIGURE 5-9. A. Patient with pseudotumor cerebri due to minocycline ingestion with a swollen disc and a hemimacular star. |
Sarcoidosis is a granulomatous inflammatory process that is identified pathologically by a noncaseating granuloma. Epithelioid and giant cells are characteristic findings on histopathologic studies.
The optic neuropathy of sarcoidosis is produced by one of the three possible mechanisms.
Unilateral or bilateral decreased vision that is usually slowly progressive but may progress rapidly
Decreased color perception
Decreased visual acuity
An RAPD if unilateral or asymmetric
Acquired dyschromatopsia
Central scotoma or other variations of nerve fiber bundle defects are plotted on perimetric evaluation.
Signs of anterior granulomatous uveitis with flare, cells, or mutton fat KP present
Optic disc may be normal if the process is retrobulbar, swollen and hyperemic if the optic disc is inflamed, or show the presence of a sarcoid granuloma. Optic atrophy is often the end result of these optic neuropathies.
A vitreous inflammatory reaction can be seen at times. Lesions at the level of the retinal pigment epithelium (RPE) produce white areas in the fundus (Fig. 5-12). The retinal changes described in the past as venous sheathing (candle wax drippings) are actually retinal or choroidal lesions that at times coalesce (Fig. 5-13).
Nonoptic nerve manifestations of sarcoidosis are listed in Table 5-9.
Other ocular signs of sarcoidosis are listed in Table 5-10.
MRI will show enhancement of the optic nerve or the portion of the anterior visual pathway that is involved (Fig. 5-14). The meninges may be thickened and show abnormal enhancement.
Serum ACE levels may be abnormally high.
Chest X-ray or imaging will often show hilar adenopathy or pulmonary nodules or infiltrates.
Pulmonary function studies often are abnormal.
Gallium scan documents involvement of the lung and lacrimal glands in many patients.
Definitive diagnosis is established by biopsy. Tissue is usually obtained from the enlarged hilar lymph nodes via bronchoscopy or from the lacrimal glands if they appear involved by the process (Fig. 5-15). Blind conjunctival biopsy has a very low yield, while biopsy of a conjunctival lesion (granuloma) visible on the slit lamp examination has a much higher yield.
TABLE 5-9. Other Neuro-ophthalmic Manifestations of Sarcoidosis | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||
Corticosteroids are the mainstay of treatment in this disorder. They should be given systemically for the optic neuropathy.
If uveitis coexists, it should be treated with a topical or periocular corticosteroid.
If corticosteroids fail to halt or reverse the process, agents such as methotrexate should be tried.
Recently, there is increasing evidence that rituximab, an antiCD20 antibody, is beneficial.
TABLE 5-10. Other Ocular Features of Sarcoidosis | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
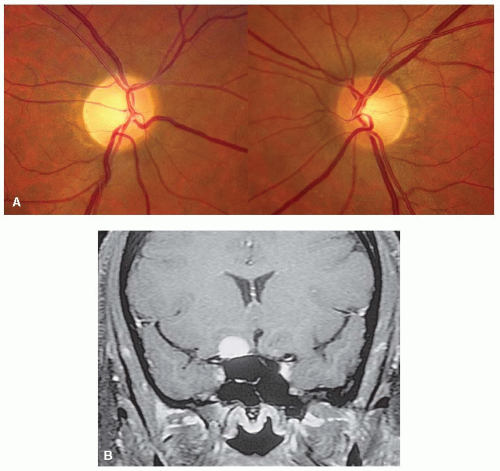
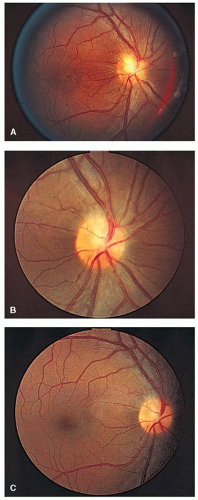 FIGURE 5-11. Right optic disc of patient with biopsy-proven sarcoidosis showing a granuloma of the optic disc (A) that disappeared after 2 years of treatment with systemic corticosteroids (B and C). |
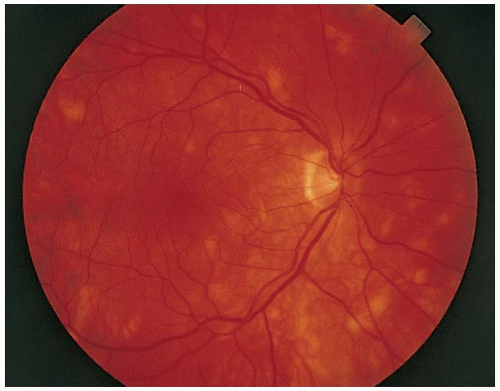 FIGURE 5-12. White lesions in the posterior pole at the level of the retinal pigment epithelium. (Courtesy of Tamara Vrabek, MD.) |
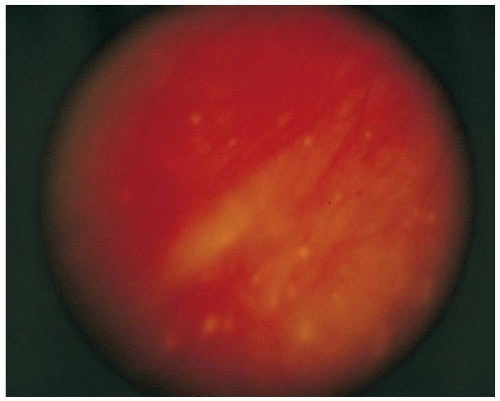 FIGURE 5-13. Retinal pigment epithelium atrophy that may coalesce to form “candle wax spots” in the fundus of patients with sarcoidosis. (Courtesy of Tamara Vrabek, MD.) |
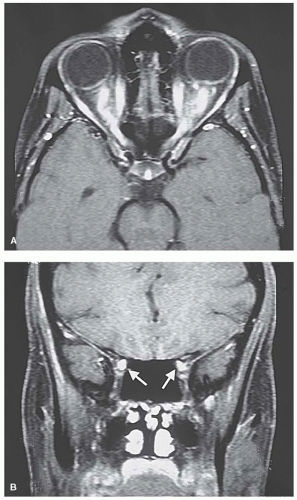 FIGURE 5-14. MRI axial (A) and coronal (B) of patient with sarcoidosis and decreased vision bilaterally showing enhancement of both optic nerves along their entire course. |
Optic neuritis occurs during the secondary and tertiary stage of infection. We consider that syphilitic optic neuropathy implies neurosyphilis.
