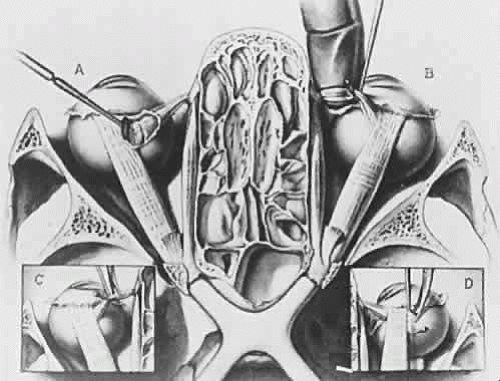
Ocular Motility
Herschel P. Goldstein
Alan B. Scott
THE ORBIT
The medial orbital walls are generally parallel; the lateral orbital walls diverge one from the other at an angle of approximately 90 degrees (Fig. 1). The orbits reach adult proportions at age 13 to 15 years, with the visual axes of the two eyes separated by 60 to 70 mm. The orbits are closer to one another in infancy, and in some children an especially narrow interocular distance leads to an appearance of the eyes being crossed, or esotropia. Growth of the nasal sinuses widens this distance. Displacement or rotation of the orbits can carry the extraocular muscles and vary the normal geometry of their action. For example, if the orbits are rotated in craniofacial disorders, the normal horizontal action of the horizontal rectus muscles takes on also a vertical action. Imaging of the bones and muscles by computed tomography (CT) or by magnetic resonance imaging (MRI) is useful to tell these variations from normal.
BASIC GEOMETRY OF EYE POSITIONS
NOMENCLATURE
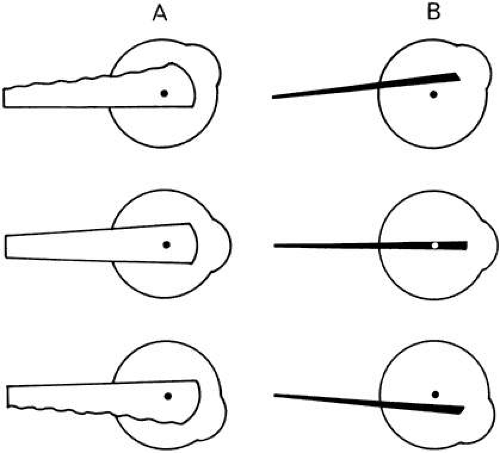
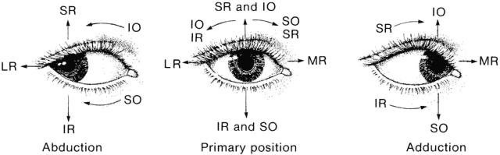
Monocular eye movements are called ductions. Binocular eye movements in the same direction are called versions or gaze movements. Normal eyes maintain the same relative alignment in various positions of gaze: comitance. When the eyes move unequally because of muscle weakness or restrictions, incomitance results. Eye movements in contrary directions are called vergences. A tropia is a manifest deviation of the visual axes. A tendency to deviation of the visual axes held in check by the fusion mechanism is a phoria. See Figure 2 for these and additional terms.
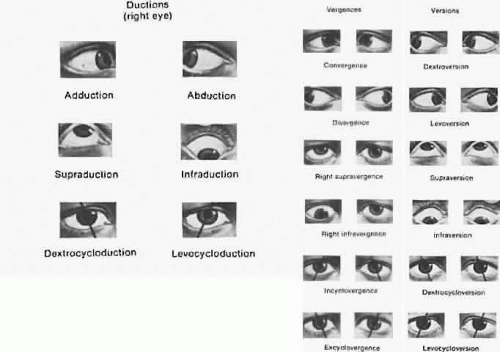 Fig. 2. Ocular movements. (Duke-Elder S: System of Ophthalmology. Vol 6. Ocular Motility and Strabismus. London: Henry Kimpton, 1973.) |
In agonist-antagonist relationships, the muscles pull in opposite directions, for instance, medial rectus versus lateral rectus. To a substantial degree, vestibular gaze and control of oculomotor function are arranged around these muscles as pairs that control rotation in horizontal, vertical, and torsional directions.
Synergists are agonist muscles pulling in the same direction. Muscles often are synergists for one direction, such as the superior rectus and inferior oblique for supraduction, while at the same time acting as antagonists for their horizontal and torsional vectors.
GLOBE AND ORBITAL AXES
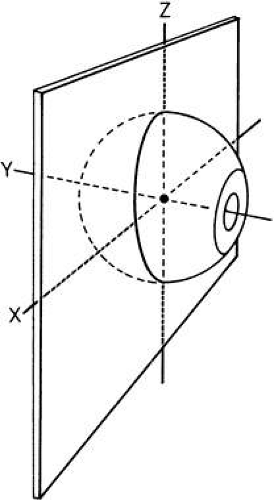
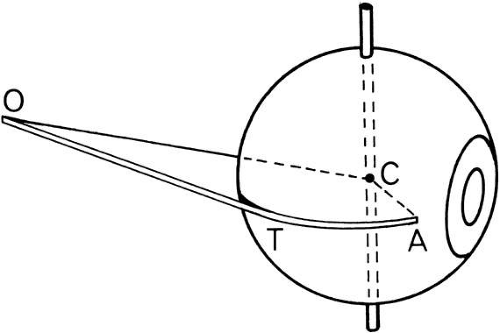
The eye is a sphere rotating around three orbital axes: vertical rotation around the horizontal (x) axis, cyclorotation around the anterior-posterior (AP) or y, axis, and horizontal rotation around the vertical (z) axis. The eye has a similar set of axes, which rotate with the eye (Fig. 3). The study by Howard and Templeton provides further description of axis systems.1 The eye and its center of rotation can also be translated by small amounts in other directions: medial to lateral, up to down, and inward to outward.
POSITIONS OF THE EYE
The global axes can be compared with the orbital axes in various gaze positions by marking a cross on the front of the cornea to tell how the eye is oriented (Fig. 4).
Primary Position
In this position, the two sets of axes, global and orbital, coincide exactly. The geometric AP axis of the eye is normally close to the visual axis and the latter is taken as the y axis. An example is a soldier standing at attention with his eyes straight ahead on the horizon: the eyes in this position approximate the primary position.
Secondary Positions
These positions rotate around the horizontal or vertical axis only. Movement of any amount vertically straight upward or downward leaves the horizontal (x) axis in its same position and tilts the vertical (z) and visual (y) axes in the vertical plane. A similar situation occurs in purely horizontal rotation away from the primary position. No rotation occurs around the y axis.
Tertiary Positions
All other positions are termed tertiary positions. If the right eye is turned obliquely up to the right, the horizontal and vertical axes of the eye now no longer stay parallel to the vertical and horizontal axes of the orbit. They tilt relative to the vertical meridian that exists in the new position. This is called cyclorotation or torsion. The eye has rotated around all three axes, including its line of sight, the y axis.
DONDER’S LAW AND LISTING’S LAW
Donders found that for any gaze position, there is a predictable amount of torsion, and that this is independent of how the eye travels to that gaze position.2 The amount of torsion increases with increased movement of gaze from the primary position. For an oblique position of 10 degrees vertical and 10 degrees horizontal, there is 1 degree of torsion, for 20 degrees vertical and horizontal, 3 degrees of torsion, for 30 degrees, 8 degrees; for 40 degrees, 15. Listing proposed (and Von Helmholtz formulated and verified) a clever way to consider the torsions that Donders described.3 The image is that of a vertical plane cutting the eye into front and back halves going right through the center of rotation at the equator (see Fig. 3). Next imagine a rod attached to the center of this equatorial plane (called Listing’s plane) so that it can spin around like a hand on a clock. This rod is the axis of rotation for the eye. For an eye position straight upward, this axis is placed horizontally from the 3- to the 9-o’clock positions. For a rotation obliquely, this axis is placed at an oblique axis, say 10:30 to 4:30 positions. The advantage is that only two data are needed to describe the eye position instead of three: one, the orientation of the rotation axis, and two, how far the eye rotated in that direction. The torsion that is required to be added in a triaxial system turns out to be automatically corrected using Listing’s system. Listing’s law has been tested under a variety of circumstances and shown to be correct for vertical and horizontal movements in the frontal plane with the head erect. Listing’s law does cease to be demonstrable in abnormal innervations causing restrictions to movement and also for normal convergence positions; there is about 1 degree of excyclophoria for each 4 degrees of convergence.
The triaxial system is no problem for modern computers; all serious modeling of eye movements is currently performed using the triaxial scheme. Clinicians think in terms of horizontal, vertical, and cyclotorsional effects of the muscles in a system of three axes. Moreover, the vestibular system driving many eye movements is also organized into vertical, horizontal, and torsional subsystems.
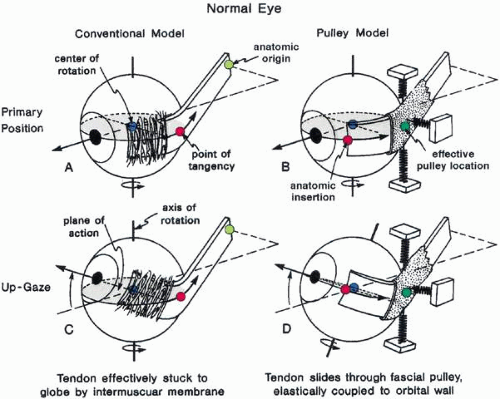
But Listing’s concept is more than a clever way to think about normal eye rotations and turns out to have some basis in anatomy. Nakayama conceived of Listing’s plane as an elastic membrane with the eye suspended in it (Fig. 5).4 If the eye is pulled in any direction, it obeys Listing’s law. Anatomically, there is a matrix of fatty fibromuscular tissue suspending the eye and the anterior third of the muscles in the orbit and attached to the orbit walls. This is very like the eye suspended in a foam rubber layer several millimeters in thickness that acts like the membrane of Nakayama. The eye is thus somewhat mechanically constrained. During normal large saccadic movements accompanied by strong head rotation, transient torsional deviations up to 25 degrees have been found,5 showing that this mechanical restraint is only partial. In addition, the eye muscles are programmed to have a certain particular set of pulls for each gaze position. Large deviations from Listing’s law occur with non-paralytic brainstem lesions altering the innervation program. So Donders’ law is also confirmed by eye muscle innervations.
POSITION OF REST
The eye positions of orthophoric, normal individuals under deep anesthesia with curare paralysis are probably the closest approximation to a true position of rest. De Groot and coworkers found an exotropia of 2.25 degrees for each eye in young adults, which increased with age.6 This is in accord with the estimate of most clinicians. In newborns whose eyes are straight when awake, the visual axes are commonly divergent up to 35 degrees in deep sleep, which become progressively straighter within only a few weeks.7 The position of the eyes under anesthesia in strabismus cases helps to show anatomic vertical, horizontal, or torsional factors underlying the alignment disorder. Differences in waking eye position from those while asleep indicate innervational factors. In general, esotropic cases are less esotropic, showing that innervational factors are dominant, and exotropia cases are about the same, showing that anatomic position is the major factor. Difference between the position of the two eyes shows the effect of unequal anatomic factors; rotation of the fundus structures indicate torsional factors.
ACTION OF MUSCLES
The pull of a muscle on the eye is determined by (1) the size of the muscle (cross-section, primarily), (2) the direction it is pulling, (3) the innervation to the muscle, and (4) the mechanical characteristics of the muscle.
SIZE
Cross-sectional areas of the extraocular muscles in square millimeters from Volkmann and Nakagawa’s measurements appear in Table 1.8,9
TABLE 1. Eye Muscle Area, Length, and Innervation | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
COMMENTS
Horizontal recti are largest. This is sensible, because they alone are horizontal movers.
Vertical recti average about 75% the size of the horizontals.
Obliques are about 50% the size of the horizontals.
Vertical recti and obliques act together as snyergists to provide vertical and torsional movement. Thus, individually they may be smaller than horizontals.
In general, antagonists such as medial and lateral rectus are similar in size, thereby balancing opposing forces.
Muscles exert force in proportion to their cross-sectional area, about 30 g/mm2, according to Huxley.10 Although a horizontal extraocular muscle of 15 mm2 cross-sectional area should be able to exert a tension of 450 g, we have never found a tension of more than 150 g, even when testing isometrically. Comparison with other muscles confirm this low force.11,12
LENGTH
The extraocular muscles are from 32 to 40 mm in length (see Table 1). Inasmuch as each 1.0 mm on the surface of the eye corresponds to 4.5 to 5 degrees of rotation, the normal amplitude of eye rotation of 45 to 50 degrees each way from the primary position requires 10 mm change of length of the muscle in each direction. This represents a contraction of 25% to 30% and an extension of 25% to 30% from the normal resting length of the muscle, nearly optimal excursions for linear performance. Sacrifice of muscle length during eye muscle operations can reduce the amplitude of eye rotation. Muscle length is not proportional to eye size, so that large myopic eyes rotate less and small microphthalmic eyes rotate farther than normal.
DIRECTION OF MUSCLE PULL
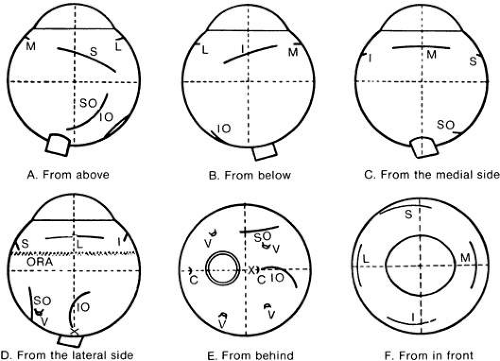
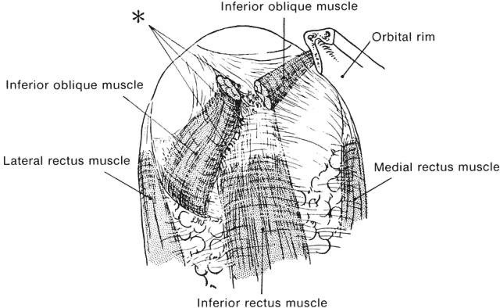
Muscle attachments and directions of pull (Fig. 6) are defined as follows:
Origin is the point of muscle attachment to the bony orbit.
Anatomic insertion occurs at the point at which the muscle blends into the sclera.
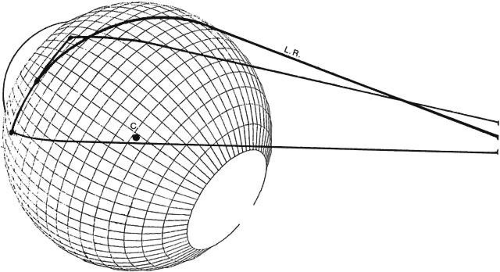
Point of tangency is the point at which the muscle first touches the globe as the muscle extends from origin to its anatomic insertion.
Arc of contact is formed as the muscle or tendon lies on the sclera between the point of tangency and the anatomic insertion (see Fig. 6). The muscle can uncoil onto and off the globe like a rope and pulley. As the globe rotates, the arc of contact may lengthen or shorten.
Functional insertion is usually the point of tangency. This point on the sclera is constantly changing as the eye rotates toward the muscle origin and away from the muscle origin, but the actual pull is exerted here. The medial rectus has a short arc of contact, about 6 mm. In theory, then, after 30 degrees of rotation, the anatomic insertion goes behind the point of tangency. Then, the anatomic insertion becomes the functional insertion. In fact, however, MRI scans show that in adduction the medial rectus does not run in a straight line from the origin to the globe but bows toward the center of the orbit, thus maintaining tangency. At the same time, the antagonist lateral rectus bows outward toward the lateral orbit wall. All rectus muscles do this to some degree.13 These effects are due to hydraulic forces on the orbital fat and to circumferential fascial tissues holding the rectus muscles together. The result is that the arc of contact of the agonist muscle on the globe so that it uncoils a little less than simple geometry would indicate.14 The fascial tissues constrain the muscle to be close to the globe and act to transfer some muscle force to the eye. If both the agonist and antagonist muscles are detached from the scleral insertion, less than a total paralysis of effect occurs.
Muscle plane. The origin, functional insertion, and center of rotation establish an imaginary plane through which the muscle pulls to move the globe (see Fig. 6).
Muscle axis of rotation is perpendicular to the muscle plane. The individual muscle pulling on the eye rotates the globe around this axis through the center of rotation. For the horizontal recti, it coincides with the vertical axis of the globe when the eye is in the primary position. For other muscles, it is oriented at various angles established by their coordinates of origin and insertion (see Table 1, Fig. 7).
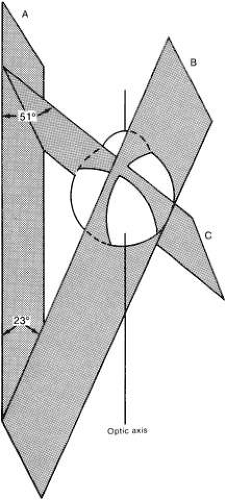 Fig. 7. Planes of medial orbital wall (A), vertical recti (B), and oblique muscles (C). (Cogan DG: Neurology of the Ocular Muscles. 2nd ed. Springfield, IL: Charles C Thomas, 1956.) |
The forces of muscle contraction and relaxation pull on the eye according to the muscle plane established by the origin, functional insertion, and center of rotation. Clinical strabismus patterns due to anatomic variation of this anatomy, which can be shown on CT or MRI scans, are not rare. For more detailed measurements of arc of contact, exact point of origin, and so forth, see Tables 1 and 2.
TABLE 2. Coordinates of Origin and Insertion of Ocular Muscles* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PRIMARY AND SECONDARY ACTIONS OF MUSCLES
One scheme is to assign rotation around each of the axes as a major or minor function of muscles:
The primary action of the horizontal rectus muscles is 99% horizontal. They have trivial secondary or tertiary actions.
The primary action of the vertical rectus muscles is 75% vertical, and they have secondary torsional and horizontal actions. The primary action of the oblique muscles is 60% cyclorotation (torsion) and they have secondary vertical and horizontal actions.
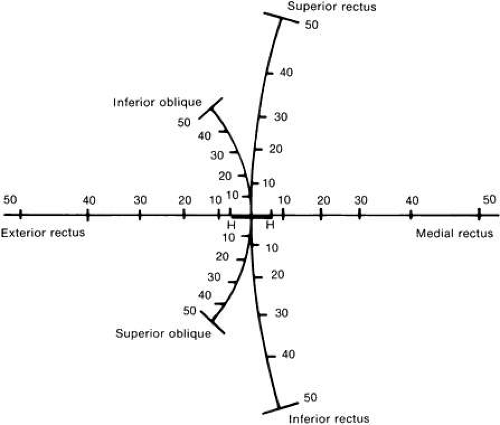
Another helpful concept of how each muscle acts from the primary position is to show what would happen if you plotted the direction of your fovea on a wall while each muscle individually pulled the eye in its muscle plane (Fig. 8).
MUSCLE ACTION FOR POSITIONS AWAY FROM THE PRIMARY POSITION
When the eye moves to new positions, the orbit axes stay fixed and do not move. The eye carries its axes and the muscle insertions along to the new position with it. The action of each muscle on the eye changes because the eye axes are now in different positions.
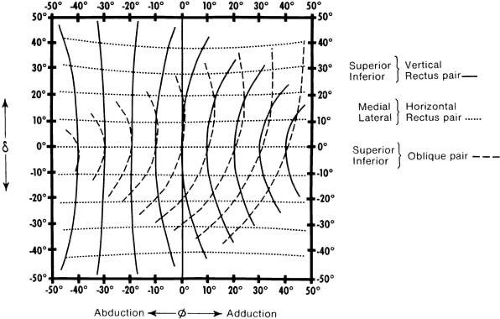
Figure 9 shows the effect that each muscle pair would have if it were the only one pulling. This graph is like that in Figure 8 but involves other gaze positions in addition to the primary position. The horizontal recti are considered as starting from various positions of vertical gaze. There is a minimal change of effect; they remain as horizontal movers. The effect of the oblique and rectus muscles is considered starting from various positions in the horizontal plane. The vertical rectus muscles have a nearly pure vertical effect when the eye is in abduction, but increasing torsional and horizontal effect as the eye moves into adduction. For the obliques, there is almost purely vertical action in extreme adduction and almost purely torsional effect in extreme abduction. The superior and inferior rectus muscles are quite symmetric, whereas the superior and inferior oblique are not quite symmetric, as a result of the long arc of contact of the inferior oblique.
STABILITY OF MUSCLE PLANES IN THE ORBIT
The anatomic muscle insertions on the globe are carried along by rotation of the eye to new positions. What about the functional origins and insertions: are they moved to the same degree as the anatomic insertions? For example, in the classic geometric analyses in Figures 8 and 9, the muscles are supposed to behave as if they were a single string connecting the origin and center of insertion and as if the muscles could slip sideways over the globe.15,16 Four factors alter this: (1) first is the width and geometry of the insertion (Fig. 10). The extraocular muscle insertions are all convex at their insertional end; (2) they are all approximately 9 to 10 mm wide; (3) the muscle almost bisects the center of rotation of the globe on its way to the insertion; and (4) these insertions are all approximately 5 to 6 mm in front of the center of rotation (inferior oblique excepted).
The practical effect of this on, for instance, the medial rectus, is to slack off the upper fibers during upward gaze and transfer the effective tension generated by the muscle from the center of the muscle to the lower fibers. Similarly, in downward gaze, muscle tension is transferred to the upper fibers, and the lower border of the muscle is slack (Fig. 11A). Were this not true, the situation would be as shown in Figure 11B: the medial rectus would become an elevator in upward gaze and a depressor in downward gaze. Similar figures show the stabilizing effect of a wide insertion on the line of pull of oblique and vertical rectus muscles. Von Helmholtz first described this attribute of the width of the extraocular muscles tending to hold the direction of pull of the muscle stable in the orbit, regardless of eye position.
MUSCLE PULLEYS
The trochlea (pulley) of the superior oblique constrains the tendon and changes its direction of pull so that a new origin is created. Less dramatic accumulations of fibrous orbital tissue surround the rectus muscles just behind the equator and extend to the orbit holding it to some degree to the orbit wall (Fig. 12). These tissues also act to limit sideslip in the rectus muscle and to limit the surgical effect of rectus muscle transposition operations in which the insertion is moved laterally. They bias the origin as the trochlea does, but to a much lesser degree, thus acting as “soft pulley” (Fig. 13). such displacement occurs in strabismus disorders, but it is not always clear whether the displaced muscle is taking the pulley with it or vice versa. These tissues have long been known to contain smooth muscle fibers.17 Innervation to these structures and their functional significance is a current topic of investigation.
MUSCLE STIFFNESS
Imagine that the medial rectus consisted only of two stiff chains. Then, when the eye rotated upward, the smallest bit of slack created by relaxation at the upper border of the medial and lateral muscle totally relaxes tension and transfers it to the lower border. Clearly, this jerkiness would be unsatisfactory, and the smooth compliant nature of the extraocular muscle and its convex anterior insertion are factors that combine to transfer this force easily and smoothly from one side of the muscle to the other. On the contrary, if the muscle were extremely flexible, like a wide elastic band, then relaxation on one side would have very little effect on the transfer of tension along the muscle insertion. In fact, the stiffness of the muscle, about 0.25 to 0.5 g per degree, seems exactly adapted. In awake patients, when the upper part of the medial rectus insertion is cut away from the globe except for 2 mm at the lower end of the insertion, the cut muscle retracts, making an angle of about 45 degrees with the original insertion. When the patient rotates his eye superiorly, the cut superior fibers fall back almost exactly onto the insertion, an indication that the inferior fibers are taking almost the entire strain. Thus, as the eye carries the medial rectus insertion up, the force moves down, keeping its pull horizontal on the globe and horizontal in the orbit.
SIDESLIP OF MUSCLES OVER THE GLOBE
The intermuscular membrane stabilizes the anterior portion of the rectus muscles from the equator forward, preventing each muscle from slipping sideways. In Figure 14, the path of the lateral rectus in gaze nasally and upward is indicated, assuming the muscle took the shortest path from origin to center of insertion. This extreme upward path is not taken, but a course nearly over the lateral meridian of the eye is maintained by the intermuscular membrane. If the intermuscular membrane is cut away at the time of surgery, the horizontal muscle slips about 2 mm vertically during vertical movements. A second factor holding the rectus muscle from slipping sideways is the adhesion or “foot plate” that attaches the global surface of the muscle to the sclera and extends some 4 to 5 mm posterior to the insertion. Abnormal vertical eye movements from such sideslip of horizontal can occur in strabismus disorders in which high forces gradually tear and obliterate the intermuscular membrane (Duane’s syndrome), and in high myopia in which expansion of the globe, usually in the upper temporal area, stretches the intermuscular membrane and pushes the lateral rectus downward. These disorders can be treated by fixing the muscle to the sclera at the equator to prevent sideslip.18,19 The superior oblique tendon has a very short length and arc of contact, and here sideslip is not a significant factor. The inferior oblique has a very long arc of contact. Indeed, the necessary length of muscle to allow extension and contraction is provided by the coiling of the inferior oblique muscle around the globe for a long distance. The ligament of Lockwood, at the crossing of the inferior oblique and inferior rectus muscles, acts to prevent A-P slip of the inferior oblique, just as the intermuscular membrane acts in terms of the rectus muscle (Fig. 15). Eye muscles imaged on MRI scans had only small sideslip when subjects fixated in various directions.20
COHESIVENESS OF THE MUSCLE
The belly of the muscle is readily split longitudinally because it contains little transverse fascial tissue, but the anterior muscle is cohesive within itself to prevent separation because otherwise, the fibers of the lateral rectus muscle would splay out almost 18 mm wide in extreme gaze into elevation and adduction. Thus, the anterior muscles, their surrounding Tenon’s fascia, and the intermuscular membranes form a continuous stabilizing band or cap around the globe extending from the muscle insertions anteriorly back to the equator. The muscle “pulley” tissue anchoring the muscles to the orbit adds to this.
FIELD OF ACTION OF MUSCLES
Even though all these muscles have some effect on the eye in every gaze position, each pulls preferentially in a specific direction, called its field of action. Torsion is not usually considered, only horizontal and vertical actions. The field of action is established by two considerations: geometry and innervation.
GEOMETRY
We have seen in our analysis that the lateral rectus pulls toward abduction almost exclusively. That is its field of action. The medial rectus pulls toward adduction almost exclusively. That is its field of action.
For vertical gaze such a simple arrangement is not present, for a combination of muscle actions exists. In 23 degrees of abduction, the superior rectus is a pure rotator around the horizontal axis of the adducted eye. This is its field of vertical action for clinical testing. If the eye rotates well vertically in this position, it must be due to a normal superior rectus. Here, the inferior oblique muscle contributes little vertical movement. In adduction, the oblique becomes an increasing elevator, but the superior rectus remains a substantial elevator even in adduction because of its angle of insertion and its greater cross-sectional area. Thus, elevation in adduction is a combination of inferior oblique and superior rectus activity. Nevertheless, vertical contribution of the oblique can be assessed clinically by comparing elevation in abduction, in which it has virtually no effect on elevation, with elevation in adduction in which it contributed 45% to 50% of elevation (Fig. 16). The superior oblique and the inferior rectus muscles have the same relationship in infraduction. The inferior rectus is virtually the sole infraductor in the abducted gaze positions, and both the superior oblique and the inferior rectus contribute as gaze is moved toward adduction.
INNERVATION TO HORIZONTAL MUSCLES
In addition to the mechanics, changes in the innervation to the muscles in various gaze directions establish what they do; this might be thought of as field of activation. For abduction, innervation progressively increases to the lateral rectus as the eye abducts from the primary position. This is true whether it starts from the primary position, or whether the eye starts from a position of slight elevation or depression: the program of innervation of the lateral rectus appears to be the same. Similarly, the medial rectus increases activity with all attempts to adduct the eye, whether the eye starts in the primary position, in moderate supraduction, or moderate infraduction.
INNERVATION TO VERTICAL MUSCLES
The elevators of the eye, the superior rectus and the inferior oblique muscles, probably receive equal innervation increments during supraduction attempts, and this is true whether the eye is straight ahead, abducted, or adducted. That is, if one establishes a horizontal line 30 degrees above the primary position, whether the eye looked at the adduction end, the middle of the line, or the abduction end, any individual vertical muscle has the same innervation. The same applies for infraduction involving the superior oblique and the inferior rectus. Thus, it must follow that the oblique muscles and the rectus muscles establish their field of vertical action primarily on the basis of mechanics, and they do not separate themselves one from another on the basis of differences in innervation reaching them in adduction versus abduction.
We occasionally find a motor unit within, say, the inferior rectus that changes its innervation in the downgaze plane slightly, increasing slightly in abduction, but decreasing with adduction. However, just the reverse of this may be seen in another patient. Consequently, minor variations from the general rule occur. Perhaps these variations act to correct differences in anatomy from one person to another. We know oblique muscle planes are at 30 degrees to the visual axis in one person and 50 degrees in another; perhaps these variations represent individual innervational adjustments that develop during infancy and visual development to achieve concomitance. These variations may have been the basis of the anecdotal reports of difference in vertical muscle innervation in abduction vs abduction.21 Data on the innervational change of vertically acting eye muscles with gaze are few and a careful systematic study remains to be done. For vertical fusion movements, evidence from the torsional position of the eye indicates that the oblique muscles are the primary movers, not the vertical rectus muscles,22,23 although electromyographic (EMG) studies remain to be done. Vertical supraduction during forced blinking (Bell’s phenomenon) appears to be carried out primarily by the oblique muscles.
AMPLITUDE OF ROTATION OF THE EYE
In normal children, the eye can rotate from the primary position about 45 to 50 degrees horizontally and downward and about 40 to 45 degrees upward. This 45 to 50 degrees of movement requires about 10 mm of contraction and 10 mm of relaxation from the primary position (4.8 degrees of rotation equals 1 mm of distance on the sclera). Supraduction diminishes with age. It is reduced a bit even by age 25 and averages only 16 degrees of supraduction at age 80. Other directions of gaze persist better. Exercise helps.24 Donders pointed out that the eyes of myopic people rotate less than this.2 The center of rotation is more posterior in myopic people, effectively increasing the radius of the eye as it rotates and thus giving less rotation for each millimeter of muscle shortening. On the contrary, microphthalmic eyes can often turn more—up to 70 degrees abduction!
FINE STRUCTURE OF MUSCLES
We know from human EMG study that eye muscles are constantly active when awake; therefore, they need much stamina. Eye position can be accurately maintained; this requires fine control. Rapid eye movements are possible: this requires high speed of muscle contraction. These attributes—precision, speed, and stamina—are physiologic properties of a motor unit reflected in the structure of the muscle fibers that comprise it. Using various histologic criteria, anatomists are able to subdivide at least five types of muscle fibers in the extraocular muscles.25
MUSCLE LAYERS
Almost all investigators find that the extraocular muscles of mammals are divided into two layers to some extent: a global or central layer, next to the eye, and a superficial or orbital layer confined to the posterior two thirds of the rectus muscles and less evident in oblique muscles. This layering is much less obvious in humans than lower species (Fig. 17). The orbital layer has a high capillary density and generally contains small fibers. Scott and Collins confirmed that, in humans, fibers that are active constantly for fixation near the primary position are predominantly in this layer.26 By the classification of Alvarado and Van Horn in cat, the orbital layer contains two fiber types.27 Both are relatively small, rich in mitochondria, and contain some lipid vacuoles. About 60% to 70% of the cells are singly innervated with good sarcoplasmic reticulum content, which allows rapid calcium ion changes with each contraction, and about 30% to 40% are multiply innervated slower fibers.
 Fig. 17. Section through the middle of rabbit superior rectus muscle. The orbital layer (top) of smaller fibers is obvious. |
The global layer contains one type of multiply innervated fiber and three types of single innervated fibers of varying size, mitochondrial supply, and sarcoplasmic reticulum content. Type 1 is a large, pale, relatively mitochondria-free type rich in endoplasmic reticulum. This should be a very fast but readily fatigued fiber, good for saccades and for short periods of extreme gaze. These fibers constitute about 30% to 40% of the total number, but they occupy about 60% of the core area and thus contribute importantly in terms of strong but temporary force production. Types 2 and 3 are progressively smaller and contain increasing amounts of mitochondria and probably decreasing amounts of endoplasmic reticulum. These would be more fatigue resistant and slower in response than those that contain a great deal of cytoplasmic reticulum.
MUSCLE FIBER ORIENTATION
The muscle fibers are mostly parallel, but some side-to-side and end-to-side junctions of muscle fibers exist, creating a network within the global layer. Many fibers thus terminate before the end of the muscle; a cross section at the center shows more fibers than cross sections near either end. Oh and Demer have recently counted the fibers in the rectus muscles of monkeys and humans and confirm this restriction of the orbital layer to the posterior two thirds of the muscle.28 Surprising was the finding of insertion of some of these orbital fibers onto the fibromuscular tissue (“pulley”) in Tenon’s fascia near the anterior one third of the muscle.
FASCIA IN MUSCLES
The endomysium surrounding these fibers contains substantial elastic tissue. The perimysium surrounding groups of muscle fibers contains more collagen fibers. Within this area run the small vessels and nerves to the muscle. These collagen fibers run along the length of the muscle; thus, their shortening by inflammation, scar or lack of being stretched by antagonist contraction (e.g., in paralysis) results in stiffening and restriction of eye movement, a common clinical problem.
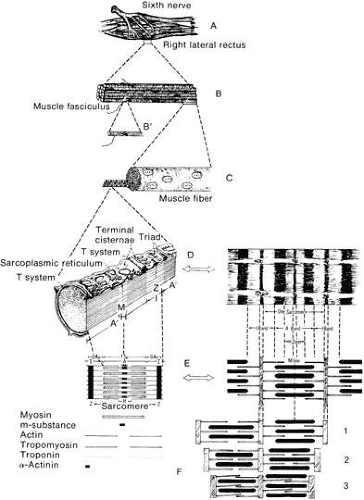
MUSCLE CONTRACTION
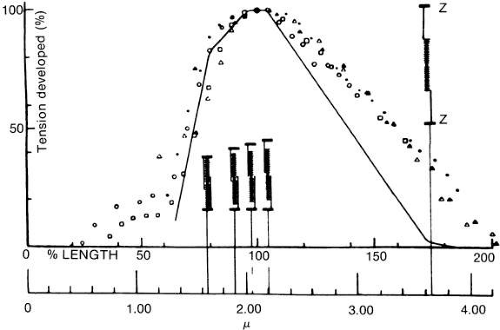
In Figure 18, the muscle is diagrammatically dissected into its components. The heavy filaments are myosin; the thin filaments, a double helix of actin with additional proteins, tropomyosin, and troponin. The sliding of these filaments past one another provides length change, especially apparent in Figure 19. As thick and thin filaments of the muscle fiber slide past one another, the bridges between these filaments maintain the strength of the muscle. If there were many crossbridges, there would be more force, and if there were fewer crossbridges, there would be less force, as seen in isometric testing. In Figure 20, one can see that as the muscle is normally stretched, the number of cross bridges being effectively used maximumizes and then gradually diminishes as further stretching occurs. Unlike many muscles that work over a large range, extraocular muscles work almost entirely in the region from 1 to 2.2; that is, the rather linear slope to the left of the peak of tension. To work within this range, sarcomeres of muscles cannot be greatly stretched or compressed from their normal length of about 2.3 μm. When muscles are maintained at a stretched or shortened length for weeks or months, they respond by adding or removing sarcomeres to regain this length of 2.3 μm. Muscles so shortened become stiff also and cannot be distinguished mechanically from fascial or fibrotic contraction.29
DIFFERENCES IN TENSION
Both singly and multiply innervated types of eye muscle fibers are composed of thick and thin filaments of about the same length. Thus, it is not surprising that they give about the same total force for each cross-sectional area.30
THE T SYSTEM
How can a wave of depolarization sweep through an eye muscle and cause a twitch in 2 msec? Invaginations of the muscle membrane, the sarcolemma, extend into the muscle fiber in a direction transverse (t) to the axis of the fibrils, carrying the wave of polarization into the muscle itself. The sarcoplasmic reticulum that stores calcium necessary for activation is associated directly with these transverse tubules. Much T-system and sarcoplasmic reticulum is present in eye muscles to allow very rapid contraction frequencies up to 400 Hz.
