Purpose
To analyze occurrence, risk factors, and course of ocular hypotony (OH) in juvenile idiopathic arthritis–associated uveitis (JIAU).
Design
Cohort study.
Methods
Epidemiologic and ophthalmologic data at baseline and during follow-up of JIAU patients with and without ocular hypotony were evaluated.
Results
OH developed in 57 of the 365 JIAU patients during the follow-up (mean 4.5 ± 3.5 years). In 40 patients with follow-up ≥12 months, OH was unrelated to previous ocular surgery: risk factors at baseline (univariate logistic regression analysis) included longer total duration of uveitis (odds ratio [OR] 1.13, P < .001), bilateral uveitis (OR 3.51, P = .009), low visual acuity (OR 5.1, P = .001), high laser-flare (LF) values (OR 1.74, P = .01), and presence of posterior synechiae (OR 3.28, P = .004). Increased anterior chamber (AC) cell and LF values were observed within 3 months prior to onset of transient (≤3 months; 37.5%) or persistent OH (>3 months; 62.5%). AC cell and LF values decreased within 3 months after onset of transient OH, while LF levels remained elevated ≥12 months in persistent OH. Optic disc edema and epiretinal membrane formation was found more frequently after OH onset.
Conclusions
OH was observed in 15.6% of JIAU patients. Longer total uveitis duration, bilateral uveitis, low visual acuity, high AC flare and LF grades, and presence of posterior synechiae at baseline were risk factors for subsequent OH. Burden of OH might be improved with immunosuppression.
Juvenile idiopathic arthritis (JIA) is the most common systemic disease associated with childhood uveitis. In previous publications, the incidence of JIA-associated uveitis (JIAU) varied between 9% and 25%. Vision-threatening complications related to JIAU have been reported in 23%–67% of cases, including cataract, glaucoma, macular edema, optic disc edema, and epiretinal membranes. Ocular hypotony (OH) associated with uveitis was noted in about 3%–9% of these patients, supposedly secondary to inflammation, ciliary traction, surgery, and atrophic processes. Different pathophysiological mechanisms underlying OH have been discussed previously. An increased uveoscleral outflow (eg, owing to elevated levels of prostaglandins during the acute inflammatory processes) may cause OH. Furthermore, secondary morphologic changes in chronic uveitis, such as cyclitic membranes, impaired ciliary body epithelium, or an atrophic ciliary body owing to chronic and/or ongoing inflammation, may decrease ciliary body secretion.
It would be desirable to determine potential predictors of OH in order to identify JIAU patients at risk already at an early stage. This information could help in optimizing the anti-inflammatory treatment for JIAU patients and potentially prevent OH and its deleterious complication ocular phthisis. The aim of this study was to evaluate occurrence, risk factors, and clinical course of ocular hypotony in a larger cohort of children with JIA-associated uveitis.
Methods
Medical records of all children with JIA-associated uveitis followed up between April 3, 2001 and November 28, 2013 at the Department of Ophthalmology at St. Franziskus Hospital, Muenster, Germany, were retrospectively reviewed. The study was conducted in accordance with the Declaration of Helsinki. Approval from the ethics committee is not required for retrospective chart reviews in Germany. Only patients who were referred to the uveitis center within the first year of uveitis onset were included in this study.
Juvenile idiopathic arthritis was defined in accordance with the International League of Associations for Rheumatology (ILAR). Rheumatologic examinations included a review of systems, laboratory tests (eg, HLA-B27, complete blood count, urinalysis, angiotensin-converting enzyme, rheumatoid factor, and antinuclear antibodies [ANA]), and chest radiograph examination, if appropriate. Patients with uveitis unrelated to JIA were excluded. Uveitis was classified according to the Standardization of Uveitis Nomenclature (SUN) Working Group as anterior, intermediate, posterior, or panuveitis. All data were reported using a standardized protocol, documenting the medical history, unilateral or bilateral disease, duration of uveitis, best-corrected visual acuity (BCVA), slit-lamp examination, tonometry, and funduscopy. The clinical assessment for each patient was verified by a second observer at each visit. BCVA was measured with age-appropriate tests (eg, Lea and E-tests for children <7 years, and numbers and Snellen tests for adolescents) and the results converted to logMAR units for statistical analysis. Intraocular pressure (IOP) was measured using Goldmann tonometry (Haag Streit, Koeniz, Switzerland) or with an iCARE TA01i tonometer (Icare Finland Oy, Vantaa, Finland). In this study, an IOP <6 mm Hg was defined as OH.
The first visit of the patient at the uveitis clinic was defined as baseline. In cases of bilateral OH, the eye with the lower pressure and more severe OH-related ophthalmologic signs was selected for further analysis, whereas in the control group the right eye was always used for the analysis.
Data about the onset, course, and duration of OH were gathered. Therefore, the ophthalmologic findings 12 and 3 months before OH onset, at the onset of OH, and also 3, 6, and 12 months thereafter were evaluated. Uveitis-related structural complications were documented, for example band keratopathy, synechiae and cataract formation, macular edema, epiretinal membrane formation, ocular hypertension (IOP ≥21 mm Hg), and glaucoma (pathologic cupping of the optic disc and/or glaucomatous visual field defects). If macular edema was suspected, it was confirmed by optical coherence tomography (Stratus OCT III; Carl Zeiss Meditec, Jena, Germany) and/or fluorescein angiography (Spectralis HRA-OCT; Heidelberg Engineering, Heidelberg, Germany). Anterior chamber cells (ACC) were graded according to SUN. Active uveitis was defined as the presence of ≥0.5+ cells in the AC. Laser-flare (LF) photometry measurements were performed with a KOWA FM-500 device (Kowa Company, Ltd, Electronics and Optics Division, Tokyo, Japan) as a routine examination at every visit according to the protocol of the Laser-Cell Flare Photometry Medical Advisory Board. LF values were expressed as photon units/millisecond (pu/ms). Patients with low LF (≤20 pu/ms) were distinguished from those with high LF (>20 pu/ms).
Any topical and systemic anti-inflammatory treatments (including dosages) were reported for each visit, as well as all previous ocular surgical procedures. At the occurrence of OH, topical corticosteroids were increased, and systemic corticosteroids and immunosuppression were adjusted to the individual uveitis course at the discretion of the treating ophthalmologists.
Statistical analysis was performed using MedCalc Statistical Software version 15.2 (MedCalc Software, Ostend, Belgium). Normal data distribution was checked by using the Kolmogorov-Smirnov test. The χ 2 test or McNemar test was applied for categorical data, and Student t test, Mann-Whitney U test, or Friedman test were used for statistical analysis of numerical data when appropriate. Correlation between 2 variables was determined by calculating Pearson correlation coefficient r. Univariate and multivariable regression analysis were performed to calculate odds ratios (OR) and the 95% confidence interval (CI) for determining baseline risk factors of OH. A stepwise backward logistic regression was used for multivariable analysis. P values less than .05 were considered to be statistically significant.
Results
Patient Characteristics
A total of 365 patients with JIA-associated uveitis were identified in the uveitis center database. Epidemiologic data for these patients are shown in Table 1 . JIAU patients were predominantly of female sex (69.6%), had early onset of arthritis, and were ANA positive (74.5%), and the majority was classified as having persistent oligoarthritis (73.0%). In 150 patients (41.2%), uveitis-related secondary complications were documented already at baseline, for example, band keratopathy (17.5%), posterior synechiae (3.8%), optic disc edema (10.7%), and epiretinal membrane formation (3.8%). The mean follow-up at our clinic was 4.5 ± 3.5 years. Ocular surgery had been performed in 28 patients (7.7%) before study inclusion and in another 28 patients (14.8%) during the subsequent observation period.
| JIA Uveitis, Total Group | JIA Uveitis, Patients Without Ocular Hypotony | JIA Uveitis, Patients With Ocular Hypotony During Follow-up | P Value a Comparing Patients With and Without Ocular Hypotony | |
|---|---|---|---|---|
| Patients, n (%) | 365 (100) | 308 (84.4) | 57 (15.6) | n/a |
| Female sex, n (%) | 254 (69.6) | 218 (70.8) | 36 (62.7) | .3 |
| Age at baseline, y, mean (SD) | 7.8 (4.1) | 7.7 (4.2) | 8.4 (4.1) | .02 |
| Age at JIA onset, y, mean (SD) | 3.7 (3.1) | 3.5 (3.0) | 4.4 (3.4) | .03 |
| Age at uveitis onset, y, mean (SD) | 5.5 (3.8) | 5.6 (4.0) | 4.8 (2.7) | .06 |
| Duration of uveitis at baseline of study, mo (SD) | 32.4 (41.9) | 30.4 (40.8) | 41.9 (46.1) | .09 |
| Polyarthritis RF positive | 3 (0.8%) | 3 (1.0%) | 0 (0%) | .5 |
| Polyarthritis RF negative | 34 (9.3%) | 29 (9.4%) | 5 (8.6%) | |
| Systemic arthritis | 1 (0.3%) | 0 (0%) | 1 (1.8%) | |
| Oligoarthritis, persistent | 266 (73.0%) | 227 (73.7%) | 39 (68.4%) | |
| Oligoarthritis, extended | 51 (14.0%) | 40 (13.0%) | 11 (19.3%) | |
| Psoriatic arthritis | 2 (0.5%) | 2 (0.6%) | 0 (0%) | |
| Enthesitis-related arthritis | 6 (1.6%) | 6 (1.9%) | 0 (0%) | |
| Undifferentiated arthritis | 2 (0.5%) | 1 (0.3) | 1 (1.8%) | |
| ANA positivity, n | 272 (74.5%) | 227 (73.7%) | 45 (78.9%) | .2 |
| Uveitis-related eye complications | 150 (41.2%) | 108 (35.1%) | 42 (73.7%) | <.001 |
| Previous eye surgery | 28 (7.7%) | 15 (4.9%) | 13 (22.8%) | <.001 |
a Mann-Whitney U test or χ 2 test (for categorical data); P < .05 statistically significant.
Out of 365 JIA patients, OH was reported in 57 patients (15.6%) ( Figure 1 , Table 1 ). Patient demographics did not differ between patients with or without OH occurrence with regard to sex, JIA subtype, ANA positivity, age at uveitis onset, and duration of uveitis at baseline ( P > .05, each), whereas OH patients were older at JIA onset, were older at study baseline, and had more uveitis-related eye complications and previous eye operations ( Table 1 ). The incidence rate for OH was 0.02/person-year in the total group of patients included in the study.
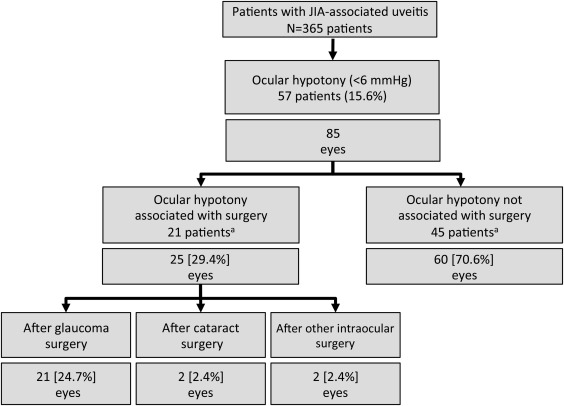
Association of Ocular Hypotony With Ocular Surgery
In 21 patients (25 eyes), OH was observed after previous ocular surgery ( Figure 1 ; 21 eyes after glaucoma surgery, 2 eyes after cataract surgery, and another 2 eyes after other intraocular operations). In these patients, OH was noted at a mean of 5.3 ± 12.4 months following ocular surgery.
Predictors for the Occurrence of Ocular Hypotony in Patients Without Ocular Surgery
To investigate the predictors for the occurrence of OH unrelated to ocular surgery, all JIAU patients who did not have prior surgical ocular interventions within the previous 12 months were further analyzed. Patients with a follow-up of ≥12 months were included in this analysis (40 patients). This group was compared with patients in whom OH did not occur and without previous surgery (293 patients with a follow-up ≥12 months). At baseline, OH patients were also younger at onset of uveitis, but the 2 groups did not differ with respect to sex, JIA type, and ANA positivity. Furthermore, IOP, ACC grade, and lens status were similar in the 2 groups ( Table 2 ).
| Parameter | JIA Uveitis, With Ocular Hypotony Not Associated With Previous Surgery (N = 40) | JIA Uveitis, Without Ocular Hypotony, No Previous Surgery (N = 293) | P Value a | Univariate Regression Analysis b | ||
|---|---|---|---|---|---|---|
| OR | 95% CI | P Value | ||||
| Female sex, n (%) | 26 (65.0) | 194 (66.1) | .9 | 1.06 | 0.48–2.2 | .95 |
| Bilateral uveitis, n (%) | 34 (85.0) | 184 (62.6) | .02 | 3.51 | 1.37–9.04 | .009 |
| Subtype of JIA | >.05 | |||||
| ANA positivity, n (%) | 33 (82.5) | 89 (77.4) | .4 | 3.34 | 0.41–27.37 | .2 |
| Age at JIA onset, y, mean (SD) | 3.5 (2.5) | 3.77 (2.9) | .9 | 0.9 | 0.79–1.02 | .08 |
| Age at uveitis onset, y, mean (SD) | 4.8 (3.0) | 5.9 (3.4) | .02 | 0.9 | 0.79–1.02 | .08 |
| Total uveitis duration (mo) between initial documentation and final follow-up at our institution (SD) | 86.5 (48.5) | 74.1 (3.8) | <.001 | 1.13 | 1.01–1.21 | <.001 |
| Uveitis duration (y) before hypotony, mean (SD) | 9.4 (15.8) | n.a. | ||||
| BCVA (mean logMAR ± SD) | 0.5 (±0.6) | 0.2 (±0.2) | .07 | 5.10 | 1.77–14.75 | .0008 |
| IOP in mm Hg (mean ± SD) | 13.4 (4.9) | 14.0 (2.7) | .2 | 0.95 | 0.85–1.06 | .36 |
| LF, photon units/msec (mean ± SD) c | 58.7 (101.8) | 39.8 (76.4) | .006 | 1.74 | 1.13–2.67 | .013 |
| ACC grade (mean ± SD) d | 0.5 (0.6) | 0.7 (0.9) | .4 | 0.67 | 0.40–1.14 | .11 |
| Anterior chamber flare (mean ± SD) d | 0.9 (1.0) | 0.5 (0.8) | .009 | 1.74 | 1.13–2.67 | .013 |
| Posterior synechiae (%) | 45.0 | 26.1 | .006 | 3.28 | 1.47–7.32 | .004 |
| Topical corticosteroids, n (%) | 27 (67.5) | 192 (65.5) | .8 | 1.2 | 0.55–2.62 | .65 |
| Systemic corticosteroids, n (%) | 11 (27.5) | 43 (14.7) | .1 | 2.27 | 0.95–5.39 | .06 |
| csDMARDs, n (%) | 27 (67.5) | 171 (58.3) | .3 | 1.61 | 0.74–3.50 | .27 |
| bDMARDs, n (%) | 2 (5.0) | 7 (2.41) | .8 | 2.02 | 0.32–12.54 | .46 |
a Mann-Whitney U test or χ 2 test (for categorical data).
b Risk of occurrence of ocular hypotony was calculated with univariate regression analysis, and corresponding odds ratios, 95% confidence intervals, and P values are shown.
Univariate logistic regression analysis found a higher OH risk in patients with bilateral uveitis (OR 3.51, 95% CI 1.37–9.04; P = .009), longer total uveitis duration (OR 1.13, 95% CI 1.01–1.21, P < .001), lower BCVA (OR 5.1, 95% CI 1.77–14.75, P = .0008), higher LF values (OR 1.74, 95% CI 1.13–2.67, P = .013), and presence of posterior synechiae at first examination (OR 3.28, 95% CI 1.47–7.32, P = .004). Results of the univariate regression analysis are shown in Table 2 .
In a multivariable logistic regression analysis, only a younger age at onset of JIA (OR 1.18, 95% CI 1.03–1.36, P = .02), bilaterality of uveitis (OR 5.3, 95% CI 1.16–23.9, P = .03), and poor BCVA at first examination (OR 4.6, 95% CI 1.05–19.7, P = .04) were independently associated with the onset of OH during the course of uveitis in JIA, whereas neither other clinical findings nor topical and systemic treatment at baseline were independently associated with a higher risk for OH ( P > .05, each). No relationships between associated risk factors, generated subgroups, and different lengths of follow-up periods have been found, whereas the risk increased with the lengths of the follow-up period.
Course of Ocular Hypotony Unrelated to Previous Eye Surgery
Eyes with OH not associated with previous surgical procedures revealed a mean IOP of 4.3 ± 2.0 mm Hg at a mean OH duration of 9.5 ± 22.7 months. In 37.5% of these eyes, OH duration was ≤3 months (transient OH) and in another 62.5% >3 months (persistent OH). A total of 4 eyes (8.9%) developed phthisis bulbi after a mean uveitis duration of 32.4 ± 25.2 months and after a mean OH duration of 20.3 ± 10.1 months, respectively.
In an attempt to define predictive factors for transient or persistent OH course, the clinical data at initial documentation at our institution were analyzed. Patients with persistent OH revealed a higher rate of bilateral uveitis (96.0% vs 73.3%; P = .04), interestingly had a higher IOP (14.2 ± 3.7 mm Hg vs 12.1 ± 6.3 mm Hg; P = .04), and had received systemic corticosteroids slightly less frequently (16.0% vs 46.7%; P = .06) at initial examination as compared with the group with transient OH, with the latter not reaching the level of significance. The groups did not differ in sex, type of JIA, age at onset of JIA or JIA-associated uveitis, BCVA, LF values, ACC grade, anterior chamber flare grade, or presence of posterior synechiae ( Table 3 ).
| Parameter | Ocular Hypotony ≤3 Months (N = 15) | Ocular Hypotony >3 Months (N = 25) | P Value a | Univariate Regression Analysis b | ||
|---|---|---|---|---|---|---|
| OR | 95% CI | P Value | ||||
| Female sex, n (%) | 9 (60.0) | 17 (68.0) | .9 | 1.18 | 0.3–4.69 | .81 |
| Bilateral uveitis, n (%) | 11 (73.3) | 24 (96.0) | .04 | 12.0 | 1.24–116.2 | .03 |
| Subtype of JIA | >.05 | |||||
| Age at JIA onset, y, mean (SD) | 3.5 (3.6) | 3.5 (1.8) | .3 | 1.0 | 0.75–1.35 | .98 |
| Age at uveitis onset, y, mean (SD) | 5.6 (3.7) | 4.2 (1.9) | .1 | 1.25 | 0.98–1.60 | .05 |
| Uveitis duration (mo) at initial documentation at institution (SD) | 32.6 (35.1) | 58.9 (57.6) | .07 | 1.0 | 0.97–1.00 | .08 |
| Uveitis duration before hypotony, y, mean (SD) | 2.1 (2.4) | 3.7 (2.8) | .08 | 0.79 | 0.59–1.04 | .1 |
| BCVA (mean logMAR ± SD) | 0.4 (0.5) | 0.5 (0.7) | .5 | 0.76 | 0.24–2.40 | .64 |
| Intraocular pressure in mm Hg (mean ± SD) | 12.1 (6.3) | 14.2 (3.7) | .04 | 0.91 | 0.78–1.06 | .21 |
| LFM, photon units/msec (mean ± SD) c | 81.7 (149.2) | 45.3 (59.8) | .6 | 1.00 | 0.99–1.01 | .33 |
| ACC grade (mean ± SD) d | 0.6 (0.6) | 0.4 (0.5) | .4 | 1.66 | 0.5–5.47 | .41 |
| Anterior chamber flare (mean ± SD) d | 1.1 (0.8) | 0.7 (1.1) | .1 | 1.45 | 0.71–2.97 | .31 |
| Posterior synechiae (%) | 60.0 | 36.0 | .5 | 0.5 | 0.12–2.06 | .34 |
| Topical steroids, n (%) | 10 (66.7) | 17 (68.0) | .9 | 0.85 | 0.2–3.56 | .82 |
| Systemic corticosteroids, n (%) | 7 (46.7) | 4 (16.0) | .06 | 5.25 | 1.18–23.46 | .03 |
| csDMARDs, n (%) | 9 (60) | 18 (72.0) | .9 | 1.43 | 0.35–5.79 | .62 |
| bDMARDs, n (%) | 1 (6.67) | 1 (4.0) | .7 | 1.85 | 0.11–32.0 | .67 |
a Mann-Whitney U test or χ 2 test (for categorical data).
b Univariate regression analysis was performed to assess predicting factors for transient (≤3 months) and persistent (>3 months) ocular hypotony. Odds ratios, 95% confidence intervals, and P values are shown.
Univariate logistic regression analysis revealed a higher risk for transient OH for patients with bilateral onset of uveitis (OR 12.0, 95% CI 1.24–116.2, P = .03) and systemic use of corticosteroids (OR 5.25, 95% CI 1.18–23.46, P = .03). Table 3 summarizes baseline characteristics and results of univariate regression analysis owing to course of ocular hypotony. Multivariable regression analysis did not reveal any significant association of these parameters with the time courses of OH.
Ophthalmologic Findings Before and After Onset of Ocular Hypotony
No significant difference in BCVA values prior to OH and during the subsequent follow-up examinations was found ( Tables 4 and 5 ). The mean ACC grades at the time point of OH onset were higher than at the visits 12 and 3 months before OH developed ( P < .05, each; Table 4 ). In 64.5% of eyes with OH, LF values remained elevated up to 12 months after OH onset.
| 12 Months Before Onset of Hypotony | 3 Months Before Onset of Hypotony | P Value a , b | At Onset of Hypotony | P Value a , c | |
|---|---|---|---|---|---|
| BCVA logMAR (mean ± SD) | 0.41 ± 0.58 | 0.45 ± 0.37 | ns | 0.53 ± 0.54 | ns |
| BCVA loss ≥0.2 logMAR (10 letters) (%) | 30.8 | 25.0 | ns | ||
| IOP in mm Hg (mean ± SD) | 9.9 ± 4.7 | 10.2 ± 3.8 | ns | 4.3 ± 4.8 | <.05 |
| AC cell grade (mean ± SD) | 0.4 ± 0.8 | 0.4 ± 0.6 | ns | 0.9 ± 1.0 | <.05 |
| LFM in photon units/msec (mean ± SD) | 51.9 ± 55.2 | 89.5 ± 139.1 | <.05 | 98.7 ± 124.9 | <.05 |
| LFM change ≥20 (%) | 70.4 | 77.8 | ns | 80.9 | ns |
| Keratic precipitates (%) | 36.0 | 44.4 | ns | 51.2 | ns |
| Posterior synechiae (%) | 52.2 | 47.4 | ns | 61.9 | ns |
| Band keratopathy (%) | 57.7 | 62.2 | ns | 56.4 | ns |
| Cataract formation (%) | 25.8 | 20.8 | ns | 21.4 | ns |
| Aphakia (%) | 19.4 | 20.8 | 14.3 | ||
| IOL (%) | 22.6 | 25.0 | 26.2 | ||
| Iris rubeosis (%) | 0 | 0 | ns | 5.9 | ns |
| Optic disc edema (%) | 31.8 | 37.5 | ns | 55.2 | ns |
| Macular edema (%) | 9.5 | 9.3 | ns | 14.8 | ns |
| Epiretinal membrane formation (%) | 19.1 | 13.3 | ns | 32.3 | ns |
a Friedman test or McNemar test (for categorical data).
b Twelve months prior to OH vs 3 months prior to OH.
| Onset of Hypotony | 3 Months After Onset of Hypotony | P Value a , b | 6 Months After Onset of Hypotony | P Value a , b | 12 Months After Onset of Hypotony | P Value a , b | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Duration of hypotony (mo) | ≤3 | >3 | ≤3 | >3 | ≤3 | >3 | ≤3 | >3 | |||
| BCVA (mean ± SD) | 0.44 ± 0.36 | 0.57 ± 0.61 | 0.49 ± 0.6 | 0.75 ± 0.54 | ns c , d , e | 0.33 ± 0.39 | 0.44 ± 0.53 | ns c , d , e | 0.57 ± 0.7 | 0.61 ± 0.59 | ns c , d , e |
| IOP (mm Hg ± SD) | 4.9 ± 1.5 | 4.0 ± 2.2 | 11.7 ± 3.4 | 5.1 ± 3.5 | <.05 c , e ns d | 11.4 ± 3.7 | 7.4 ± 3.7 | ns c <.05 d , e | 11.45 ± 4.1 | 7.89 ± 4.0 | ns c , e <.05 d |
| ACC (mean ± SD) | 1.1 ± 1.1 | 1.0 ± 1.0 | 0.6 ± 1.1 | 0.9 ± 0.3 | <.05 c , e ns d | 0.3 ± 0.7 | 0.4 ± 0.9 | ns c , e <.05 d | 0.4 ± 1.0 | 0.3 ± 0.5 | ns c , d , e |
| LFM (photon units/msec) (mean ± SD) | 94.4 ± 117.1 | 108.3 ± 129.9 | 72.3 ± 104.8 | 78.9 ± 106.5 | ns c , d , e | 45.4 ± 57.4 | 80.9 ± 108.5 | <.05 c , e ns d | 25.6 ± 16.9 | 81.9 ± 109.2 | <.05 c , e ns d |
| LFM change ≥20 (%) | 57.1 | 18.9 | <.05 e | 0 | 44.4 | <.05 e | 20.0 | 21.4 | ns e | ||
| Keratic precipitates (%) | 57.1 | 48.2 | 40.0 | 45.5 | ns c , d , e | 25.0 | 25.0 | ns c , d , e | 30.0 | 41.2 | ns c , d , e |
| Posterior synechiae (%) | 64.3 | 60.7 | 54.0 | 54.6 | ns c , d , e | 60.0 | 57.6 | ns c , d , e | 58.0 | 57.9 | ns c , d , e |
| Band keratopathy (%) | 41.7 | 62.9 | 43.3 | 62.2 | ns c , d , e | 46.2 | 62.9 | ns c , d , e | 46.4 | 61.1 | ns c , d , e |
| Cataract (%) | 26.7 | 27.9 | 27.0 | 27.3 | ns c , d , e | 26.7 | 26.1 | ns c , d , e | 31.7 | 38.9 | ns c , d , e |
| Iris rubeosis (%) | 4.2 | 6.9 | 0 | 0 | ns c , d | 0 | 0 | ns c , d | 0 | 3.3 | ns c , d |
| Optic disc edema (%) | 40.0 | 63.2 | 30.0 | 58.3 | ns c , d <.05 e | 30.0 | 50.0 | ns c , d <.05 e | 11.11 | 23.08 | ns c , d , e |
| Macular edema (%) | 10.0 | 17.6 | 9.1 | 9.0 | ns c , d | 5.0 | 6.7 | ns c , d | 5.0 | 6.2 | ns c , d |
| Epiretinal membrane (%) | 20.0 | 38.1 | 22.2 | 40.0 | ns c , d <.05 e | 18.2 | 41.2 | ns c , d <.05 e | 30 | 46.7 | ns c , d , e |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


