Fig. 1
The “domino effect” of Pierre Robin sequence (Courtesy of David Low, MD)
The triad of micrognathia, glossoptosis, and tongue-based airway obstruction (TBAO) was initially described in 1939 by Dr. Pierre Robin, a French somatologist, who implicated the small mandible as the causative deformity leading ultimately to airway compromise [2, 3]. This “domino” effect is therefore known as Pierre Robin sequence (PRS). A sequence is constellation of abnormalities which are linked from an inciting anomaly or deformity. In the case of Pierre Robin sequence, failure of normal mandibular development sets off a cascade of anatomic changes in utero which result in the varying and seemingly unrelated physical findings (Fig. 2). {Figueroa 1991}.
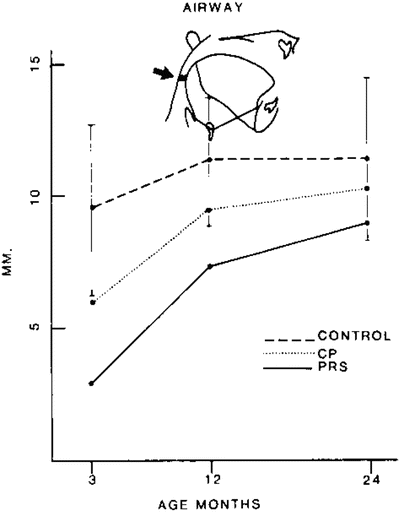

Fig. 2
Catch-up growth (Reprinted from [40] Permission granted from Allen Press Publishing Services)
The etiology of this mandibular deformity is the topic of much debate. Several studies have attributed the micrognathia seen in Pierre Robin sequence to intrauterine deformation or extrinsic factors rather than intrinsic growth restriction of the mandible itself [4]. From between the 6th to 12th week of gestation, the human embryo transitions from a position of extreme neck flexion where the mandible is buried in the upper thorax to a position of gradual extension [5]. This period coincides with rapid mandibular growth that allows the tongue to descend and the palatal shelves to fuse. Several factors including multiple gestation, oligohydramnios, and cervical hemivertebrae may restrict this extension [6]. These studies point to the phenomenon of “catch-up growth” as evidence that extrinsic compression plays an important role in the deformity.
Increasingly, attention has focused on genetic cause of PRS. In addition to the syndrome-associated cases of PRS such as those patients with Stickler or Treacher Collins syndromes who demonstrate intrinsic mandibular growth deficiency, non-syndromic PRS may also have a genetic component. Investigators have demonstrated increased frequency of palatal clefts in the parents of patients with PRS [7] as well as several novel genetic mutations present in a proportion of such patients [8, 9].
No matter the cause, PRS is a challenging disease process which can result in acute airway compromise, chronic obstructive sleep apnea, cor pulmonale, anoxic brain injury, and even death [10]. In the neonatal period, this entity can be particularly devastating, often necessitating emergent postnatal intubation in severe cases. Even in patients who do not require emergent airway management, chronic hypoxia, feeding intolerance, and failure to thrive may be common [11]. In one of Pierre Robin’s early publications, he describes the grim prognosis of a severely affected child: “I have never seen a child live more than 16–18 months who presented hypoplasia as such the lower maxilla was pushed more than 1 cm behind the upper”[2].
History and Epidemiology
Congenital micrognathia is associated with over 100 known syndromes [12]; however, in the setting of tongue-based airway obstruction as is seen with Pierre Robin sequence, it is estimated to occur in 1 in 8,500 to 1 in 14,000 live births [13]. In 40–60 % of cases, PRS occurs in isolation, as the only disease process. However, over half of patients may carry syndromic diagnoses which not only play a role in disease pathogenesis but may contribute to increased disease severity. The most common of these associated syndromes, for example, is Stickler syndrome, which has prevalence of 1 in 8,000 live births in the general population but is present in over 30 % of patients with PRS. Table 1 lists several of the major syndromes commonly associated with PRS.
Table 1
Frequency of associated syndromes
Frequency in general populationa | Frequency in Robin sequenceb | |
|---|---|---|
Stickler | 1:8,000 | 1:3 |
Velocardiofacial Syndrome | 1:2,000 | 1:9 |
Fetal alcohol | 1:1,000 | 1:10 |
Treacher Collins Syndrome | 1:25,000 | 1:20 |
Undefined syndrome | – | 1:3 |
Classification
There is no widely accepted classification system to stratify disease severity in patients with Pierre Robin sequence. As mentioned previously, several studies have associated worse outcomes in PRS patients who also carry syndromic diagnoses, and most centers now routinely recommend genetic screening after diagnosis. However, there is a wide variability in the severity of many syndromic diagnoses, and the simple presence or absence of a syndrome, alone, does little to differentiate such patients. Patients with Stickler or Nager syndromes often have more severe presentations than in PRS patients without accompanying syndromes, whereas many syndromes seen in these patients may contribute little or not at all to disease severity. Although many studies demonstrate worse outcomes in syndromic PRS as a whole, we caution against making management decisions based solely on the presence or absence of a syndrome.
Indeed, the key to differentiating the impact of micrognathia on the neonatal airway is to quantify its degree of deformity, and those syndromes which adversely affect outcomes in Pierre Robin sequence are all associated with macroglossia or micrognathia. In order to assess the severity of anatomic mandibular deficiency, several authors advocate for the measurement of maxillary-mandibular discrepancy (MMD) as a simple, reliable, and reproducible method for stratification [14]. From the “worms eye” position looking superiorly, the mandible is gently closed to the maxilla and the distance between the midline mandibular alveoli and maxillary alveoli is measured. The realization that maxillary projection may also be inadequate in some forms of PRS, however, limits the utility of this tool [15]. In contrast, others recommend a clinical grading scale based on the severity of airway compromise [16].
The only validated classification system for patients with Pierre Robin system was developed as a means of determining which patients would benefit from surgical intervention. The GILLS criteria [17, 18] assesses for the presence of five factors which are shown to predict which patients might benefit from tongue-lip adhesion (TLA) surgery and which patients required direct tracheostomy (Table 2). Those patients with scores of two or less had a 100 % chance of success with the procedure whereas those patients with scores of three or more had close to a 50 % failure rate requiring tracheostomy.
Table 2
GILLS scoring system for tongue-based airway obstruction
GILLS Criteria | |
|---|---|
1 | Presence of GER |
2 | Preoperative intubation |
3 | Late presentation (>2 weeks old) |
4 | Low birth weight (<2,500 g) |
5 | Syndromic diagnosis |
Clinical Presentation
As stated previously, patients with Pierre Robin sequence demonstrate a wide spectrum of disease severity ranging from subclinical presentation to frank life-threatening respiratory compromise. Although the degree of micrognathia plays an important role in the development of respiratory symptoms, this is often a subtle finding in the neonatal period and observation for clinical signs of distress is imperative.
In the immediate postnatal period, patients with severe forms of Pierre Robin sequence will often display signs of distress such as grunting and crying, obvious apneic episodes, and even cyanosis. Those less severely affected may demonstrate increased work of breathing including supra- and substernal and intercostal retractions as well as cervical hyperextension. Symptoms are often positional with some improvement in prone position. Some neonates show few signs of respiratory compromise while awake. However, as resting tone decreases during phases of deep sleep, the tongue may assume a more posterior posture leading to obstruction.
In the first weeks of life, patients with moderate obstruction may present during an initial well child visit with inadequate weight gain, cachexia, or even failure to thrive sometimes without a history of apneic events. There are several reasons for feeding difficulties in PRS. First, the small mandible and tongue malposition as well as the presence of a palatal cleft pose significant physical restrictions on infant suckling. Indeed, primary oropharyngeal dysmotility has been noted in some patients. Second, feeding in infancy poses substantial metabolic demands, and patients with subnormal oxygenation may present with “exercise-induced” anorexia. Finally, gastroesophageal reflux (GER), although common in many infants, is especially challenging in those with Robin sequence with a reported incidence as high as 85 %. [19] As the tongue falls back, infants may increase inspiratory pressures in order to overcome the obstruction. The increased negative intrathoracic pressure then overcomes lower esophageal sphincter tone, and the gastric contents are “sucked” into the esophagus and the bronchial tree [6]. Such subclinical aspiration can be a substantial source of morbidity.
Cleft palate, although not a diagnostic criteria, occurs in the majority of patients with PRS, so much so that it is often incorrectly included among its essential characteristics. It is, however, distinct from other forms of palatal clefting with or without cleft lip [20]. The cleft palate associated with Pierre Robin sequence most often is wide and U-shaped, a result of failure of the palatal shelves to fuse in early gestation (8–12 weeks) due to the presence of an abnormally positioned tongue.
Evaluation and Clinical Approach
Timely multidisciplinary team evaluation—including Neonatology, Genetics, Pulmonology, Otolaryngology, the Feeding Team, and Plastic Surgery—is essential to maintaining adequate oxygenation and weight gain in patients with Pierre Robin sequence. A thorough history and physical exam should be performed to assess for potential syndromic association as well as evaluate overall appearance and tone. Indeed, hypotonia has the potential to exacerbate airway obstruction in affected patients and, in severe cases, may suggest poor response to interventions which address the upper airway. A complete airway exam is also an imperative first step in evaluation to determine the need for adjunctive airway support including high flow supplemental oxygen, continuous positive airway pressure, and intubation if necessary. Continuous pulse-oxymetry should also be employed early on in order to fully evaluate the number and degree of obstructive events.
Plain lateral radiographs with soft tissue windows may help characterize degree of micrognathia as well as assess for severity of glossoptosis by allowing for visualization of a patent or occluded airway stripe. In neonates the airway stripe should measure approximately 4 mm [21]. Although such an evaluation may provide insight into the degree of upper airway compromise, it is not substitution for direct visualization of the airway.
Bedside fiberoptic naso-endoscopy is an important tool to localize the level of airway obstruction and should be performed prior to any definitive intervention. In addition to a noninvasive way to visualize the lower airway, endoscopic evaluation of the entirety of the upper airway can rule out other causes of obstruction such as choanal atresia. Additionally, direct micro-laryngo-bronchoscopy (MLB) is essential to evaluate subglottic structures and to rule out the presence of laryngomalacia, tracheomalacia, and other pathologies [22]. A jaw-thrust maneuver performed under anesthesia at the time of MLB can help determine the extent to which mandibular advancement can improve tongue base position. Presence of lower airway pathology may significantly alter management decisions.
Diagnosis
The diagnosis of Pierre Robin sequence relies on the clinical finding of micrognathia in the setting of tongue-based airway obstruction. Although the presence of micrognathia is easy to establish on exam, and airway obstruction is readily apparent in severe cases, the extent to which the tongue base contributes to airway compromise can be difficult to determine. For this reason, diagnostic airway endoscopy is essential to confirm the presence of glossoptosis and rule out other sources of upper or lower airway obstruction.
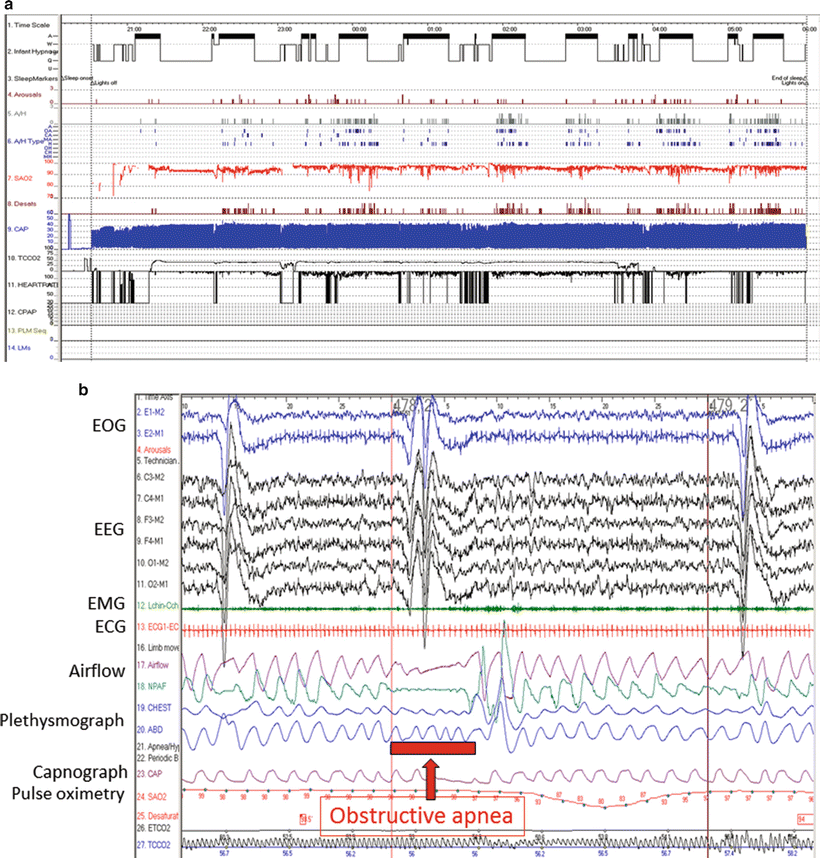
Mild to moderate cases of Pierre Robin sequence, however, where profound desaturation and cyanosis are often absent, present diagnostic challenges to clinicians. In these settings, polysomnography (PSG) has been utilized to establish and characterize the presence of apneic events and to quantify the frequency and degree of airway obstruction [14]. Sixteen lead polysomnography, in particular offers a thorough evaluation of neonatal sleep and respiratory function. It consists of electroencephalographic, electrooculographic, electromyographic, electrocardiographic monitoring as well as detailed analysis of respiratory performance and tissue oxygenation throughout the sleep cycle. It is utilized to quantify the number of apnea and hypopnea events per hour (apnea–hypopnea index) as well as the severity of obstruction as measured through oxygen saturation and end tidal and transcutaneous carbon dioxide measurements (Fig. 3). Indeed, PSG plays an important role in establishing the presence of subtle obstructive events that may be overlooked clinically during wakefulness and is crucial for quantifying the severity of airway obstruction during sleep [23–25]. Additionally, PSG can differentiate central apneas secondary to brainstem dysfunction and the obstructive events that may be improved with intervention. Many practitioners now advocate for an expanded role for polysomnography beyond mild presentations of the disease citing its utility as an objective way to measure improvement after airway interventions.