MITOCHONDRIA
Mitochondria provide power to the muscle by oxidative phosphorylation. Because muscle contraction is powered by dephosphorylation of adenosine triphosphate, mitochondria are one source of energy. Thus, the more mitochondria present, the more power and stamina are provided. The extraocular muscles have an extremely high content of oxidative enzymes associated with the mitochondria.
SINGLE AND MULTIPLE INNERVATION
In certain eye muscle fibers, the filaments are separated into bundles. Obvious under light microscopy, this fibrillar pattern (Fibrillenstruktur) is generally associated with rapid, twitch-type contraction. A horizontal view of such a fiber (Fig. 21) reveals the extensive mitochondrial involvement and the differentiation into small bundles. Most of these fibers have single nerve innervation by only one motor endplate. Such a fiber is adapted for rapid, short-term use. In other fibers, the fibrils are not divided much into separate bundles, giving a rather uniform field of distribution (Felderstruktur). Some of these muscle fibers contract only in local areas of the muscle by local innervation around multiple nerve endings along the fiber, without an overall depolarization and twitch. In Figure 22, such a fiber is seen in cross section, again emphasizing the lack of separation into myofibrils. Such fibers would be less capable of high frequence contraction but more capable of slow, steady, stamina-oriented tasks.
BLOOD FLOW AND FATIGUE
Blood flow in the extraocular muscles must be exceedingly high to account for the rapid metabolism. Indeed, average blood flow per gram of extraocular muscles proportionally to that of other skeletal muscles and was second only to myocardium.31 Eye muscles are very resistant to fatigue under laboratory conditions. Extreme gaze to one side for several minutes, however, gives rise to unstable fixation in humans (end-gaze nystagmus). This is unequal in the two eyes, so it appears to be a fatigue of the muscles or motor units rather than of gaze function.
CHANGES IN EXTRAOCULAR MUSCLE WITH AGE AND DISEASE
Miller found alterations in staining characteristics, occasionally degenerating nerve fibers, and disturbance of the orderly fiber alignment and direction of human muscles after age 30.32 With age, the collagen fibers within the muscle itself increased. As motor nerve fibers drop out with neuron death, the remaining nerve fibers send sprouts to form new neuromuscular endings on the denervated muscle fibers. This process preserves muscle strength with aging. A similar sprouting in partial nerve damage leads to prolonged recovery with large motor units. The different types of muscle fiber are probably susceptible to diseases at varying rates. For example, muscular dystrophies associated with mitochondrial abnormalities should especially involve extraocular muscles. Cogan and colleagues have proposed a susceptibility difference seen also in myasthenia gravis.33
THE MOTOR UNIT
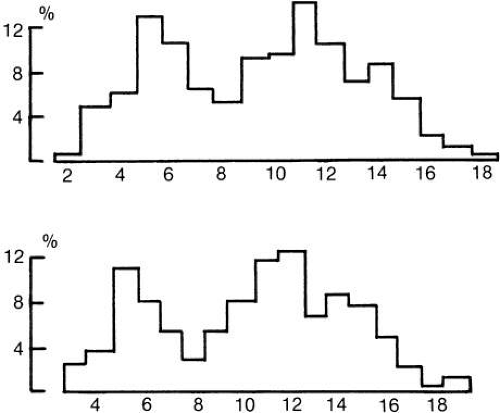
The motor neuron cell body, axon fibers, and muscle fibers that they supply are collectively called a motor unit.34 In skeletal muscles, the muscle fibers comprising a motor unit are spread widely about in the muscle, perhaps to even out mechanical pull. In eye muscles, however, the muscle fibers in a single unit are probably in rather close bundles.35 Tiny movements of a recording electrode within an eye muscle markedly reduce recorded activity of a particular unit, a finding inconsistent with a very “spread-out” distribution. Much of our understanding of motor unit physiology comes through EMG. When the usual EMG electrode records muscle activity, it picks up extracellular signals of twitch-type muscle fibers. Changes in muscle membrane potential that remain local and are not propagated might escape detection; we might thus fail to understand the function of some multi-innervated fibers. However, investigators find a pattern of activity in the ocular motor nuclei (where all these motor units are represented) that correlates well with the EMG pattern found in humans.36,37 The number of muscle fibers supplied by any particular nerve fiber varies; one nerve fiber may supply as many as 140 skeletal muscle fibers. On the average in human extraocular muscles, the ratio has been estimated to be about one nerve fiber to six muscle fibers. Distribution of the nerve fiber sizes in nerves to human extraocular muscles is a bimodal (Fig. 23).38 Muehlendyck has calculated the innervation ratio of the larger muscle fibers and that of the smaller muscle fibers on the basis of the assumption that larger nerve fibers go to the larger muscle fibers and small nerve fibers to the small muscle fibers.39 This ratio is one nerve fiber for each three or four small muscle fibers, good for fine tuning of position, and one nerve fiber to ten or more layer muscle fibers, good to provide the force for saccades.
It would be advantageous to employ physically small motor units to grade with accuracy the amount of contraction or relaxation around the primary position. On the contrary, it would be greatly advantageous for a rapid saccadic eye movement to recruit a motor unit large in size (not only is the number of fibers in the fast unit greater, but their size is also much larger), and thus tension would rise rapidly for the saccadic eye movement. On the basis of these considerations, Robinson’s scheme indicates the possible participation of motor units of different muscle fiber anatomy as the eye moves from out of the field of action of the particular muscle (“off”) into the field of action of the muscle (“on”) (Fig. 24).40 This is consistent with the data found in humans as reported by Scott and Collins.41
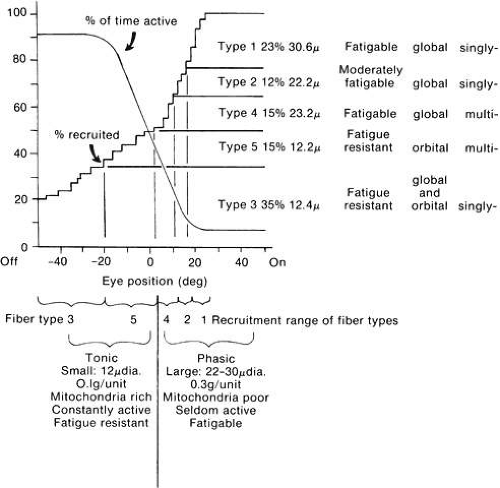 Fig. 24. Hypothetic functional arrangement of eye muscle fiber types. Data on five types taken from Alvarado and Van Horn27 are at the upper right: percentage of fibers of each type, their average diameter, their fatigability, their location in global or orbital layers, and whether they are singly or multiply innervated (Peachy L: In Bach-y-Rita P, Collins CC, Hyde JE [eds]: The Control of Eye Movements. New York: Academic Press, 1971). Upper left, one curve indicates percentage of motor units recruited into activity for any angle of gaze. The other curve estimates percentage of time (e.g., in a day’s activity) that a motor unit is active for units recruited at different thresholds. Below this are indicated eye position ranges over which various fiber types are recruited. It is proposed that type 3 is recruited first, type 1 last, and the two multiply innervated types, 4 and 5, around the primary position, and that the most useful definition of tonic and phasic is to distinguish fatigable from fatigue-resistant fibers. This in turn means that fibers that are recruited below the primary position are tonic, and those above, phasic. (Robinson DA: Personal communication, 1977.) |
PURPOSE AND TYPES OF EYE MOVEMENTS
The successful use of vision comes, in part, from being able to control gaze, that is the position in space to which the eyes are pointed. Gaze results from the sum of body, head-on-body, and eye-in-head movement. Primates, and humans in particular, show great variability in the relative mix of body, head, and eye movement used to establish gaze. A normal person told not to move the head makes perfectly good gaze changes to targets within the orbital range using eye movement only. In contrast, gaze control in nonhumans is more stereotypical and almost always involves combined eye and head movement. The human oculomotor system shares, to some degree, gaze-controlling strategies with the most primitive vertebrate forms and thus reflects a long evolutionary history. A handful of interacting oculomotor subsystems exists, each with its own purpose. The vestibular system compensates for movements of the head (the vestibulo-ocular reflex or VOR). The optokinetic system compensates for motion of the visual surround. The smooth pursuit system, an additional visual tracking system, moves the eyes to track a small visible target smoothly. The saccadic system abruptly moves the eyes from one point in the orbit to another. The vergence system aligns the visual axes of the eyes on the target as the target’s distance changes. The fixation system holds the eye on a stationary target. Natural eye movement is a simultaneous integration of these basic types.
The smooth pursuit, vestibular, optokinetic and saccadic systems produce conjugate eye movements: both eyes move similarly. The vergence system, in contrast, produces disjunctive eye movements: each eye moves differently. Assessment of eye movement behavior would be simple if, for each oculomotor subsystems, a strict relation could be made between an eye movement type and its adequate stimulus. Unfortunately, eye movements result from a complex interaction of physical stimuli, which often activate multiple subsystems, and unique psychological factors, which modulate responses to the same stimulus. Furthermore, no physical stimulus is necessary to make some types of eye movements. Saccades, for example can be executed in a goal-directed fashion voluntarily in the dark to imagined targets; smooth pursuit, on the contrary, cannot be executed without a moving target by most. The psychological factors involved are often ill-defined and beyond rigorous control. Not too surprisingly then, in the clinic, assessing intrinsic oculomotor capability poses a formidable challenge. Some patients may have difficulty in understanding directions, others may have difficulty concentrating on the task at hand and others may be less willing to cooperate. In all these particulars, children are especially troublesome.
EYE MOVEMENT PSYCHOPHYSICS
The purpose of this section is to survey overall oculomotor capacity ignorant of the underlying biologic machine.
VESTIBULO-OCULAR REFLEX
Except for those virtually eyeless animals, all vertebrates possess the standard six extraocular muscles, whether lateral or frontal eyed, developed for roles as predator or prey.42 They all make reflexive, compensatory eye movements in response to a movement of the head. All three sorts of natural head movement are more or less compensated: head rotation activates the angular VOR (AVOR); head translation activates the linear VOR (LVOR); and head tilt in a coronal plane activates the torsional VOR (TVOR). Associated eye movements are compensatory because, for example, to a rightward head movement, the eyes move reflexively leftward in an attempt to steady the visual image on the retina.
There are numerous variations on this theme. Usually, VOR performance is better when combined with visual cues. This reflex response is referred to as visually enhanced VOR (VVOR). When the result of a deliberate head movement, the VOR is called active, or passive when the result of external forces moving the head. Active and passive VORs may differ given that the former can be influenced simultaneously by the cervico-ocular reflex and perhaps by internal command signals (efference copy), but the effect of either in normals appears small and is not usually considered.43,44 Active or passive head movement can be either transient, that is, the head moves from one steady position (or velocity) to another as, for example, one might orient toward something far to one side, or oscillatory as, for example, one might track an insect flying nearby. Additionally, the head (and body) can be passively rotated. Such continuous rotation in one direction is unnatural for humans but not necessarily so for fish swimming in tight circles or being carried by swirling currents.
Vestibular Stimulation in Darkness
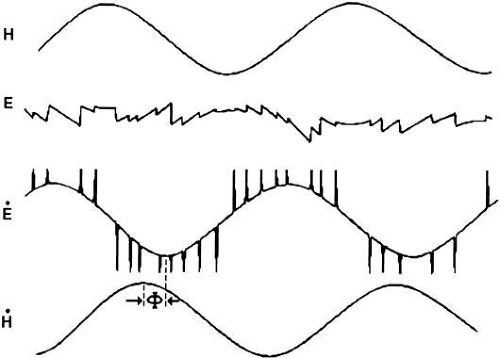
The rationale behind using sinusoidal stimuli is that theoretically, any more complicated head movement can be represented by a sum of sinusoidals, the so-called Fourier expansion, and so if superposition* would hold for the VOR, the response to any complex stimulus can be predicted. Indeed, superposition has been shown to hold for the VOR in darkness.45 Figure 25 depicts the eye movements measured during large-amplitude sinusoidal head rotation in darkness. As shown in trace E of that figure, segments of compensatory eye movement (slow phases) are interrupted from time to time by abrupt resetting eye movements (quick phases) in the opposite direction. The composite behavior is called vestibular nystagmus. (Nystagmus derives from the Greek word meaning “to doze,” which is in this sense extended to eye movements because of the similarity to the characteristic diphasic head bobbing seen as one dozes while sitting upright.)
VOR efficacy can be assessed using various methods. A perfectly compensatory VOR would have the eye rotate in the orbit by an amount equal to, but opposite, the head movement: images of visual targets would be locked on the retina. Because of the ensuing nystagmus, comparing eye excursion, as in trace E, with head excursion would provide a poor measure. From eye position data, cumulative slow phase position can be derived by first extracting the quick phases.46 A simpler method compares eye velocity (E) to head velocity (H). By comparing traces (ignoring those spikes arising from the quick phases) and H, for example, the compensatory nature of the VOR is easily appreciated (see Fig. 25). If the eye was on target before the head started moving, then a system designed to match eye velocity to head velocity perfectly would also keep the eye on target during head movement. Thus, a measure based on velocity seems reasonable and is widely used. At a given stimulus frequency and amplitude, two metrics, VOR gain and phase, characterize eye movement. Most authors calculate gain by |E|/|H| where |E| and |H| are the magnitude of eye velocity and head velocity, respectively. A perfectly compensatory VOR has a gain of 1. For sinusoidal stimuli, phase measures the relative shift between the E and H signals in degrees. A perfectly compensatory VOR has a phase of 180 degrees. Some authors report phase with respect to 180 degrees (such as Φ in Fig. 25). Phase can be estimated by noting corresponding points on the E and H traces or by direct calculation using mathematic approaches, such as that provided by Fourier analysis. A Bode plot summarizes, in two graphs, the VOR gain and phase measurements plotted against frequency.
During small-amplitude oscillation in the dark, the occurrence of quick phases depends on the subject’s instructions. Quick phases were absent when study subjects were asked to imagine a stationary target but occurred often when these people performed mental arithmetic.47 Quick phases, therefore, are not intrinsically linked to vestibular stimulation.
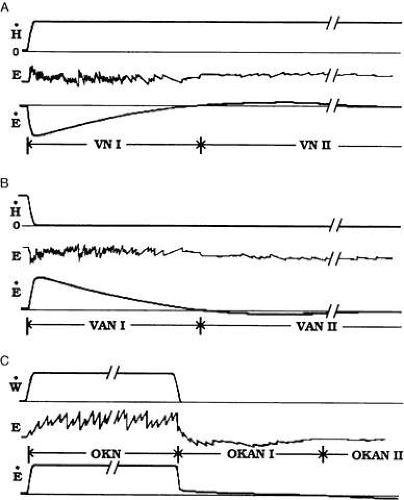
Other aspects of the VOR are revealed by rotating a normal, upright human subject continuously at a constant velocity about an earth-vertical axis in darkness. This elicits an initially brisk vestibular nystagmus (Fig. 26A). As rotation progresses, the slow phase velocity of the per-rotatory nystagmus decreases to 0 following a roughly exponential time course (see VN I in Fig. 26A). The exponential time constant, Tvor, measures about 10–15 seconds. In many subjects a small-amplitude nystagmus builds and decays anew with anticompensatory slow phases (see VN II in Fig. 26A). When rotation ceases, postrotatory nystagmus starts with slow phases directed oppositely to the previous, per-rotatory slow phases (see Fig. 26B). Slow phase velocity during postrotatory nystagmus follows a time course that looks much like the mirror image of the per-rotatory pattern. Many subjects demonstrate a late reversed postrotatory nystagmus, as well.
Efficiency of the VOR
Of all oculomotor subsystems, the VOR responds most quickly to stimulation: eye movement commences within 14 msec after head movement.48 VOR gain is rarely unity and unfortunately, VOR gain not only depends on stimulus parameters but also the subject’s mind set. For example, VOR gain in the dark for passive sinusoidal head movement at 0.3 Hz measured 0.94 when the subject was instructed to imagine a stationary target but fell to 0.65 when the subject performed mental arithmetic.47 In alert and motivated normal subjects, VOR gain, in darkness, during high velocity, transient head turns, is near unity to about 350 degrees/sec and does not depend on whether the movement was active or passive.49 With higher head velocity, eye velocity tends to increase (and could exceed 500 degrees/sec), in addition to becoming more variable.
Considering that the eyes are set apart and that the axes of eye rotation are not coaxial with the axis of head rotation, to compensate for head movement while the eyes are converged on a near target requires horizontal VOR gain greater than unity to be compensatory, reaching a value near two for targets close to the eye. Such gain increases have been measured.50 This high gain has been shown to depend on convergence rather than accommodation and to decrease with frequency of head shake above 1 or 2 Hz.51
Torsional Vestibulo-Ocular Reflex
Rotation of the head about a naso-occipital axis gives rise to a weak torsional VOR presumably mediated by the utricule. Tosional VOR is conjugate and counterrolls the eyes about 20% of head tilt within 30 degrees of the vertical.53
Vestibulo-Ocular Reflex Plasticity
If not for the adaptability of the VOR, use of correction spectacles would be impossible. Not only do lenses change the focus of visual images, they likewise change, by virtue of their minimizing or magnifying power, the amount of eye movement required to compensate for head movement. As an exaggerated example, if a subject is fitted with spectacle lenses with × 2 power then a 10-degrees/sec compensatory head movement would require a 20-degrees/sec eye movement in the opposite direction, twice the velocity needed without the lenses. VOR plasticity likely did not come about so that correction lenses could be worn but instead imparted an animal with the ongoing capability of using vision to adjust VOR gain as necessary.
VOR Maturation
Eye movement recordings suggest that the VOR in infants differs somewhat from the VOR in adults. The VOR time constant in newborns is short, less than 1 second compared with about 10 seconds in adults, approaching adult levels within 3 months after birth.54
OPTOKINETIC SYSTEM
Optokinetic eye movements, by definition, are driven by movement in the visual scene. Thus, all oculomotor subsystems except the VOR come into play when tracking a visual target with the head stationary. One of these subsystems, the optokinetic system (OKS) evolved early in vertebrate history in fish55 and supplements the VOR by eliciting compensatory eye movements in response to large-field retinal stimulation, the rationale perhaps being that whenever the images of most everything visible move within a “stationary” world, then most likely the eyes moved in space rather than the other way around. To a suddenly presented stimulus such as a full visual field of horizontally translating vertical stripes, the optokinetic response is the rapid onset of optokinetic nystagmus (OKN) with slow phases in the direction of stripe motion (Fig. 26C). In humans, peak slow phase eye velocity is reached during the first slow phase.56 As long as the stimulation continues, OKN continues unabated. The ratio of slow phase velocity to stimulus velocity defines OKN gain, which is close to unity for stimulus velocity up to 60 to 90 degrees/sec. Instructing subjects to pick out and follow stripes (look mode) results in large amplitude, low frequency (more than 40 degrees at about 1 Hz) nystagmus while instructing subjects to stare straight ahead results in small amplitude, high frequency (less than 20 degrees at about 2 Hz) nystagmus.57 Without instruction, most subjects naturally adopt the stare mode. If all lights are extinguished during OKN, nystagmus, in most subjects, does not stop entirely but continues as optokinetic after-nystagmus (OKAN) with an attenuated slow phase velocity that slowly decays away in about 1 minute (see Fig. 26C). The initial OKAN slow phase velocity depends not just on the OKN stimulus velocity but also on the OKN stimulus duration; longer duration OKN stimulation (up to 1 minute) leads to higher velocity slow phases during OKAN. The latter property has been called velocity storage.56 Two integrated signal pathways have been postulated to explain OKN and OKAN. The so-called “direct” signal pathway produces the nearly instantaneous, rapid rise of eye velocity at stimulus onset. The so-called “indirect” signal pathway slowly charges the velocity storage element during OKN and discharges the “stored velocity” during OKAN.58 If OKN stimulation is prolonged, OKAN, which beats in the same direction as the OKN, is followed in some subjects by optokinetic after-after nystagmus (OKAAN), which beats in the opposite direction (Fig. 26C). (There is no agreed upon terminology of optokinetic after-nystagmus. Other terms for OKAN include primary OKAN and OKAN I and other terms for OKAAN include secondary OKAN, reversed OKAN, and OKAN II.) OKAAN is thought to reflect OKS habituation.59 Interestingly, labrynthine disease abolishes OKAN when the optokinetic stimulus produced OKN with fast phases toward the affected ear.60 A particularly curious observation is the sensation perceived during full-field optokinetic stimulation. At stimulus onset human subjects report that only the drum is moving but after a short while most subjects report the sensation of slow self rotation in a direction opposite to the drum’s rotation, a phenomenon called circular vection. Circular vection builds until the subject is convinced that the drum is stationary and it is the subject who is rotating exclusively. Nothing in the OKN record, however, reflects the development of circular vection.
SACCADIC SYSTEM
The saccadic system generates all reorienting eye movements. When these eye movements are used to acquire novel visual targets or correct for positioning inaccuracies and drift they are referred to as saccades. The French word saccade means jerk and appropriately describes the eye’s behavior. When these movements reset the eye during nystagmus, the resulting phenomena are called quick phases. Physiologically, saccades and naturally induced quick phases are not known to be different.61 Saccades are often called fast or rapid eye movements but this seems poor terminology because the vestibular system can generate “slow” phases of comparable velocity.49
Amplitude Range of Saccadic Eye Movement
Normally, saccades can start at any orbital position and go to any other. The smallest voluntary saccade is reported to be about 0.05 degrees.62 The smallest involuntary saccade (termed a microsaccade) measures about two thirds smaller, only about 0.017 degrees.63 In the natural setting, however, eye position errors smaller than about 0.5 degrees may not elicit a saccade at all, despite the detection of such errors by the visual system and the ability of the saccadic system to make tiny saccades.64 Young refers to this as an “indifference threshold.” On average, saccades are much larger than this but are usually less than 15 degrees.65
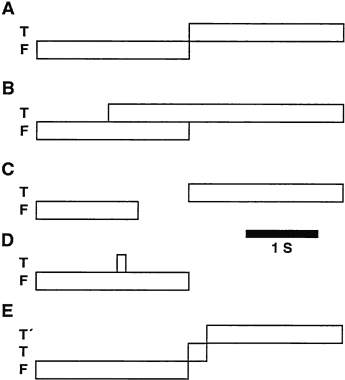
Saccade Waveform
Figure 27 shows the approximate timing of the more common tests used to elicit and study saccades. Each bar in the figure represents the appearance of a small visible light at some position. Each example begins with a subject fixating a small light at position F. In A (step test), the light at F is extinguished and instantaneously the light appears at target position T. The distance between F and T is the step size. Figure 28 depicts a set of normal voluntary horizontal saccades recorded during a step test where the position of T was varied along a horizontal line through F. As seen in Figure 28, horizontal saccades of different sizes are not scale replicas of each other. The early part of the trajectories coincide but larger saccades have longer durations than smaller ones. Not as obvious in Figure 28, larger saccades have higher peak velocities. A plot of average saccadic peak velocity against saccadic amplitude (Fig. 29) reveals a saturating curvilinear function called the saccadic main sequence, a term borrowed from the literature of astronomy. The data for saccades less than 20 degrees have been fit by θmax = 684 (1 – e-0.1) where max is peak eye velocity, θmax is saccade amplitude and e is the base of the natural logarithm (
2.718).66 Peak saccadic velocity reaches about 700 degrees/sec for saccades larger than 20 degrees.67 In comparison, 30 degree saccades produced by a rhesus monkey can exceed 900 degrees/sec.68 The velocity profile is somewhat variable from saccade to saccade but typically peak velocity occurs at or slightly before the midway point of the saccade. In additon, Figure 28 illustrates the tendency for the eye to: overshoot the steady state position for small saccades (common for saccades smaller than 10 degrees, see bottom trace of Fig. 28 at 30 msec); and fall short of the target by about 10% during large saccades necessitating an involuntary corrective saccade after the primary saccade (common for saccades larger than 20 degrees, see top trace of Fig. 28 at 230 msec). Careful measurement of eye velocity has shown that after a saccade, with overshoot or not, the eye is usually still moving, thus creating some ambiguity in marking the end of the saccade.69 Indeed, no entirely satisfactory criteria determine the end of a saccade. Often a velocity threshold is applied, taking as the end of the saccade the point at which eye velocity falls below a selected value, for instance, 5 degrees/sec.66 Saccade duration, Tdur, in seconds, is linearly related to saccade amplitude, ΔρgU, in degrees70:
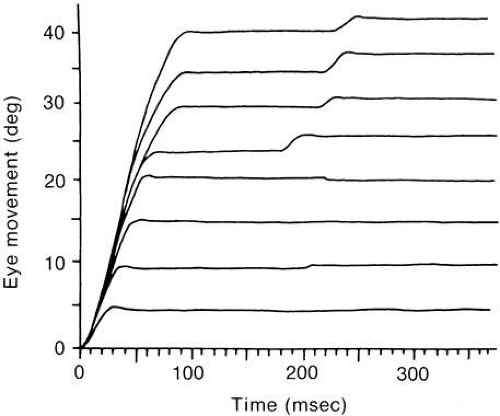 Fig. 27. Common saccade tests. Bars in each case depicts duration of the fixation (F) or target (T or T’) spot. A, Step test; B, Overlap test; C, Gap test; D, Delayed memory; E, Adaptation test. |
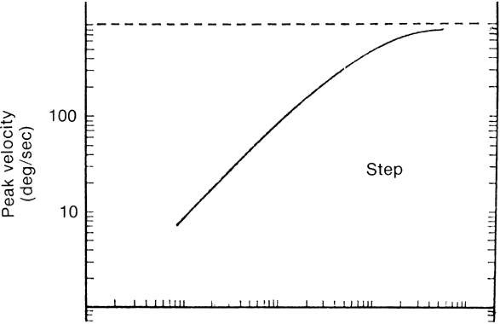 Fig. 28. Superimposed tracings of saccadic eye movements in eight steps from 5 degrees to 40 degrees. (Robinson DA: the mechanics of human saccadic eye movement. J Physiol Lond 174: 245, 1964.) |
 Fig. 29. Main sequence for eye movements. (Bahill T, Clark MR, Stark L: Mathematical Biosci 24:191, 1975.) |
Tdur = 0.0022Δθ + 0.021
A saccade’s step gain is the ratio of the saccade’s amplitude to the target step size and is measured at a point in time after any postsaccadic drift has subsided. Saccades that land immediately on target are called orthometric or normometric. Those falling short of the target are called hypometric whereas those going past the target are called hypermetric. Often a saccade reaches a final position after a period of postsaccadic drift. Slow postsaccadic drift is also called a glissade. Fast postsaccadic drift in a direction opposite to that of the original saccade, as seen in Figure 28, lower trace, is often called dynamic overshoot. Dynamic overshoot and slow drift can both occur after the same saccade.69
The mechanism giving rise to dynamic overshoot in small saccades is moot. The observation that the decaying phase of the overshoot fits on the main sequence has led some researchers to suggest the overshoot is saccadic and results from a deliberate effort to generate gaze shifts in the least amount of time.71,72 The report that the overshoot, when found, is almost exclusively observed in the abducting eye69—an apparent violation of Hering’s law—led to a computer modeling study showing that overshoot in one eye with no overshoot in the other can be explained simply by mechanical differences of the two orbits, that is, the overshoot arises passively.73
The reaction to a stepped target may not be just a single saccade although it commonly is for small saccades. For large target steps, subjects often make two saccades: an initial primary saccade covering about 90% of the displacement followed by a secondary saccade to the target (see Fig. 28, top four traces). Rarely, some normal subjects require a tertiary saccade to acquire the target. Although any saccade that reduces the error between the current eye position and the target could be called a corrective (or correction) saccade, the term is not usually used in referring to the large primary saccade. That corrective saccades persist in total darkness suggests that the corrective saccade is not driven by actual retinal error but is preprogrammed, generated in anticipation of error after the primary saccade.74 Why the primary saccade is ordinarily hypometric is not known but appears purposeful because when feedback is used to move the target position systematically after the saccade is under way to the position that would be normometric for the primary saccade, the saccadic system, after only a few exposures reduces the amplitude of the primary saccade to, in effect, reestablish the primary hypometric saccade.75 Corrective saccades lie on the main sequence suggesting they are executed using the same machinery as the primary saccades.
Saccade Latency and Refractoriness
When subjects are repeatedly tested with unpredictable saccadic targets, the distribution of latency between the appearance of the target and the start of the saccade typically is multimodal. Three latency modes are recognized: the shortest peak between 100 and 135 msec, express saccades; a second one between 140 and 180 msec, fast regular saccades, and a less common third peak about 200 msec, slow regular. Saccades with shorter latency are typically considered anticipatory. The relative frequency in each mode depends on the specific saccade test. If the duration of F in Figure 27A is random, the eye, after an average latency interval of about 200 msec makes a saccadic eye movement toward T.76 Many factors influence saccadic latency, some involve the physical properties of the target, others involve cues to predict a target’s future movement from its past movement. For random target steps larger than 1 degree, saccadic latency is largely independent of saccade amplitude with only small increases for large (e.g., more than 40 degree) saccades.77 For random target steps less than 1 degree, latency (and its variability) dramatically increases to about 400 msec for a 0.06 to 0.2 degree target step.78 When subjects can anticipate an upcoming saccade, especially its direction, latencies are reduced. In an extreme case in which the target is stepped back and forth between two points repeatedly, subjects predict the target motion and can “track” with almost no latency at all.79
If the light at F is held on after the target appears at T (overlap test, see Fig. 27B), the average saccadic latency is longer than when no overlap exists.81 Alternatively, inserting a short interval of time (100 to 200 msec) between the offset of light at F and the appearance of T (gap test, Fig. 27C), decreases the average saccadic latency of most subjects because the express saccades are favored, even when anticipatory saccades are removed.80 Making target selection harder by presenting T along with other potential targets increases average latency and eliminates express saccades.81
The sequence in Figure 27D can test for short-term memory in the saccadic system. Here the light at T appears transiently whereas F remains visible. The subject is usually instructed to look to the remembered position of T as soon as F goes out. Subjects can be instructed to make prosaccades to the target or antisaccades opposite the target. An antisaccades task tests a subject’s ability to override visual grasp reflexes. In the clinic, saccadic latency to visual targets may provide a sensitive indicator of optic neuropathy where, for example, above normal latency has been correlated with afferent conduction loss.82
Oblique Saccades
Oblique saccades in humans are normally curved83 with the horizontal and vertical components generally synchronized.84 Curiously, the duration of the orthogonal components usually differs: for saccades smaller than 15 degrees the horizontal and vertical component durations seem to be independent84,85 but for saccades larger than 15 degrees the shorter duration component is stretched toward the longer duration component.84 Rhesus monkeys, in contrast, tend to make straight oblique saccades.86
Saccadic Plasticity
Saccades are so fast that visual feedback is not possible during saccade execution to ensure accuracy. Instead, the brain uses postsaccadic visual cues to guide adjustments in the preprogramming of future saccades: both static errors (hypo- or hypermetric saccades) and dynamic errors (excessive postsaccadic drift) can be eliminated.87 Adaptation to static errors can be studied with the sequence in Figure 27E. Here the detection by circuitry of the saccade toward T triggers a second jump of the target to position T’. When T’ is sufficiently close to T subjects remain consciously unaware of the second jump despite generation of appropriate corrective saccades. If this test is repeated over and over, the brain gradually adjusts the saccade to approximate the saccade previously made to a target step from F directly to T’.
Vision during Saccades
As we look around a continuously lit scene we do not sense the smeared retinal image caused by the eye’s high velocity during saccades. When scene illumination is confined entirely to the duration of the saccade, however, a smeared image is indeed perceived.88 Experiments wherein patterned visual stimuli were flashed only during saccades have shown that detection thresholds were raised a mere 0.5 log units above the detection threshold when the stimuli were presented at fixation.89 Thus, vision is not blanked during saccades. Other experiments have demonstrated that clear images presented before and after a saccade mask the appreciation of image smear during the saccade.92
SMOOTH PURSUIT SYSTEM
When using eye movements only, a normal subject tracks a small moving target with a mixture of smooth pursuit, saccadic, and vergence eye movements. The overall strategy is to maximize acuity by keeping the target close to the fovea while reducing retinal image slip. For a particular subject, the relative mix of smooth and saccadic eye movement to track a target moving on a uniform background depends primarily on target velocity and regularity. The smooth pursuit subsystem (SPS) creates the conjugate smooth pursuit component. SPS gain, the ratio of eye velocity to target velocity, seldom is unity so that as the eye drifts away from the target saccades are triggered to reduce the accumulated error.91 SPS gain decreases with increasing target velocity, increasing target eccentricity with respect to the fovea, decreasing target movement predictability and decreasing attention. SPS gain is also lower when the moving target is viewed against a stationary structured background.90 With an alert subject, as SPS gain decreases for whatever reason, the frequency of catch-up saccades increases, thus keeping the target close to the fovea.
Higher SPS gain when tracking predictably as opposed to randomly moving targets hints at a fundamentally complicated system: a simpler control system would depend only on the target’s instantaneous kinematics. Further along this same line, the observation that the SPS can be, to some extent and with practice, driven by perception in lieu of retinal image motion along the pursuit direction92,93 has not yet been explored beyond psychophysics and is too often ignored. Reliance on an internal model of target motion probably stems from the somewhat longer time needed by the visual system to process motion cues. Generally, though, voluntary smooth pursuit requires some motion cues, either visual, proprioceptive, or auditory. Nevertheless, target image slip on the retina is considered the primary input to the SPS but constant retinal error can be an adequate stimulus as well.94,95
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree