Fig. 6.1
(a–d) MRI sagittal (a) and coronal (b) and axial (c) images and PET-CT (d) display left nasopharyngeal lesion filling the Rosenmuller fossa invading through the pharyngobasilar fascia and infiltrating the cavernous sinus measuring 17 × 7 × 26 mm
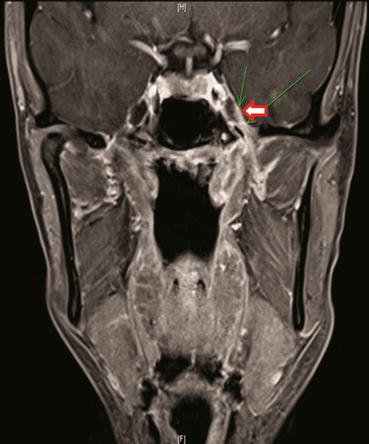
Fig. 6.2
MRI coronal image displays minimally extending intracranially through foramen ovale
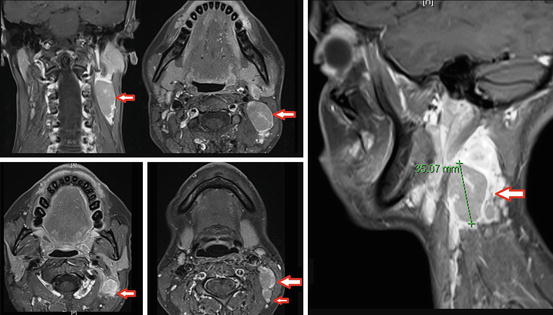
Fig. 6.3
MRI images display bilateral nodal disease, largest on left level 2 measuring 3.5 cm
A biopsy was performed from the left nasopharyngeal lesion confirming the undifferentiated nonkeratinizing carcinoma (WHO Type III).
She was staged as T4N2M0, advanced stage nasopharyngeal cancer .
2 Evidence-Based Treatment Approaches
Radiotherapy (RT) alone is the standard treatment for stage I (Category IB). Concurrent chemoradiotherapy (CRT) is the current standard of care for non-metastatic advanced stage (stages III, IVA, and IVB) nasopharyngeal carcinoma, with or without induction or adjuvant chemotherapy (Category IA). For intermediate-risk disease , stage II, it is recommended to give CRT instead of RT alone (Category 2B).
The MDACC series initially clarified the efficacy of RT alone for T1 category, while RT alone was capable of 5-year local control rates of 93, 79, 68, and 53 % for T1, T2, T3, and T4, respectively. Sanguinetti at al pointed out that advanced T category, squamous histology, and cranial nerve deficits were poor prognostic factors for local control [1].
The major data for combined CRT approach is derived from phase III trial of 230 stage II (T1–2N1M0 or T2N0M0 disease with parapharyngeal space involvement; 13 % is stage III based on AJCC 2010 TNM) nasopharyngeal cancer patients randomized into RT plus concurrent weekly cisplatin (30 mg/m2) versus RT alone [2]. The addition of concurrent cisplatin significantly improved overall survival from 85.8 % for RT alone to 94.5 % for CRT at 5 years; based on improvement in distant metastasis free survival (94.8 % vs. 83.9 %) without any difference in locoregional relapse-free survival (93.0 % vs. 91.1 %).
Addition of chemotherapy to definitive RT increases overall survival by 4–6 % at 5 years while decreases the risk of event by 10 % and the risk of death by 18 %. CRT demonstrated the most pronounced benefit in comparison to induction chemotherapy or adjuvant chemotherapy [3].
Intergroup 0099 (RTOG 8817) trial established a benefit from concurrent (cisplatin 100 mg/m2 on days 1, 22, and 43) and adjuvant (cisplatin 80 mg/m2 on day 1 and fluorouracil 1,000 mg/m2/day, days 1–4, every 4 weeks for three cycles) chemoradiotherapy (70 Gy, 35–39 fractions, 1.8–2.0 Gy/fraction/day) for stage III and IV nasopharyngeal cancer in comparison to RT alone (CRT vs. RT at 3 year, progression-free survival: 69 % vs. 24 %, overall survival: 78 % vs. 47 %; at 5 years in Table 6.1) [4]. Five-year OS and DFS favored CRT arms over RT alone in other Singapore [5], Taiwan [6], Hong Kong [7, 8], and China [9] randomized trials (Table 6.1).
Table 6.1
Prospective randomized trials of concurrent chemoradiotherapy for locally advanced nasopharyngeal cases
Trials | n | Standard arm (RT) | Randomization arm (CRT) | RT | CRT | p | C | CRT | p |
|---|---|---|---|---|---|---|---|---|---|
Disease-free survival | Disease-free survival | Overall survival | Overall survival | ||||||
150 | 70 Gy | 70 Gy with cisplatin + 3 cycles of cisplatin/5FU | 29 % | 58 % | <0.001 | 37 % | 67 % | 0.005 | |
Singapore trial [5] | 221 | 70 Gy | 70 Gy with cisplatin + 3 cycles of cisplatin/5FU | 53 % (3 years) | 72 % (3 years) | 0.01 | 65 % | 80 % (3 years) | 0.01 |
Taiwan trial [6] | 284 | 70–74 Gy | 70–74 Gy with cisplatin | 53 % | 72 % | 0.0012 | (3 years) | 72 % | 0.0022 |
Hong Kong trial [7] | 348 | 66 Gy | 66 Gy with cisplatin + 3 cycles of cisplatin/5FU | 62 % | 72 % | 0.027 | 54 % | 78 % | 0.97 |
China trial [9] | 115 | 70–74 Gy + 10 Gy Boost | 70–74 Gy + 10 Gy Boost with oxaliplatin | 83 % (2 years) | 96 % (2 years) | 0.02 | 77 % | 100 % | 0.01 |
Hong Kong trial [8] | 350 | 66 Gy + 10–20 Gy boost | 66 Gy + 10–20 Gy boost with cisplatin | 52 % | 60 % | NS | 78 % | 70 % | 0.065 |
China trial [11] | 506 | 60–66 Gy with cisplatin | 60–66 Gy with cisplatin + 3 cycles of cisplatin/5FU | 84 % (2 years) | 86 % (2 years) | 0.13 | NA | NA | NS |
Induction chemotherapy followed by RT alone approach has failed to show an overall survival benefit compared to RT alone (Table 6.2), but induction chemotherapy in addition to RT slightly improved relapse-free and disease-specific survival [17]. However, sequential therapy of induction chemotherapy followed by concurrent chemoradiotherapy started to be a popular alternative to concurrent chemoradiotherapy with adjuvant chemotherapy [18]. A recent phase II randomized trial comparing induction chemotherapy with docetaxel and cisplatin followed by weekly cisplatin concomitant with RT to weekly cisplatin concomitant with RT. Aside from similar quality of life scores, a trend toward improved 3-year progression-free survival with sequential therapy (88 % vs. 60 %, p = 0.12) and significant increase in overall survival with sequential therapy (94 % vs. 68 %) were evident [15].
Table 6.2
Prospective randomized trials of neoadjuvant chemotherapy for locally advanced nasopharyngeal cases
Trials | n | Standard arm (RT) | Randomization arm (C→RT) | RT | C→RT | p | C | C→RT | p |
|---|---|---|---|---|---|---|---|---|---|
Disease-free survival | Overall survival | ||||||||
International nasopharyngeal cancer study [12] | 339 | 70 Gy | 3 cycles of cisplatin + epirubicin + bleomycin and then 70 Gy | 40 % (2 years) | 54 % (2 years) | <0.001 | NA | NA | NS |
China study [13] | 456 | 70 Gy | 2–3 cycles of cisplatin + bleomycin + 5FU and then 70 Gy | 49 % | 59 % | 0.05 | 56 % | 63 % | 0.11 |
AOCOA [14] | 334 | 70 Gy | 2–3 cycles of cisplatin + epirubicin and then 70 Gy | 42 % (3 years) | 48 % (3 years) | NS | 71 % (3 years) | 78 % (3 years) | NS |
Hong Kong trial [15] | 65 | 70 Gy with cisplatin | 2 cycles of docetaxel + cisplatin and then 70 Gy with cisplatin | 88.2 % (3 years) | 59.5 % (3 years) | 0.12 | 67.7 % (3 years) | 94.1 % (3 years) | 0.012 |
China trial [16] | 338 | 70 Gy with cisplatin/5FU + 4 cycles of cisplatin/5FU | 2 cycles of cisplatin/5FU, then 70 Gy with cisplatin/5FU + 4 cycles of cisplatin/5FU | 78.5 % | 82.5 % | 0.16 | 95.9 % (3 years) | 94.5 % (3 years) | 0.54 < div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|