Fig. 19.1
Original single-plate Molteno implant, 1967
1.
A stage of hypotony, lasting 10 days.
2.
The hypertensive stage lasting 6–10 weeks.
3.
The stable stage in which the intraocular pressure remained steady or fell slowly. This began after 8–12 weeks.
The hypertensive stage coincided with the formation of a thick-walled hyperemic bleb. Thus the early implants produced bleb pathophysiology, which has continued in all the upgraded implants that have developed over the years. The unchanged factors over the years have been the tissue and the aqueous, and thus the “Epstein factor(s)” continues to play a major role in bleb function related to implant use.
Systemic steroids were used in some of these initial implants. Prednisone 30 mg was given daily beginning 1 week after operation and was maintained in gradually diminishing dosage until the eye was completely quiet after 6–12 weeks.
Systemic steroids had a marked effect in every case giving a shorter hypertensive phase, thinner bleb, and stable intraocular pressure of between 10 and 23 mm. This observation of the use of anti-inflammatory medication was to play a much more significant role in the yet to be redesigned Molteno device. Complications of the initial implant included corneal dystrophy due to the tube as well as uveitis and discomfort from the large anteriorly situated bleb. In older patients the implant was removed usually at 3 months, or later, when the bleb was well established, to avoid the complications listed. Removal was not done in young patients as this resulted in a tendency for the bleb to fibrose. The initial implant had made headway in the treatment of refractory cases of glaucoma, and when the results obtained with the implant were compared to those of the Scheie procedure as an initial procedure, only a small difference in favor of the implant was noted. However, a marked difference in the success obtained with the implant over the Scheie was seen in secondary glaucomas, previously failed glaucoma procedures, and buphthalmic cases. The anterior implant was technically difficult to implant, had to be removed at a later date, and combined with the complications of corneal dystrophy, uveitis, and bleb dysesthesia encouraged Molteno to modify the implant. The availability of medical grade silicon and polypropylene allowed Molteno to develop the long-tube implant, which became the prototype of all the long-tube implants available today.
Long-Tube Implants
The long-tube implant was developed because more suitable materials, polypropylene and silicone, became available. This allowed the delivery system from plate to anterior chamber to become more flexible, which in turn provided the opportunity to move the plate away from the limbal area to a more posterior location [5]. The long-tube implant plate now had a larger plate surface 13.5 mm, resulting in a larger bleb and more drainage. The polypropylene plate and the silicone delivery systems were easy to handle surgically and, more importantly, afforded the opportunity of placement in a quadrant of the eye where paralimbal surgery may not have been possible. Indeed this new design would stand the test of time and become the prototype for a new generation of long-tube implants. The development of the long-tube implant resulted in the introduction of a bleb different from those seen with standard glaucoma surgeries. The function of the tube was to deliver aqueous from the anterior chamber to the surface of the plate, situated away from the locations that had been prevalent with standard glaucoma procedures. The function of the plate was to initiate the presence of a bleb, generally as large or larger than the plate surface. This bleb would then regulate the flow of aqueous to the surrounding conjunctiva, and this in turn would be regulated by the final permeability of the fibrovascular lining of the bleb wall. The implant thus now became simply a conduit for delivering aqueous from the anterior chamber to the subconjunctival space, and the process of bleb formation, as well as surgical aspects related to the implant itself, would become the important aspects which would drive the search for mechanisms to improve the function of these new glaucoma implants. Molteno predominantly, and other researchers to a lesser extent, would become involved in the further evolution of Molteno’s discovery.
Molteno made the observation that the bleb formed around the implant depended on the age of the patient and to some degree on the severity of the glaucoma, in particular to the level of the intraocular pressure immediately prior to inserting the implant [6]. Infants and elderly patients form thin-walled blebs, which function well. Older children and fit young adults were found to produce a series of changes in the blebs after implant surgery, which often led to the formation of a fibrosed bleb. Based on these observations Molteno began extensive research on the pathophysiology of the blebs and discovered that the changes that accompany formation of a bleb over an implant can be named according to the intraocular pressure within the eye, at that particular stage of bleb formation [6].
The initial stage of bleb formation is the hypotensive stage, lasting approximately 7–10 days, and is characterized by vascular congestion and edema of the tissues over the plate. This is followed by a period of steadily rising pressure, disappearance of the edema, and the formation of the bleb itself. About 4–6 weeks after implant insertion, the blood vessels over the bleb become congested, and the intraocular pressure rises to usher in the hypertensive phase. This phase lasts from 2 to 4 weeks after which time the bleb becomes less congested; the pressure falls until a stable plateau is reached about 3–6 months after implant insertion. This results in the stable stage, hopefully remaining for the rest of the patient’s life.
Two major problems associated with the use of the long-tube implants were immediate postoperative hypotony and the development of bleb fibrosis as mentioned in some patients. Methods for dealing with these two problems therefore required solutions.
Control of Hypotony in the Immediate Postoperative Period
In the beginning the single-plate implant was used without restricting flow to the surface of the plate. Shallow to flat anterior chambers were the norm in the postoperative period. Homeostasis of aqueous flow usually took 10–14 days, after which, due to early capsule development over the plate, the anterior chamber began to reform. The earliest attempt at restricting aqueous flow was a modification of the dissection of the conjunctiva in the area of the plate, consisting of decreasing the size of the dissection in the area of plate deposition, by inserting a single Weck-Cel sponge into the area to accomplish the dissection, rather than utilizing scissors to dissect a much larger space [7]. This technique did help to limit postoperative flow into the surrounding tissue, thereby decreasing the potential for shallow or flat anterior chambers but was by no means ideal.
Molteno developed a two-stage procedure for use in those patients where medical control of intraocular pressure was sufficient to allow the eye to tolerate a delay of 5 weeks prior to allowing aqueous to reach the plate surface [8]. This technique consisted of inserting the implant rather than connecting the tube to the anterior chamber, to place it beneath a rectus muscle but to connect it to the anterior chamber at a second operation 5–6 weeks later when a sufficient capsule had developed over the plate, thereby preventing postoperative hypotony. This procedure, although effective, required two surgeries and was replaced by the “vicryl tie” procedure [9]. Herein the tube was constricted by placing a 5-0 vicryl suture tightly around the tube prior to inserting it into the anterior chamber at the time of implant insertion. This resulted in delaying aqueous access to the plate surface for 3–5 weeks, the usual time taken for dissolution of the suture, at which time a sufficient capsule had developed over the plate, once again preventing excessive hypotony.
The ability to control postoperative hypotony allowed Molteno to increase the drainage area by introducing a multiple plate system. The concept being if one plate lowers pressure, then surely two or more plates will multiply the pressure-lowering effect. Molteno tried using as many as four plates, one in each eye quadrant, but found that one or two were sufficient. The introduction of the double-plate implant would usher in a phase of glaucoma implant development which would change the thinking related to glaucoma implants, resulting in the development of different implants, a process that is still continuing, and revert to the all important pathophysiology of bleb formation and its importance [10].
The Influence of the Double-Plate Molteno Implant on Glaucoma Implant Research and Development
The double-plate implant consisted of two single plates joined together by a silicone tube. Aqueous would therefore pass from the surface of one plate to the other plate, and this flow could be restricted by placing a vicryl suture around the connecting tube, thereby restricting flow until a sufficient capsule had formed over the second plate. A major problem with the double-plate implant was the need to isolate the superior rectus muscle, allowing placement of the second plate beneath the muscle into the adjacent eye quadrant. This technique was that used by Molteno and by most surgeons using double-plate implants. This procedure was complicated and time consuming. Placement over the muscle, which was much easier and just as effective, was suggested, but most surgeons continued to use the standard “under” the muscle technique. The first Molteno implant was inserted into an eye in the USA by the author who was a resident with Molteno. Molteno mailed this implant to the author, with instructions on its use. The author subsequently mailed an implant to Baerveldt, also with instructions on its use. A study published by the doctors working with Baerveldt concluded that the two-plate implant was better in intraocular pressure control than the single plate, and therefore this implant became the implant of choice when glaucoma implants were chosen [11]. The surgical difficulty of placing the second implant persisted. Baerveldt concluded that a single-plate implant, with at least the surface area of the double plate or larger, would be more practical and could be placed into a single quadrant. Thus the Baerveldt implant came into being. The single-plate implant would subsequently become the future of all glaucoma implants, including the Molteno implant.
The problem of hypotony with the introduction of larger single-plate implants became more important. Control of hypotony resulted in further developmental advances in design of implants as well as in surgical innovations.
Molteno developed the dual chamber implant, prior to the introduction of the Baerveldt implant [12, 13] (Fig. 19.2). This implant was available in both a single- and double-plate model.


Fig. 19.2
Dual chamber Molteno3 implant
The dual chamber implant is placed with the anterior edge of its plate in line with the insertion of the rectus muscles. This allows Tenon’s capsule to be pulled tightly over the anterior situated chamber, which has a posterior pressure ridge. This pressure ridge when covered by Tenon’s capsule acts as a pressure-sensitive valve which regulates the flow of fluid into the main bleb cavity. The intraocular pressure at which aqueous flows into the main chamber depends on the tension of the Tenon’s capsule over the anterior chamber as well as the swelling of covering tissue in the postoperative period. The pressure ridge needs to be of sufficient height to produce this valve action. The swelling of the tissue decreases allowing a bleb to form over the anterior chamber, as a capsule has developed enclosing the space. This prevents flow into the second chamber and prevents hypotony. As the intraocular pressure increases, the aqueous will flow over the ridge into the main chamber maintaining a low intraocular pressure. The dual chamber implant will not function as a valve unless it is used as Molteno suggested, which is by pulling Tenon’s tightly over the ridge and suturing it to the muscle insertion. Without this technique the dual ridge implant acts as a non-dual chamber device. The pressure at which aqueous flows across the pressure ridge depends on the turgor and elasticity of the overlying tissue. Molteno has found that irrespective of variation in tissue over the chamber, that pressure during the first postoperative week is maintained at 6–22 mmHg.
The advantage of the dual ridge valve is that it can clear itself rapidly after blockage by inflammatory exudate or solid blood clot. Unfortunately the dual ridge implant had not been used as proposed by Molteno, resulting in the search for a different valve mechanism leading to the development of the Ahmed implant, with its pressure-sensitive valve. The Ahmed implant differed from the Molteno implants in that it was a single-quadrant larger single plate with a valve mechanism [14]. Double-plate Ahmed implants were introduced at a later date.
The Baerveldt implant required a mechanism to prevent postoperative hypotony, as did all the Molteno implants including the dual chamber implant, the latter due to the failure in adopting the correct means of insertion of the implant. Use of intraluminal stents and tubal occlusion with circumferential sutures became the usual means of hypotony control. The tying off of tubes required a mechanism to relieve postoperative increases in pressure, prior to tube opening. Sherwood suggested making a relieving slit in the tube anterior to the area of occlusion, allowing aqueous to escape into the tissue surrounding the tube, lowering the intraocular pressure [15] (Fig. 19.3). This slit needs to be made on the lateral side of the tube to prevent occlusion of the overlying patch material. As a result of the restriction of aqueous flow to the plate surface until pressure had normalized in the eye, it became apparent that the hypertensive phase, as described previously, became less marked and often did not occur at all. This phenomenon appeared to be due to a decrease in the release of proinflammatory substances to the plate surface, as will be explained later in the chapter. The discovery that double plates had a better pressure-lowering potential than single-plate implants, as pointed out earlier, led to the concept that bigger is better. This in turn resulted in the development of large single-plate implants, with different sizes available in the Molteno and Baerveldt groups. However with further studies relating to the efficacy of size, it became apparent that beyond a size equivalent to the 300 mm Baerveldt, that additional pressure lowering was minimal if at all present [16]. The single-plate implants used most frequently at the present time include the single-plate Molteno3 implants 175 and 230 mm2, the Baerveldt implant 250 and 350 mm2, and the Ahmed and Krupin implants both 184 mm2. A newer single-plate Molteno implant, a modified Molteno3, will be described later (Fig. 19.4). Very little additional pressure lowering is obtained with implants larger than 175 mm2. The introduction of the single-plate larger-size implants has made the surgical procedure easier, as well as freeing up an additional superior quadrant should an additional implant be required.
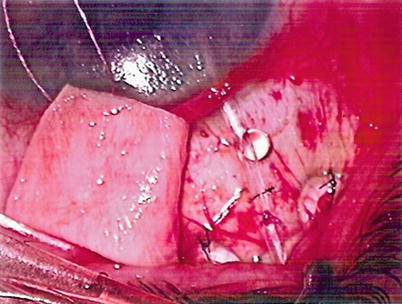
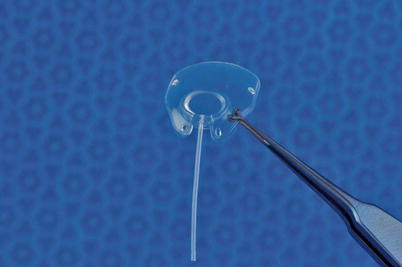

Fig. 19.3
Aqueous bubble exiting anterior chamber via Sherwood slit in silicone tube

Fig. 19.4
Molteno3 with low ridge and anteriorly placed suture holes
The introduction of delayed release of aqueous to the plate surface was observed to result in a less intensive tissue reaction over the plate, especially if the intraocular pressure was elevated. Molteno made the observation that the intensity of the inflammation depended on the patient’s age and the intensity of the glaucoma. The immediate release of aqueous onto the plate surface in the presence of high IOP resulted in a more fibrosed and less efficient bleb. This reaction occurs as a result of the passage of proinflammatory cytokines present in glaucomatous aqueous. In infants and the elderly, without severe glaucoma, the tube opening is followed by low-normal IOP and 3–4 weeks later moderately elevated IOP. In severe glaucoma and young healthy patients, the pressure rises sooner and is higher with a more marked inflammatory reaction over the plate. Molteno found that in these patients, this reaction and the hypertensive phase could be modified with the use of anti-fibrosis systemic medication.
The Control of Postoperative Inflammation and the Hypertensive Phase of the Bleb
Molteno found that the use of a systemic combination of drugs, comprising steroids, nonsteroidal anti-inflammatories, and colchicine, given in combination, reduced postoperative inflammation [6]. Furthermore the additional use of topically applied epinephrine and atropine had an additive effect. According to Molteno the medication should not be given prior to the flow of aqueous onto the plate surface as this will prevent the formation of a preformed bleb lining, resulting in increased hypotony after the tube opens. The use of Molteno’s anti-fibrosis drug regimen, albeit effective, has not been adopted universally. The reason for this is that the drugs need to be taken within 4–5 days after the operation and continued usually for a period of 6 weeks. There are multiple side effects related to their use, and often the patients need to be hospitalized in order to be monitored, an impractical situation in many countries.
Maintaining a low IOP after the tube opens will result in a more functional bleb. This can be achieved with the use of hypotensive medications used from the time of implant insertion for 12 weeks, a period that will include the hypertensive phase. An alternative method has been introduced to treat the hypertensive phase, consisting of removing aqueous from the bleb, using a 30-gauge needle, a procedure that can be done in the office and which can be repeated weekly or less frequently until the stable stage of the bleb is reached [17]. The use of this technique has resulted in the development of thinner and more functional blebs. Aqueous removed from blebs during the hypertensive phase has been shown to contain cytokines, namely, TGFβ (transforming growth factor beta) and PGE2 (prostaglandin E2) [18]. Recent research has discovered additional cytokines in glaucomatous aqueous, the most consistent being interleukin-8 and monocyte chemotactic protein (MCP-1 and MCP-2), all of which are proinflammatory (current research to be published). Therefore tapping the bleb not only lowers the IOP but also at the same time removes these proinflammatory products resulting in less postoperative inflammation and subsequently thinner and more functional blebs.
Clinical observation, relating to the effect of the hypertensive phase of the bleb, has indicated that a hypertensive phase in which the pressure is very high and prolonged will result in a final bleb with a thicker lining rendering it less effective. Another observation made is that the hypertensive phase is more marked if the aqueous is allowed to reach the plate surface at the time of surgery [19]. As previously stated, “glaucomatous aqueous” has been shown to contain proinflammatory cytokines, which will result in an early intense inflammatory reaction over the plate producing an inflamed bleb wall, which in turn results in a more intense hypertensive phase. Preventing aqueous from reaching the plate surface, until the IOP has been lowered and thereby containing less cytokines, has proven to decrease the intensity of the inflammatory reaction over the plate as well as a less intense hypertensive phase.
Development of Ancillary Methods for Bleb Fibrosis Control
The understanding of bleb physiology has resulted in the development of ancillary methods for bleb fibrosis control. The implant per se only really acts as a conduit for transporting aqueous from the anterior chamber to the subconjunctival space as has been previously mentioned. The main factors involved in bleb formation are the aqueous and the tissue over the plate. The initial aqueous is proinflammatory due to its contents of cytokines, and the tissue, most specifically Tenon’s capsule, contains the messenger RNA (mRNA) for tumor necrosis factor-beta (TGFβ), a proinflammatory cytokine [20]. Controlling aqueous flow to the plate and manipulating Tenon’s capsule or a combination of both was thought to offer promise in the control of bleb fibrosis.
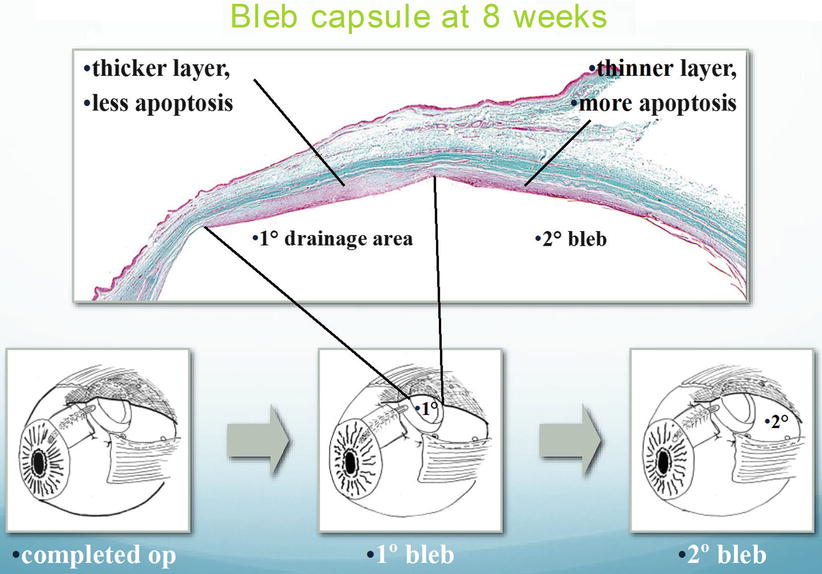
Preventing aqueous from reaching the plate surface in non-valved implants is accomplished by tubal occlusion as has been described. Prior to this technique Molteno utilized a two-stage procedure to prevent aqueous flow to the plate. Following placement of the implant, the tube was not introduced into the eye, but only done so 6 weeks later when a thin fibrous capsule had developed over the plate. This technique was associated with actively lowering the IOP as much as possible with medications, so that when the tube was finally introduced into the eye, the IOP was lower than the IOP at the original surgery, thereby lowering cytokine levels and thus the fibrotic potential of the aqueous. Because the two-stage procedure was originally designed to prevent postoperative hypotony, with the advent of the vicryl tie method, this technique was abandoned by Molteno. However it also led to the development of the dual chamber implant. Although developed to control hypotony as previously explained, the dual chamber implant also prevented the flow of aqueous to the second chamber, and this occurred usually after the IOP had been lowered by its isolation in the anterior chamber and subsequent formation of a draining bleb over this chamber, before allowing flow to the main plate surface. This resulted in a different type of aqueous flowing onto the plate, one with less cytokines, resulting in the formation of a thinner and more efficient bleb. Molteno has demonstrated this occurrence with histology gleaned from a postmortem eye (Fig. 19.5).