The treatment of laryngotracheal stenosis has evolved over the past several decades. Advances in technology, equipment and medication have increased our ability to treat some airway stenosis in a minimally invasive manner, and at times by an endoscopic method. With improved precision, our results with postoperative voice and swallowing should improve.
The treatment of laryngotracheal stenosis (LTS) has been a persistent challenge in pediatric otolaryngology. As a result, the management strategies have undergone constant scrutiny and refinement in the last several decades. The initial description of cricoid framework expansion using costal cartilage was described by Fearon and Cotton in 1972. Although the initial open techniques for laryngotracheal reconstruction (LTR) had a high success rate for decannulation, the surgical approach recommended total laryngofissure with prolonged postoperative stenting to prevent restenosis and ensure graft stability. Displacement of the epiglottic petiole can result in blunting of the anteroposterior dimension of the supraglottis at the level of the false vocal cords. Production of scar in the lamina propria inhibits normal vocal cord vibration. Not surprisingly, the resulting anterior commissure blunting, arytenoid fixation, vocal fold asymmetry, and glottic insufficiency often contributed to patient dysphonia. The compromised voice quality was subjectively described as low-pitched, breathy, or hoarse . Objective evaluation of voice quality with fiberoptic laryngoscopy and videolaryngostroboscopy revealed that both voice quality and speech intelligibility were compromised postoperatively . Swallowing difficulties were also encountered in the perioperative period because of long-term stenting.
Minimally invasive open techniques
With improvement of decannulation rates, the issues of phonation and deglutition were the next to be addressed. Modifications of laryngotracheal reconstructive surgery were developed, which included short-term stenting, single-stage LTR without indwelling stents, and limited laryngofissure. The initial shift from long to short Aboulker stents sought to improve swallowing ability in patients who required prolonged stenting and allowed decannulation because the shorter stents were not secured to the tracheostomy tubes. Nonetheless, shorter stents carried the reported risk of circumferential suprastomal granulation tissue when left in place for over 4 weeks. In cases of posterior glottic stenosis, short-term stenting became possible with the placement of a posterior cartilage graft. The subsequent transition toward single-stage LTR without stent placement was initially limited to select patients with greater lumen diameter, normal vocal cord mobility, and no significant glottic involvement. The two-surgeon technique, to address the concern of anterior commissure preservation, was first described in 1998 . This method involved more precise airway incisions with endoscopic guidance by the assistant surgeon ( Fig. 1 ). Direct visualization of the stenotic area by the endoscopist enabled the primary surgeon to avoid anterior commissure disruption when possible.

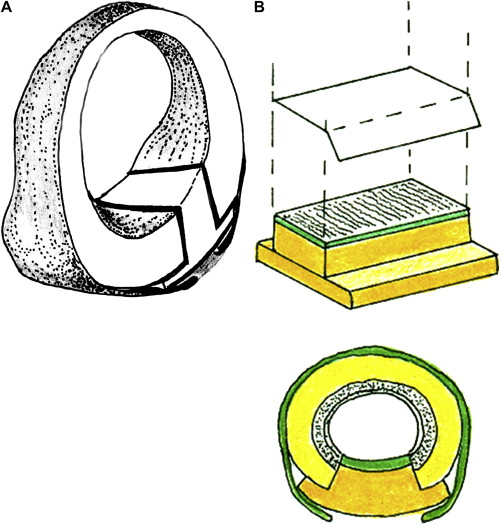
Further advancements in airway reconstruction include the popularization of “sutureless” posterior cricoid grafting. The technique was initially criticized as dangerous because of the risk of graft protrusion into the airway without stabilization with multiple sutures. This method has proved, however, to be both effective and successful . The cartilage graft is fashioned with overlying perichondrium in a boat shape with flanges to avoid prolapse into the airway ( Fig. 2 A). The use of a template for the graft size and shape and precise measurements of depth, length, and width of defect can also ease graft placement ( Fig. 2 B). This surgical modification enables easier placement of grafts into a subperichondrial pocket and avoids the difficult placement of posterior graft sutures with resultant granulation formation. More recently, sutureless posterior cricoid grafting with flanged costal cartilage has been described for the management of isolated posterior glottic stenosis in children .
Presently, the mainstay of treatment for moderate to severe LTS is augmentation with anterior and posterior cartilage grafts. Nonetheless, the desire both to improve voice quality and swallowing function postoperatively and to avoid external scarring in children has fueled the quest for alternative methods. For less severe cases, a variety of noninvasive technique instruments including the laser and microdebrider have been applied successfully.
Endoscopic techniques: less is more
The 1980s heralded the availability of endoscopic techniques, which were first applied to the removal of airway foreign bodies. The idea of endoscopic-assisted surgery developed in the following decade. The application of endoscopy to pediatric laryngologic surgery has revolutionized the field. Initially described as an assistive technology for limiting laryngofissure, endoscopic approaches now may often obviate the need for open surgical intervention.
Endoscopic repair of subglottic, posterior, and glottic stenosis and vocal cord paralysis has been successful when patients are chosen appropriately. There are various advantages to endoscopic repair of these laryngotracheal lesions. Many of these procedures can be performed without requiring intubation. As a result, these endoscopic procedures can be performed as an outpatient as opposed to open reconstruction, which may require postoperative care in the PICU setting with or without postoperative intubation or ventilation. In open single-stage LTR, nasotracheal intubation may be used postoperatively as an airway stent. During this period, it is common practice to use heavy sedation, physical restraints, and occasionally neuromuscular blockade to minimize the risk of accidental decannulation. Mechanical ventilation is necessary when pharmacologic restraints are used. Several complications including pulmonary atelectasis, pneumonia, airleak syndromes, sedative medication withdrawal, and prolonged neuromuscular weakness have been reported. These complications may lead to prolonged PICU and hospital stay, which may be avoided in selective outpatient endoscopic management.
Endoscopic arytenoidectomy or cordotomy was initially described in children for the management of bilateral vocal cord paralysis . Endoscopic repair for low-grade posterior glottic stenosis using a carbon-dioxide (CO 2 ) laser was also reported in the literature . The extent of arytenoid resection ranges from complete arytenoidectomy to vaporization of the vocal process with lateralization of the vocal cord and preservation of the lateral body of the arytenoid to decrease risk of aspiration. Endoscopic laser resection is also applied as an adjunctive procedure to augment open repair. As discussed later, there are a wide range of procedures using the laser and microdebrider that are now performed entirely by an endoscopic approach.
Endoscopic posterior cricoid split was first described by April and colleagues in a canine model in 1991 using the holmium:YAG laser. Previous endoscopic attempts with CO 2 laser had poor results because of circumferential scarring, tracheomalacia, and loss of cartilage. More recently, Inglis and colleagues achieved successful decannulation of two patients with posterior glottic stenosis and one patient with posterior and subglottic stenosis by endoscopic posterior cricoid split with rib cartilage grafting. A “safety suture” was used to retrieve the graft if displaced below the larynx during surgery. The advantage of this minimally invasive technique is that it avoids destruction of the vocal cords or arytenoids in addition to obviating the need for an open approach.
Endoscopic management of bilateral vocal cord paralysis with CO 2 laser arytenoidectomy was described by Ossoff and colleagues in 1984. This technique involves vaporization of the arytenoids to the cricoid cartilage, the vocal process, and a limited area of the vocalis muscle. Dennis and Kashima later described successful decannulation with CO 2 laser posterior partial cordectomy. A 3.5- to 4-mm C-shape wedge of posterior vocal cord is excised from the free border of the membranous cord extending 4 mm laterally anterior to the vocal process.
Laser
The laser has a wide range of applications in the pediatric airway from the nasopharynx to the distal bronchi. It is a useful tool in the treatment of choanal atresia and recurrent respiratory papillomatosis; the unroofing or marsupilization of laryngeal, epiglottic, and vallecular sacular cysts; the management of anterior and posterior glottic scarring and subglottic stenosis; and the vaporization of suprastomal granulation tissue. The laser is used to manage bilateral vocal cord paralysis and posterior glottic stenosis with arytenoid fixation.
Otolaryngology was the first discipline successfully to apply the CO 2 laser clinically. After collaborating to develop the endoscopic attachment to the CO 2 laser, Jako performed the first in vivo experiments on the canine larynx in 1969. In 1971, after Bredemeir devised the stereo laser endoscope, which enabled used of the manipulating arm of the laser to the operative microscope, Strong and Jako began applying the CO 2 laser to laryngeal microsurgery. Further improvements of the laser delivery system by Ossoff and Karlan enabled precise aiming of the laser beam under direct visualization of the area of the lesion through the bronchoscope. The modern joystick controls allow the surgeon precisely and easily to manipulate the laser beam. Other advancements include specialized endoscopes with continuous smoke evacuation systems. Refinements in laser laryngoscopic surgery have generally been established in the adult population before application to the pediatric airway. The laser enables precise ablative maneuvers on the airway in a “hands off” fashion and in a relatively bloodless field.
Minimizing laser exposure time and power and using the smallest spot size are some of the principles that should be applied to laser laryngologic surgery. This is specifically important in trying to preserve the underlying submucosa, muscle, and cartilage in the pediatric airway.
Carbon dioxide laser
The CO 2 laser has a shallow depth of penetration with a minimal nonspecific thermal effect, which minimizes postprocedure edema. These characteristics make it ideal for the narrow diameter and shallow tissue of the pediatric airway. The CO 2 laser requires an aiming beam because of its invisible wavelength (10.6 μm). It is absorbed mainly by tissue water and can only coagulate vessels up to 5 mm. The CO 2 laser has been used as an alternative to the transpalatal approach to vaporize the bony and membranous plates in bilateral choanal atresia and it has been applied to the debridement of obstructive lesions of recurrent respiratory papillomatosis with the advantage of producing minimal scarring despite repetitive use . Recently, waveguides have been developed to deliver this wavelength, which may enhance the ability to use it in the larynx and subglottis.
Anterior and posterior glottic webs can be addressed with the use of the CO 2 laser to perform a micro trapdoor technique maximally to preserve mucosa . A mucosal flap is elevated and the underlying scar is vaporized and the mucosa is redraped. As always, the lowest power (3–5 W), spot size (250 μm), and pulse durations (0.2 or less) should be used to minimize the risk of scar reformation.
Acquired subglottic stenosis may result from emergency tracheotomy, trauma, and prolonged or repeated intubation. Laser ablation of Cotton grade 1 or 2 acquired and congenital subglottic stenosis has shown to be effective in noncircumferential lesions that are no longer than 1 cm in length. A staged quadrant-by-quadrant method may be used at 6-week intervals similar to the micro trapdoor technique described previously . The major complications from laser management of subglottic stenosis are restenosis, thermal damage, and secondary scarring.
KTP laser
The KTP laser emits a visible wavelength (532 μm) that is preferentially absorbed by hemoglobin. This characteristic makes the KTP laser very useful in the treatment of vascular lesions, such as subglottic hemangiomas. A fiberoptic delivery system enables its use in the distal airway using a ventilating bronchoscope. As compared with the CO 2 laser, its tissue penetration is significantly deeper (4 versus 0.9 mm). In a series of 12 patients with subglottic hemangioma in which patients were treated with KTP laser or CO 2 laser, all patients had resolution of symptoms and all five tracheotomy-dependent patients were decannulated and there were no complications noted in the KTP laser group . The delivery system of the KTP laser provides greater adaptability and precision than the CO 2 laser. The KTP fiber can also be applied through a rigid bronchoscope to vaporize a dense subglottic scar in a three-quadrant pattern.
The KTP laser has higher tissue absorption compared with CO 2 laser. Special attention must be paid to avoid thermal penetration of the tracheal wall and transmural injury. Granulation tissue results from repetitive trauma or friction. The KTP laser can be used by a hand-held method to ablate tracheotomy parastomal disease or through an endoscope for suprastomal disease.
Neodymium:yttrium-aluminum-garnet and argon lasers
Various negative characteristics of these two lasers limit their application to the pediatric airway. The risk of transmural injury is too high given the 4-mm depth of penetration of the neodymium:yttrium-aluminum-garnet laser. In the canine model, the argon laser has demonstrated a high rate of tracheal perforation. The lateral and deep tissue extension is unpredictable with loss of visual clues to the extent of tissue damage and extensive postoperative edema.
Future direction
Several new wavelengths that combine the optimum tissue absorption and penetration characteristics with the best delivery systems are under investigation. For example, the Er:Yag laser has a pulsed nature that allows cooling between interval pulses resulting in less tissue injury. The Ho:YSGC, Er:Yag, pulsed dye, diode, and XECL excimer lasers and photodynamic therapy are some of the other modalities being testing.
Microdebrider
The application of the microdebrider to the pediatric airway is relatively new. Initially used in orthopedic and temporomandibular joint surgery for several decades, Reuben Setliff is credited with introducing the modality to otolaryngologists in the United States for use in endoscopic sinus surgery. The microdebrider allows for precise and rapid removal of many obstructing airway lesions. It is simultaneously able to remove debris and blood allowing better visualization of the operative field during dissection. In general, it is an easily mastered surgical tool with most of the same applications in the pediatric airway as the lasers mentioned previously. Complications of thermal modalities (eg, laser), which include airway injury, tracheoesophageal fistulas, and airway fires, can be avoided. Modifications and extensions to the tip length and angulation have facilitated the expansion of its use in the airway. Sufficient tissue for biopsy may be obtained as long as orientation of the specimen is not necessary. Following its establishment in the adult airway, the microdebrider was applied to the endoscopic treatment of a number of pediatric airway lesions.
The use of the laryngeal microdebrider in the treatment of recurrent respiratory papillomatosis was first reported in 1999 . It has now become an invaluable tool in the treatment of this difficult disease. A major advantage to the microdebrider is its ability to engage papilloma only and avoid damage to the healthy underlying tissue. This minimizes collateral mucosal injury. Additionally, it avoids the risk to the staff of laser plume.
Granulation tissue is particularly easy to remove with the microdebrider. The microdebrider has been successfully used in the adult and pediatric airway to remove suprastomal and peristomal granulation tissue . Airway obstruction following LTR is often caused by granulation tissue formation and the microdebrider has been used for repeated management of this complication. Stenoses of the glottis, subglottis, and trachea, and airway cysts have also been effectively addressed with the microdebrider. It has been shown to reduce operative time compared with the laser . In the presence of flammable airway foreign bodies, the microdebrider may be applied without fear of combustion.
In contrast, there are notable disadvantages to the microdebrider. It is a cold technique and may be contraindicated when hemostasis is of particular concern. With current equipment, the microdebrider cannot be used through a ventilating bronchoscope. Nonetheless, most pediatric tracheal lesions can be reached through the suspended laryngoscope with the extended blade. Future modifications of the instrument blades may enable more extensive use in pediatric bronchoscopy.
Balloon laryngoplasty
The open surgical options for subglottic stenosis include tracheotomy to bypass the narrowing, and cricoid split and single-stage laryngotracheoplasty to widen the airway are well established. Endoscopic techniques for the management of this problem are appealing because they are significantly less invasive. The literature describing the use of the laser as the primary endoscopic tool for subglottic stenosis reports variable success. Balloon laryngoplasty is an endoscopic technique that may provide an alternative or temporizing measure in infants with acquired subglottic stenosis.
Successful balloon dilatation of tracheal stenosis has been reported in the pediatric literature for congenital stenosis and postsurgical restenosis . Limited literature is available on the use of balloon laryngoplasty as the primary treatment for subglottic stenosis. Recently, a 70% success rate was reported for balloon laryngloplasty in infants with acquired Cotton grade 2 or 3 subglottic stenosis . After dilation, topical steroids were applied to the previously stenotic segment and the patients remained intubated for 24 to 48 hours. All patients empirically received systemic steroids and a proton-pump inhibitor in the postoperative period to reduce postoperative inflammation.
It has been suggested that balloon laryngoplasty is only effective to interrupt the process of mature scar formation during the evolution of acquired subglottic stenosis. To this point, it has not been reported in the literature for cases of long-standing, congenital subglottic stenosis or cases with cartilaginous involvement.
Adjuvant therapies
Mitomycin
Postoperative wound healing poses one of the main obstacles to success in airway surgery. Recurrent tracheal granulation tissue formation and cicatrix formation after LTR result in intraluminal scarring and narrowing in initially patent airways. Mitomycin C (MMC), a potent antibiotic derived from the Streptomyces caespitosus bacteria, can modify wound healing at the molecular level and has been used to interfere with postsurgical scar formation. Available since the 1960s as a systemic chemotherapeutic agent in the treatment of solid tumors, MMC was first applied topically to the treatment of superficial bladder tumors. Later, ophthalmologists pioneered its use in preventing pterygium recurrence after surgical excision. Its use was first reported in the otolaryngology literature for the treatment of tracheal cicatrix after tracheal reconstruction in a small case series . All patients were successfully decannulated. In this initial study, topical MMC was applied intraoperatively on saturated pledgets at a dose of 0.1 mg/mL for 2 minutes to the area where the scar had been lysed. All patients in the study had previously failed standard stenting methods and postoperative laser and dilatation treatments.
Since then, a number of randomized prospective animal studies have shown impressive results in the prevention of postoperative glottic and subglottic stenosis following electrocautery and laser injury to the airway . The mechanism of the drug’s anticellular action has not been definitively characterized. It is known, however, to be a prodrug that is activated into toxic forms that produce oxygen-free radicals, create single-strand DNA breaks, and cross-link DNA. Fibroblast studies suggest similar DNA-destructive effects and MMC-induced apoptosis in fibroblast cells .
Although initially only applied to revision cases, MMC is now used routinely as a topical therapy in primary airway cases in which postoperative scarring is of significant concern. Various concentrations and durations of application have been used because optimum guidelines have not been firmly established with prospective studies. Currently, 0.4 mg/mL is the recommended concentration at many institutions, but higher concentrations have been used. Mitomycin therapy has been shown to be a useful adjunct following cold-knife and laser ablation of lesions of the glottic, subglottic, trachea, and bronchi. In cases of laryngeal webbing, MMC application after web lysis may enable avoidance of keel placement and prolonged postoperative intubation. In cases of nasal vestibular stenosis, choanal atresia, and pharyngeal flap revision, instillation of MMC following repair may obviate the need for stenting . Most importantly, although it is primarily a chemotherapeutic agent when used intravenously, MMC applied topically has demonstrated a good safety profile with normal epithelialization of treated tissue.
Many mitomycin analogues are being developed as antitumor agents. New delivery vehicles, such as gels and sustained-release films, show promise for future application. Chitosan has been studied as a drug carrier for MMC because of its biocompatibility, low toxicity, and long-term retention in the body. A recent study in canines demonstrated that chitosan is more effective than MMC in preventing anterior glottic stenosis after laser phonomicrosurgery .
Antireflux therapy
Gastroesophageal reflux disease (GERD) has been suggested as a critical factor in the development of subglottic stenosis. Multiple animal models have demonstrated that exposure of the subglottic mucosa to gastric acid causes mucosal ulceration and delayed healing and other cellular changes . There is evidence to suggest that there is a higher incidence of GERD in adults and children with laryngeal and tracheal stenosis. In 36 children undergoing LTR for subglottic stenosis at the Children’s Hospital of Pittsburgh, 21 (80%) of 26 patients tested for GERD had at least one positive result . This finding is significantly higher than the 20% estimated incidence of pathologic infantile reflux. In another series of children with subglottic stenosis who underwent esophageal biopsy, 23 (68%) of 34 patients had positive biopsies for GERD. These studies suggest a possible relationship between GERD and subglottic stenosis .
There is also evidence to suggest a relationship between airway obstruction and GERD. In a guinea pig model, esophageal acid triggered the release of substance P, which initiated airway plasma extravasation. Edema of the laryngeal mucosa from gastric acid may cause sufficient airway to compromise an already tenuous postsurgical airway. Moreover, in children with partial airway obstruction, mucosal thickening or edema from GERD may convert a marginal airway to a critical one. Although the data are limited, most agree that aggressive antireflux with proton pump inhibitors is an important component of any medical plan for these pediatric airway patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