The general ocular signs of syphilis are listed in Table 5-11.
Decreased vision, which may be unilateral or bilateral
Decreased color perception
Central scotomas
An RAPD if unilateral or asymmetric
At times, a vitreous cellular reaction can exist, especially with papillitis and in HIV-positive patients.
Neuroretinitis can occur in patients with secondary and tertiary syphilis. In secondary syphilis, it can occur as part of syphilitic meningitis and may be either a unilateral isolated phenomenon (with or without uveitis) or bilateral.
Meningitis produces involvement of other cranial nerves or signs of meningeal irritation.
The syphilitic patient, in addition, may manifest the following optic nerve abnormalities:
Optic perineuritis (perioptic neuritis): The optic disc is swollen but visual acuity is normal and there are no other clinical signs of optic neuropathy. This is usually a manifestation of secondary syphilis.
Papillitis: This anterior optic neuritis may be indistinguishable from demyelinating optic neuritis (Fig. 5-16). It usually produces rapid visual loss. However, papillitis secondary to syphillis does not show spontaneous resolution. It may be seen in both secondary and tertiary syphilis.
Neuroretinitis: May be accompanied by vitritis and is encountered in both secondary and tertiary syphilis.
Retrobulbar optic neuritis: Appears as a typical optic neuropathy except that visual
loss may be rapid and profound in secondary syphilis. In tertiary disease, it is a slowly progressive phenomenon.
Papilledema: May accompany the meningitis in secondary and tertiary disease. Its appearance may be confused with optic perineuritis until lumbar puncture documents increased intracranial pressure.
TABLE 5-11. Ocular Manifestations of Syphilis | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||
The best method to prove a syphilitic infection is to demonstrate the spirochete in a
tissue biopsy or CSF. This is not usually feasible, so indirect methods of testing for syphilis are routinely employed. Syphilitic optic neuropathy implies neurosyphilis and suspected cases should undergo CSF analysis.
The serologic tests for syphilis are listed in Table 5-12. Specific testing recommendations appear in Table 5-13.
TABLE 5-12. Tests for Syphilis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-13. CSF Testing in Syphilitic Optic Neuropathy | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||
It is strongly advised that staging and treatment of syphilis be conducted by, or in consultation with, an infectious disease specialist as there is no univerisally established criteria for the diagnosis of neuro-syphilis. However, a general overview of the indications for and the methods of treatment are as follows.
FTA-ABS positive and VDRL negative
If active syphilitic signs and abnormal CSF
Concurrent HIV: These patients may have negative FTA-ABS and VDRL (diagnosis is then made on clinical grounds)
FTA-ABS positive and VDRL positive
If previous VDRL titer (>1:8) did not decrease fourfold within 1 year of appropriate treatment
If previous titer is not available, present titer greater than 1:4, and treatment was more than several years ago
Neurosyphilis may be diagnosed in the presence of a positive serum FTA-ABS and either
CSF-VDRL positive,
greater than 5 WBC/mm3 in CSF,
CSF protein greater than 45 mg/dL.
Treatment consists of the following:
Intravenous aqueous crystalline penicillin G2 to 4 million U, q4hr, for 10 to 14 days, followed by intramuscular benzathine PCN, 2.4 million U weekly for 3 weeks.
If the CSF is normal, only the IM benzathine PCN, 2.4 million U weekly for 3 weeks is required.
There is an indication that the dosage of penicillin should be higher in HIV-positive patients.
Follow-up: Patients with neurosyphilis need repeated lumbar punctures every 6 months until the white cell count returns to normal. If it does not decrease, retreatment may be indicated.
NAION is thought to be the result of vascular insufficiency in the posterior ciliary circulation affecting the distal optic nerve, although this has not been proven.
The incidence of NAION is between 2 to 10 per 100,000 persons over the age of 50. The average age of onset is between 55 and 65 (range 40 to 70) years, although it is becoming more frequently diagnosed in younger patients with known risk factors.
Risk factors that are thought to be important include the following:
Small cup-to-disc ratio and small optic discs (also referred to as congenitally anomalous discs or “discs at risk”) (Fig. 5-17). This is probably the most important risk factor associated with NAION.
Hypertension
Diabetes mellitus
Hypercholesterolemia
Other vascular risk factors: Conditions associated with small vessel disease and coagulopathies may be important, although the evidence is not conclusive.
Following profound blood loss (either spontaneous, a result of surgery, or severe hypotension)
Post-cataract surgery
Optic disc drusen (appear to predispose to NAION)
The association of phosphodiesterase type 5 drugs [sildenafil (Viagra®), tadalafil (Cialis®)] with NAION is controversial. The World Health Organization has labeled the association “possible.”
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree