Medical Therapy for Obstructive Sleep Apnea
Ryan J. Soose
Patrick J. Strollo
INTRODUCTION
Obstructive sleep apnea (OSA) is a chronic condition that will require management across the lifespan. Like most chronic diseases, prevention of OSA remains the primary goal and most attractive treatment from a public health perspective. At this juncture, management of the large population of patients diagnosed with sleep-related breathing disorders (as well as those who are still undiagnosed) remains the critical focus of most sleep physicians. As with hypertension (HTN), diabetes, and other chronic conditions, the individual treatment plan is not aimed at “cure” but rather (a) symptom and quality of life improvement and (b) reduction of cardiovascular and general health risk.
Both physician and patient understanding of OSA is the cornerstone for successful management. This approach allows for customization of each individual’s treatment plan depending on their symptoms, airway anatomy, disease severity, and medical comorbidities. Across the lifespan, the same patient, with the same apnea hypopnea index (AHI), may benefit from different discrete treatment options, or a combination of treatment options, depending on the clinical context at that particular stage of life.
Positive airway pressure (PAP) therapy and oral appliances comprise the foundation of OSA medical management in the majority of patients. Nevertheless, there are many other management tools that can be employed to optimize clinical results, particularly when used in combination with PAP or oral appliance therapy. An individualized approach considering all available treatment options provides the most effective long-term management strategy for many patients.
POSITIVE AIRWAY PRESSURE
Introduction
The treatment of OSA with continuous positive airway pressure (CPAP) was first described by Sullivan et al. in 1981 (1). The introduction of CPAP marked an enormous advance in the management of OSA. The first commercially available PAP systems were marketed in the mid-1980s. CPAP rapidly became the mainstay of sleep apnea therapy and is still the most widely used treatment modality today.
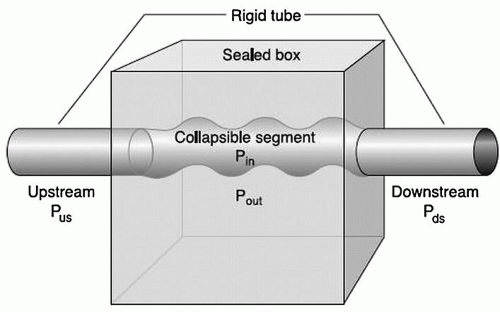
In the Starling resistor model of the pathophysiology of OSA (Fig. 136.1), the collapsible segment of the tube (pharynx) is bound by an upstream and downstream segment with a corresponding upstream and downstream pressure (Pus and Pds) (2).
Airway occlusion occurs when the surrounding tissue pressure, Pout (comprising tongue, pharyngeal muscles, parapharyngeal fat, mucosal edema, etc.), becomes greater than the intraluminal pressure (Pin).
The critical closing pressure (Pcrit) is represented by the Pin when airway obstruction occurs. Inspiratory airflow depends directly on the difference between Pus and Pcrit, and therefore, effective therapy for OSA requires widening the differential between Pus and Pcrit. This goal can be accomplished in two ways: (a) increasing Pus or (b) reducing Pcrit.
PAP works by accomplishing the former with the application of increased pressure at the airway opening. PAP systems for the treatment of sleep apnea consist of a generator that directs airflow downstream to the patient via tubing and an interface. Positive pressure is introduced into the upper airway providing a pneumatic splint through the entire length of the collapsible pharyngeal segment and thus maintaining airway patency and effective control of breathing. The splinting effect constitutes the primary mechanism of therapeutic action, although augmentation of lung volume may play a secondary beneficial role as well (3, 4).
Modalities
Continuous Positive Airway Pressure
Continuous positive airway pressure (CPAP), or fixedpressure CPAP, was the first modality described and still the most widely used. This modality delivers the same
pressure to the patient during both inspiration and expiration and remains unchanged throughout the night.
pressure to the patient during both inspiration and expiration and remains unchanged throughout the night.
Bilevel Positive Pressure
Bilevel positive pressure (BIPAP) provides the ability to independently adjust the inspiratory (IPAP) and expiratory (EPAP) pressure such that the pressure delivered during exhalation is lower than that delivered during inhalation (5). Several studies have confirmed the effectiveness of BIPAP in the treatment of OSA in adults (6, 7, 8, 9).
