Phenotype
MCTC
MCT
Proteases
Tryptase (+++)
Tryptase (++)
Chymase (+)
Chymase (−)
Carboxypeptidase A3 (++)
Carboxypeptidase A3 (+?)
Cathepsin G (+)
Distribution
Skin (++)
Skin (−)
Intestinal submucosa (+)
Intestinal submucosa (++)
Intestinal mucosa (−)
Intestinal mucosa (++)
Alveolar wall (++)
Alveolar wall (−)
Bronchial subepithelium (+)
Bronchial subepithelium (+)
Dispersed lung mast cells (−)
Dispersed lung mast cells (++)
Tonsils (++)
Tonsils (++)
Nasal mucosa (−)
Nasal mucosa (++)
Relation to pathology
Increased in fibrotic diseases
Increased around the site of T cell aviation
Unchanged in allergic and parasitic diseases
Increased in allergic and parasitic diseases
Unchanged in chronic immunodeficiency diseases
Decreased in chronic immunodeficiency diseases
Response to non-immunological stimuli
Substance P (+)
Substance P (−)
C5a (+)
C5a (−)
PAF (−)a
PAF (+)a
5.3 Role of MCs in Acute Allergic Reactions
MCs express more than 105 high-affinity IgE receptor (FcεRI) per cell. When MCs that have been sensitized with some specific IgE antibody are challenged with the specific allergen, they are activated by cross-linking of FcεRI molecules. Thus, activated MCs evoke immediate-type reaction by releasing their granules in which histamine, neutral proteases, and heparin had been stored. Then, lipid mediators such as cysteinyl leukotriene (cys-LT) or prostaglandin D2 (PGD2) are synthesized on their membranes and are released into microenvironment within several minutes.
Released histamine and lipid mediators cause acute allergic symptoms such as nasal discharge, bronchospasms, and urticaria. Histamine plays an essential role in acute skin allergic reactions, whereas cys-LT plays a pivotal role in bronchoconstriction. MCs almost exclusively express PGD2 synthase compared to all other cell types. Although the role of PGD2 in immediate-type reaction is unclear, it serves as chemoattractant for eosinophils, basophils, and Th2 cells.
Human MCs also exclusively express tryptase, one of the neutral proteases, among all human cell types. Tryptase constitutes 10 % of the MC by protein weight (Hawrylowicz et al. 2006). Proteoglycan (human MCs use “eosinophil” major basic protein instead of proteoglycan molecules) serves as a core protein in the crystalloid structure of the MC granules by binding to heparin and neutral proteases (Nakajima et al. 2002). The MC tryptase acts as trypsin-like enzyme and thereby causes tissue remodeling such as abnormal proliferation of airway smooth muscles (Brightling et al. 2002).
5.4 Role of MCs in Allergic Inflammation
MCs secrete a variety of cytokines and chemokines several hours after allergen-induced degranulation via transcription of these genes. The representative cytokines/chemokines which are produced by activated human MCs are Th2 cytokines such as IL-5, IL-13, and GM-CSF and CC chemokines such as CCL1/I-309, CCL2/monocyte chemoattractant protein-1, CCL3/macrophage inflammatory protein (MIP-)1α, and CCL4/MIP-1β. Activated human MCs also secrete a substantial amount of CXCL8/IL-8 (Nakajima et al. 2002; Bischoff 2007). MCs can store and release some of cytokines such as tumor necrosis factor (TNF)-α during degranulation process. Regarding IL-4 production, it seems reproducible using mouse MCs. However, only a few groups succeeded to immunohistochemically demonstrate the presence of IL-4 on human MCs (Bradding et al. 1992; Pawankar et al. 1997). In any case, at least in human, basophils are more potent producers of IL-4. Instead, IL-4 potently activates human MC function and maturation. Human MCs can produce a substantial amount of another Th2 cytokine, IL-13, in response to IgE-mediated stimuli, and the IL-13 production is markedly enhanced by preincubation with IL-4 (Bischoff 2007). However, these cytokines and chemokines are not unique to MCs and are produced by other cell types. During antigen stimulation, more Th2 cytokines would be produced by proliferating T cells. We should consider the relative role of MCs in the allergic or innate-type inflammation by understanding cytokines/chemokines produced by other immune cell types and epithelial-mesenchymal tissues. For example, epithelial-mesenchymal tissue-derived thymic stromal lymphopoietin (TSLP) and IL-33 are now recognized as most important cytokines for both innate-type and allergic inflammation occurred in allergic diseases such as asthma. These two cytokines and CC chemokines such as CCL17 and CCL23 are produced in response to external stimuli and Th2 cytokines such as IL-13 and stimulate the chemotaxis and development of Th2 cells and the function of MCs. Other than Th2 cytokines and CC chemokines, tryptase, cys-LT, and TNF-α also stimulate epithelial-mesenchymal tissue (Hawrylowicz et al. 2006; Oboki et al. 2010; Takai 2012; Ito et al. 2012) (Fig. 5.1).
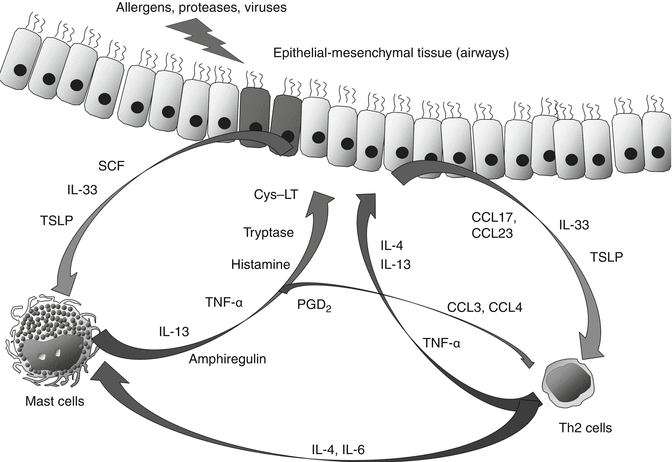

Fig. 5.1
Mutual stimulatory effect of mast cells, Th2 cells and epithelial-mesenchymal tissue on chronic allergic inflammation. These cell types are stimulating each other by releasing cytokines and mediators to form allergic inflammation in the airway. Among such cytokines, IL-13, IL-4 and IL-33 play a key role
Although human MCs do not normally produce cytokines in response to other cytokines such as IL-4 without FcεRI cross-linking, it should be noted that IL-33, which are released during necrosis of epithelial-mesenchymal tissue, alone stimulate MCs to release a variety of cytokines such as IL-13 (Iikura et al. 2007). Regarding other innate immune responses, mouse MCs are proven to play an essential role in protection against microbial infection via Toll-like receptor (TLR)s (Supajatura et al. 2002; Nakajima et al. 1997; Krämer et al. 2008). Human MCs can express functional TLR4 after preincubation with IFN-γ. These MCs can produce more TNF-a, CCL5, CXCL10, and CXCL11 compared to IgE dependently activated MCs (Okumura et al. 2003).
Topical use of glucocorticoid (GC) is the first line therapy for allergic diseases such as asthma and allergic rhinitis. Although GC do not block the degranulation of MCs, these drugs downregulate the gene expression of FcεRI in MCs and thereby downregulate IgE-mediated activation of MCs. More notably, glucocorticoid can inhibit gene expression of a variety of cytokines in MCs. Even in short time incubation, GC blocks the nuclear factor-κB (NF-κB)-dependent gene expression of cytokines, such as IL-13, CXCL8/IL-8, and GM-CSF. On the other hand, GC does not inhibit nuclear factor-activated T (NFAT)-dependent gene expression of cytokines, such as CCL1, CCL3, and CCL4.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


