Fig. 6.1
The mechanisms of host defense system from innate to adaptive immunity. PMN polymorphonuclear cells, CTL cytotoxic lymphocytes, Mo macrophages, DC dendritic cells
6.1.3 Recruitment of Macrophages into Peripheral Mucosal Inflammatory Sites
Macrophages are recruited into peripheral mucosal inflammatory sites with a wide range of different stimuli. If microbial infection takes place, neutrophil infiltration precedes and releases toxic agents designed to kill extracellular pathogens, and then macrophages come and evacuate degradated pathogens and apoptotic neutrophils. The tissue-entering process of these cells is called as chemotaxis. Chemokines are essential for the recruitment of inflammatory cells into the peripheral mucosal inflammatory sites (Sallusto and Baggiolini 2008). Chemokines are subdivided, based on the core cysteine motifs that form disulfide bonds to fold the molecule. CC chemokines have two adjacent cysteines, while in CXC chemokines, there is an intervening amino acid. Chemokine receptors are classified in accordance, CCR, CXCR, and CX3CR families. Expression of specific chemokine receptors on different populations of macrophages and dendritic cells provides different kinds of effector mechanism for their differential recruitment in response to different signals and, consequently, might modify inflammatory reaction in mucosal sites such as respiratory or digestive tract.
6.1.4 Phagocytosis
Phagocytosis is a front-line defense against pathogen attack, so almost by definition, a pathogen is an infectious agent that avoids being killed by phagocytosis. Phagocytosis is a process that requires a mechanism for self-nonself discrimination (Aderem and Underhill 1999). Macrophages possess numerous receptors that allow the direct recognition of particles based upon novel sugars, lipids, protein sequences, and concentrations of charge that are unique to pathogens (so-called pathogen-associated molecular patterns) (Fig. 6.2a, b). Particles may also be recognized indirectly if they are coated with opsonins such as specific antibodies or complement components.
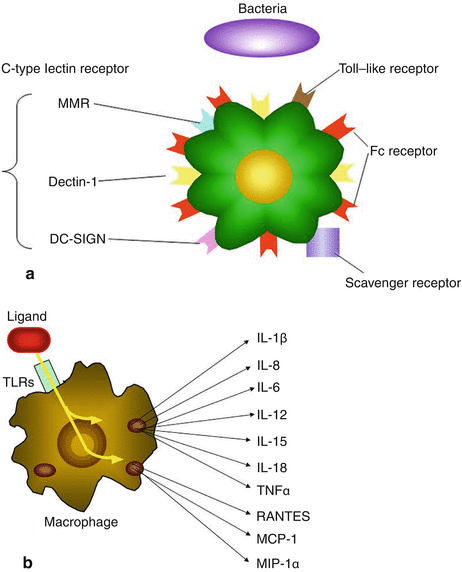

Fig. 6.2
(a) Pattern recognition receptors on macrophages. (b) Cytokines and chemokines produced by macrophage via Toll-like receptors
6.1.5 Antigen Presentation by Macrophages and Dendritic Cells
It is generally accepted that antigens derived from extracellular sources must be taken up, processed by macrophages (phagocytic antigen-presenting cells) and dendritic cells (nonphagocytic or much less-phagocytic antigen-presenting cells), and afterwards presented to T lymphocytes (Fig. 6.3). The process of uptake, processing, and presentation is now well understood (Jutras and Desjardins 2005; Hume 2008). However, recognition of the antigen major histocompatibility complex (MHC)-II complex by the T cell receptor is not sufficient to trigger T cell activation. Moreover, T cell activation needs second co-stimulatory signals from the APC in the form of specific cytokines and coreceptors.
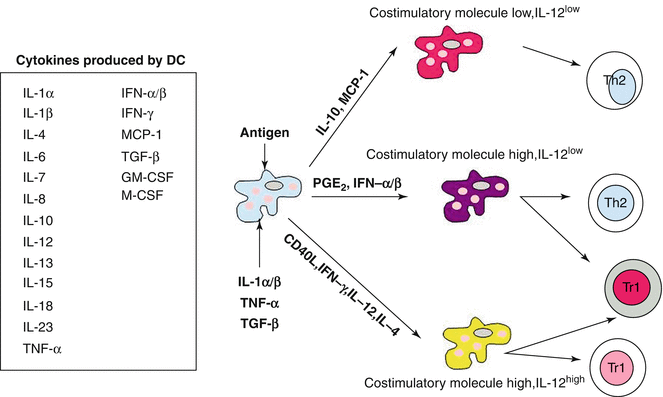

Fig. 6.3
Schematic function of dendritic cells
6.1.6 Macrophage Activation
Activated macrophages are strongly positive for class II-MHC molecules and adapted to kill microorganisms and tumor cells and present antigen to T lymphocytes. The classical macrophage activating factor, produced by stimulated Th1 lymphocytes and NK cells, is interferon-gamma (Schroder et al. 2004). Classical macrophage activation, involving a synergistic interaction between interferon-gamma and a pathogen molecule such as lipopolysaccharide (LPS), is just one of the numerous interactions that occur between distinct stimuli. We know that LPS acts on macrophages to initiate a cascade of inflammatory processes that are essential for innate immunity in upper respiratory tract such as middle and inner ear as well (Kawauchi et al. 1998; Kawai and Akira 2010). T cell products are, of course, only part of the story of macrophage activation. Macrophages respond directly to pathogen-associated molecular patterns (PAMPs). They recognize them through the plasma membrane and cytoplasmic receptors such as the Toll-like receptors and intracellular receptors of the NOD-like receptor (NLR) family (Chen et al. 2009).
6.1.7 Role of Macrophages in Induction of Immune Tolerance
Immunological tolerance is described as no ability of acquired immunity to respond to specific antigens. Central tolerance induction occurs in the thymus for T cells and the bone marrow for B cells. The main mechanism for central tolerance in T cells is the induction of T cell death. Dendritic cells (DCs) are found in abundance in the thymus, where newly produced T cells are educated to become functional CD4+ T or CD8+ T cells and undergo selection to eliminate clones against self. Low-affinity reactive T cells are positively selected and allowed to survive and reach the periphery. The mechanism of peripheral tolerance is a little different from central one but includes T cell death, anergy, and active suppression by regulatory T cells (Tregs). In this mechanism, DCs could contribute by inducing apoptosis in T cells and by producing IL-10 that induces Tregs (Lipscomb and Masten 2002).
6.2 Part II. Distribution of Macrophages in Murine and Human Nasal Mucosa
There are so many reports as regards the actual distribution of macrophages and dendritic cells in murine and human nasal mucosa, by employing immunohistochemistry with various specific antibodies to those cells. Ichimiya and Kawauchi reported in their article that in nasal mucosa of conventional (CV) mice, Mac-1 positive macrophages, mast cells, and all cell types of lymphocyte subsets were present (Ichimiya et al. 1991). But, in the nasal mucosa of specific-pathogen-free mice, all cell types were fewer in number than those of CV mice. And they concluded that macrophages and lymphocytes are mobilized to nasal mucosa, responding to continuous antigenic stimuli, and play an important role in the local defense mechanism of the upper respiratory tract. The analysis of macrophages in human nasal mucosa is abundant, and all published articles demonstrated the significant contribution of macrophages to provoke immune responses and control inflammation in nasal mucosa. Albegger investigated to find out macrophages and lymphocytes in cluster formation of human nasal polyps, employing a light and electron microscopy (Albegger 1977). And his data indicated that cell clusters were consisted mainly of macrophages and lymphoid cells. In their study, within the clusters, the cells showed intimate physical contacts being performed by microvilli with varying length, suggesting cell-to-cell interaction. These cell clusters may remind us of morphologically those found in vitro and in vivo in the course of immune responses. Jahnsen et al. in their histological study of human nasal mucosa demonstrated the dense network of human leukocyte antigen-DR+ cells with dendritic morphology not only in the epithelium but also in the lamina propria (Jahnsen et al. 2004). In addition, they also reported that, in both compartments, these cells could be divided into two main populations based on their phenotypic characteristics: the majority expressed a macrophage-like phenotype (CD11b+CD14+CD64+CD68+RFD7+), whereas the smaller population was predominantly constituted by CD1c+CD11c+ immature DCs. Krysko and Bachert aimed to determine macrophage phenotypes in nasal mucosa of chronic rhinosinusitis with nasal polyp (CRSwNP) and chronic rhinosinusitis without polyp (CRSsNP) and to examine phagocytosis of Staphylococcus aureus (S. aureus) in these pathologies (Krysko et al. 2011). They reported that more M2 macrophages were present in CRSwNP than in CRSsNP. This also was positively correlated with increased levels of IL-5, ECP, and locally produced IgE and decreased levels of IL-6, IL-1β, and IFN-γ. In their study, phagocytosis of S. aureus by human tissue-derived macrophages was reduced in CRSwNP as compared to macrophages from the control inferior turbinates. Furthermore, they concluded that decreased phagocytosis of S. aureus and an M2 activation phenotype in CRSwNP could potentially contribute to the persistence of chronic inflammation in CRSwNP.
6.3 Part III. Modification of Macrophages and Dendritic Cells and Its Clinical Impact on Inflammatory Disorders Such as Allergic Rhinitis
In this part, I would like to introduce a couple of our experimental data in mice as regards how macrophages or dendritic cells are modifying the sinonasal inflammation such as allergic rhinitis and rhinosinusitis. Mature DCs are established as unrivaled APCs in the initiation of immune responses, whereas steady-state DCs are demonstrated to induce peripheral T cell tolerance and consequently attenuate autoimmune-mediated inflammation in animal experiments (Yogev et al. 2012; Mizuno et al. 2012).
In our series of animal experiments, macrophage activation with OK-432 and its effect on allergic rhinitis (Kawauchi et al. 2009) and regulatory role of lymphoid chemokines CCL19 and CCL21 in the control of allergic rhinitis (Takamura et al. 2007; Kawauchi et al. 2011) are introduced as examples, in order to explain how macrophages or DCs modify the sinonasal inflammation such as allergic rhinitis and rhinosinusitis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