Most BIPAP devices are used in one of two modes: spontaneous (S-mode) or spontaneous-timed (ST-mode). In S-mode, IPAP is delivered in response to a patient “trigger.” In ST-mode, the patient may trigger the delivery of IPAP; in addition, the physician may set the device so that IPAP is delivered at prescribed intervals if a spontaneously triggered breath does not occur within that interval. This backup respiratory rate provided in the ST-mode is not commonly employed in the treatment of classic OSA but may be beneficial in selected patient groups with more complex sleep-disordered breathing, such as those with neuromuscular disorders, chest wall deformities, or congestive heart failure (CHF) with Cheyne-Stokes breathing.
Autotitrating Continuous Positive Airway Pressure
The pressure required to maintain airflow and control of breathing may vary throughout the night, as well as from night to night, and may be dependent on patient factors such as sleep staging, body position, and alcohol consumption. In autotitrating devices, the physician sets a prescribed pressure range, allowing the delivered pressure to vary throughout the night in order to meet the patient’s changing pressure requirements in real time. Autotitrating devices employ computer algorithms to detect changes in airflow and adjust the delivered pressure accordingly. Autotitrating devices are available in both autotitrating continuous positive airway pressure (APAP) and autotitrating BIPAP (auto bilevel) forms.
One theoretical advantage of autotitrating technology is the reduction in the overall cumulative pressure exposure during the night and subsequent improvement in patient comfort and compliance. Compared to fixed CPAP, studies of APAP show similar effectiveness on both symptomatic and polysomnographic measures and a small (2 cm H2O) average reduction in pressure. On an individual basis, some patients strongly prefer APAP over traditional fixed CPAP or BIPAP. The literature has not yet supported an improvement in adherence with autotitrating devices compared to fixed-pressure devices in CPAP naïve patients (10, 11).
Methods of Titration
The traditional diagnostic and therapeutic evaluation of OSA involves two separate nights in the sleep laboratory. The first represents a full-montage attended overnight polysomnography for diagnostic purposes. If indicated, the patient then returns to the sleep lab a second night for a treatment study that again involves full-montage polysomnographic recording but in conjunction with manual PAP titration by the attendant sleep technician.
One method of improving cost-effectiveness and patient access is the consideration of a split-night study in certain clinical circumstances. A split-night study combines diagnostic polysomnography and a PAP titration in one night, rather than the traditional process of two separate nights (12, 13). Proper clinical judgment in patient selection is essential, as inaccurate estimation of OSA severity and/or incomplete therapeutic titration may occur. Specific guidelines have been published by the American Academy of Sleep Medicine (AASM) for the use of split-night studies (14).
Alternatively, in-laboratory manual titration may be bypassed altogether with more recent developments in home portable monitoring and home auto-CPAP titration. Properly selected patients with a high pretest probability of OSA may undergo the initial diagnostic study at home with an unattended portable four-channel monitoring system. Additionally, the patient generally receives CPAP education and mask fitting in the clinical setting during the day. If the portable study is consistent with OSA and PAP therapy is indicated, the patient is then loaned an autotitrating device to use at home for a short period of time (e.g., 1 or 2 weeks).
The device is subsequently returned and the information downloaded with regard to effectiveness, usage, and mask leak. The patient can then receive a prescription for either fixed-pressure CPAP (usually set at the 90th percentile pressure) or an APAP as permanent therapy. At the
present time, home autotitration appears to be at least as effective as traditional methods with regard to both subjective and objective outcome measures as well as adherence rates (15). Additionally, it has the potential to be associated with improved cost-savings and increased resource availability across a population.
present time, home autotitration appears to be at least as effective as traditional methods with regard to both subjective and objective outcome measures as well as adherence rates (15). Additionally, it has the potential to be associated with improved cost-savings and increased resource availability across a population.
Interfaces
A variety of types of CPAP interfaces are widely available. The most common types fall under one of three categories: nasal masks, nasal pillows, and full-face masks (Fig. 136.2). Mask selection and sizing is generally accomplished by the sleep technician addressing the patient’s comfort, concerns, and facial anatomy. Proper mask fitting, patient education, and close clinical monitoring and follow-up are essential to successful long-term management.
In one randomized crossover study comparing nasal pillows and a nasal mask, investigators reported that nasal pillows were associated with less frequent adverse effects, less air leak, and less insomnia symptoms, although adherence and functional outcome measures did not differ between the groups (16).
Effectiveness
With adequate pressure application and proper usage/adherence, PAP therapy often provides effective control of sleep-disordered breathing and subsequently accomplishes the main goals of symptom/quality of life improvement and reduction of cardiovascular and general health risks.
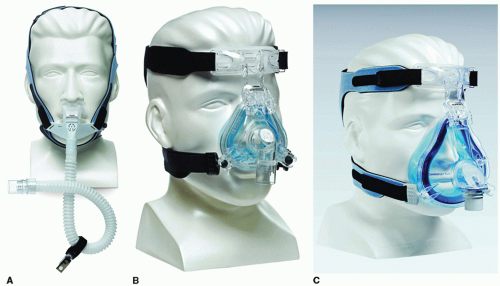 Figure 136.2 PAP interfaces: Examples of the three most common categories of mask interfaces for positive pressure therapy are depicted: (A) nasal pillows, (B) nasal mask, and (C) full-face mask. |
Symptom and Quality of Life Improvement
CPAP has been shown to improve both subjective and objective measures of OSA. On polysomnography, proper CPAP titration is associated with improved sleep continuity and architecture. During the first night of treatment, some patients may manifest an unusually large percentage of rapid eye movement (REM) sleep (“REM rebound”) as a result of sudden reversal of the airway obstruction and associated chronic sleep deprivation. CPAP often has a beneficial impact on other nocturnal aspects of OSA, including treatment of snoring, decreased nocturnal awakenings, and a reduction in nocturia (17, 18, 19, 20, 21, 22).
In studies of patients with OSA and hypersomnia, treatment of CPAP is associated with a reduction in daytime sleepiness and improvement in neurocognitive function and quality of life measures (23, 24). The effect of CPAP on objective metrics of sleep propensity during the day (e.g., multiple sleep latency test [MSLT] or maintenance of wakefulness test) is less clear, with only some studies showing an effect and a less compelling impact in patients with mild OSA (17, 23, 25). Nevertheless, despite some controversy with regard to the sleep laboratory results, the data on motor vehicle collisions, one of the most important public health risks associated with OSA, are much more convincing. A number of studies employing driving simulators have demonstrated improved performance
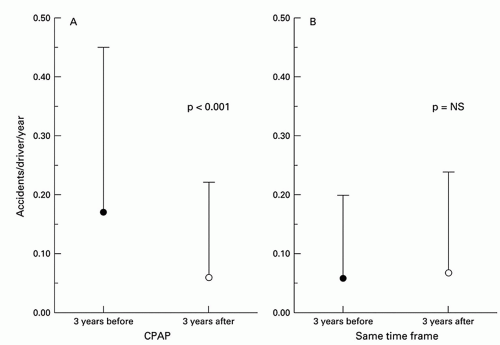
following initiation of CPAP therapy (26, 27, 28). Similarly, a comparison of the number of accidents per driver per year over the 3 years before and following CPAP therapy in OSA patients demonstrated a notable reduction, reaching levels that were comparable to those in individuals without OSA (Fig. 136.3) (29).
following initiation of CPAP therapy (26, 27, 28). Similarly, a comparison of the number of accidents per driver per year over the 3 years before and following CPAP therapy in OSA patients demonstrated a notable reduction, reaching levels that were comparable to those in individuals without OSA (Fig. 136.3) (29).
Reduction of Cardiovascular and General Health Risks
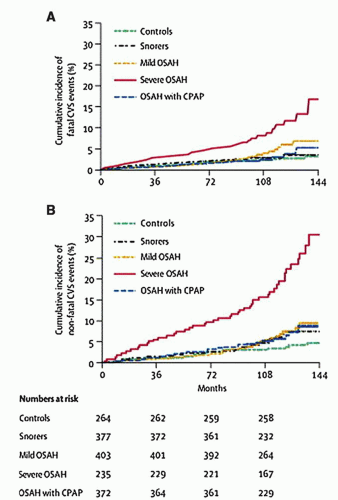
Numerous studies have shown that OSA is associated with cardiovascular disease and that successful PAP therapy results in reduced cardiovascular mortality (30, 31, 32). In a prospective cohort from the Sleep Heart Health Study, investigators showed that sleep-disordered breathing is associated with increased overall mortality as well as mortality specifically due to coronary artery disease, particularly in those patients with severe OSA (AHI greater than 30) (Fig. 136.4) (31). A large observational study showed that men with severe OSA have an increased rate of both fatal and nonfatal cardiovascular events (32). Furthermore, treatment with PAP effectively lowered this cardiovascular risk (Fig. 136.5). Finally, Peker et al. (33) assessed incident cardiovascular events in middle-aged men with OSA over 7 years and noted that the incidence of cardiovascular events in patients who were adequately treated (by PAP, uvulopalatopharyngoplasty, or oral appliances) was about 7% in contrast to about 57% in patients who were inadequately treated.
Cardiac rhythm disturbances are common in OSA patients, and the favorable response of arrhythmias to CPAP therapy, especially in the absence of structural heart disease, reinforces the linkage between OSA and rhythm
disturbances (34, 35, 36, 37, 38, 39). A recent randomized controlled trial reported that, after 1 month, CPAP therapy reduces the frequency of premature ventricular beats by nearly 60% in OSA patients with heart failure compared to patients who are not receiving CPAP, in whom there was no significant change (39).
disturbances (34, 35, 36, 37, 38, 39). A recent randomized controlled trial reported that, after 1 month, CPAP therapy reduces the frequency of premature ventricular beats by nearly 60% in OSA patients with heart failure compared to patients who are not receiving CPAP, in whom there was no significant change (39).
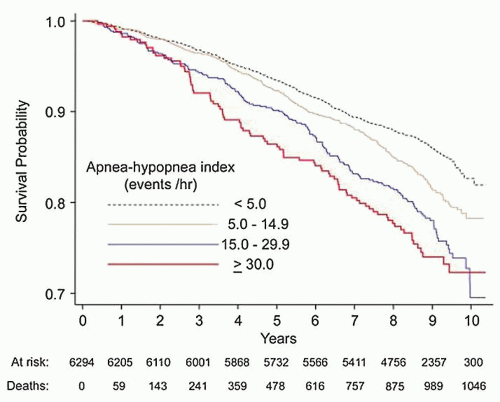 Figure 136.4 OSA severity and cardiovascular mortality: Prospective cohort analysis from the Sleep Heart Health Study showing that increased severity of OSA, based on AHI criteria, is associated with increased mortality from cardiovascular disease. (Reproduced from Punjabi NM, Caffo BS, Goodwin JL, et al. Sleepdisordered breathing and mortality: a prospective cohort study. PLoS Med 2009;6(8):e1000132, with permission.) |
Studies also support the improvement in left ventricular function after initiation of PAP therapy for patients with OSA and heart failure (40). The mechanisms by which PAP may improve cardiac function in OSA patients with heart failure include relief of sleep-related hypoxemia, elimination of cyclic increases in left ventricular afterload, reduction in sympathetic nervous system activation, and reduced inflammation.
Epidemiologic studies have provided compelling evidence that OSA is associated with increased risk for systemic HTN (41, 42, 43, 44, 45, 46). Sleep apnea has been identified by the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure as a risk factor for HTN (47). Although it is plausible that PAP therapy would ameliorate diurnal HTN by eliminating intermittent hypoxic exposure and sympathetic nervous system activation and improving sleep continuity, the literature provides conflicting data in this regard (48, 49, 50, 51).
Using a randomized, placebo-controlled design to compare ambulatory blood pressure on CPAP and sham CPAP in subjectively sleepy OSA patients, Pepperell et al. (52) observed a small but significantly greater reduction in mean blood pressure during sleep in the active CPAP-treated group. Although small, the reduction in blood pressure in the active CPAP group would have a notable public health impact on cardiovascular risk. Conversely, there are a number of well-designed studies of groups of OSA patients either with or without subjective sleepiness, and with HTN, that have failed to demonstrate a significant reduction in blood pressure with PAP therapy (53, 54, 55). In summary, it is evident that further research is required to define the effect of PAP on HTN in OSA patients.
The association between sleep-disordered breathing, including OSA, and increased risk for stroke is increasingly recognized. Data from the Wisconsin Sleep Cohort indicate that, even after adjusting for age, gender, smoking, HTN, alcohol use, and body mass index (BMI), sleep-disordered breathing is associated with increased risk for prevalent stroke (56). Further, there is increasing awareness of the association between OSA, abnormal glycemic control, type 2 diabetes mellitus, and other features of the metabolic syndrome (57). Further studies are required to determine if PAP therapy of OSA reduces risk for incident stroke and improves insulin sensitivity and glycemic control.
Adherence
Despite its documented effectiveness in improving symptoms and quality of life and reduction of cardiovascular risk, acceptance of and adherence to PAP therapy remains one of the greatest challenges in treatment. In studies using at least 4 hours of average usage per night to define adherence, a wide range of 29% to 83% of patients were considered nonadherent (58). The minimal and optimal durations of usage that confer benefit are unknown and likely vary depending on the individual patient as well as the outcome measure being evaluated. It does appear that increasing duration of use is associated with increasing levels of improvement (Fig. 136.6). At least 6 hours of use per night appears to be associated with greater cardiovascular mortality risk reduction compared with fewer hours of use per night (30).
Data card monitoring, with the use of smart cards or web-based technology, has revolutionized the assessment of PAP adherence. The data card provides information over a
selected period of time on multiple data points, including days used, hours of use per night, amount of mask leak, and a surrogate estimate of the residual AHI. Use of this technology to improve monitoring of adherence and effectiveness has the potential to dramatically improve long-term patient care. Any physician involved in the medical management of OSA should consider it part of the routine clinical practice.
selected period of time on multiple data points, including days used, hours of use per night, amount of mask leak, and a surrogate estimate of the residual AHI. Use of this technology to improve monitoring of adherence and effectiveness has the potential to dramatically improve long-term patient care. Any physician involved in the medical management of OSA should consider it part of the routine clinical practice.
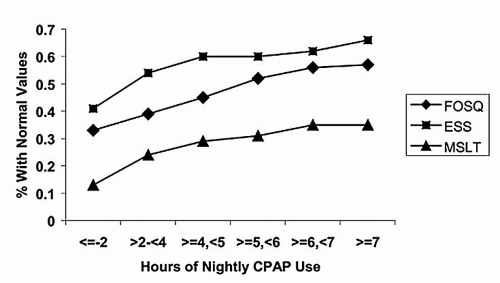 Figure 136.6 CPAP usage and dose-response of outcome measures: Cumulative proportion of participants obtaining normal threshold values on the Epworth Sleepiness Scale (ESS), MSLT, and Functional Outcomes of Sleep Questionnaire (FOSQ). (Reproduced from Weaver TE, Maislin G, Dinges DF, et al. Relationship between hours of CPAP use and achieving normal levels of sleepiness and daily functioning. Sleep 2007;30(6):711-719, with permission.) |
Objective adherence monitoring has made it apparent that patterns of adherence, or nonadherence, are established early. CPAP use in the first week after initiating therapy often predicts long-term success or failure (59). Information regarding the degree to which a patient is adherent to PAP is essential for assessment of a suboptimal clinical response. If a patient’s symptoms are inadequately resolved after the initiation of PAP treatment, possible reasons other than poor adherence include insufficient pressure delivery, mask leak, or a failure to recognize and treat a coexisting medical or sleep disorder that is also contributing to the patient’s symptoms.
Since an individual’s acceptance and adherence to PAP may be established shortly after initiating therapy, it stands to reason that close monitoring, education, and support during this period would be helpful. Studies have demonstrated improved adherence in conjunction with educational materials, available and knowledgeable staff, and patient support groups (60, 61). Patient and family member understanding of the disease process itself, the associated functional and health implications, and the reasons and options for treatment likely contribute to more successful results.
There is increasing evidence that upper airway surgery may provide a useful adjunct to improve CPAP compliance and response to therapy. Unlike past concerns that associated traditional uvulopalatopharyngoplasty techniques result in mouth leak during CPAP use, newer, less morbid, and more effective reconstructive (rather than destructive) procedures may improve medical therapy results by lowering the critical closing pressure and reducing CPAP pressure requirements (62, 63).
Side Effects and Complications
Initiation of positive pressure therapy is often associated with side effects that can impede successful management. The side effects are frequently minor and/or transient and may improve with continued use and adaptation. Others are more significant and troublesome and may negatively affect adherence and even result in discontinuation of therapy. Psychological factors, such as claustrophobia or the perceived lifestyle change associated with indefinite nightly use of a device, are common and often challenging to address (64, 65). Table 136.1




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree